 Back
BackDrug Metabolism: Principles, Mechanisms, and Pathways
Study Guide - Smart Notes

Drug Metabolism
Introduction to Drug Metabolism
Drug metabolism refers to the chemical modifications that drug molecules undergo in the body, primarily to enhance their elimination. These transformations can activate, inactivate, detoxify, or sometimes produce toxic metabolites. Metabolic reactions are categorized into Phase I (functionalization) and Phase II (conjugation) processes.
Phase I metabolism: Involves oxidation, reduction, or hydrolysis to introduce or expose functional groups, often increasing water solubility.
Phase II metabolism: Involves conjugation with endogenous molecules (e.g., glucuronic acid, sulfate, amino acids) to further increase water solubility and facilitate excretion.
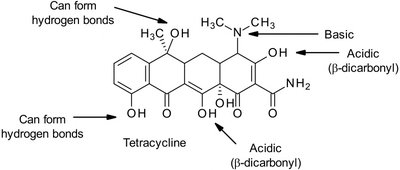
Some drugs, such as tetracycline, are sufficiently water-soluble and can be excreted unchanged due to their ionizable and hydrogen-bonding groups.
Sites of Drug Metabolism
The liver is the primary organ for drug metabolism, but enzymes are also present in the GI tract, kidney, lung, plasma, CNS, skin, placenta, and fetus. Both human and bacterial enzymes in the GI tract can metabolize drugs.
Phase I Metabolism: Oxidation
Oxidation is the most common Phase I reaction, primarily catalyzed by the cytochrome P450 (CYP450) enzyme family. These enzymes introduce oxygen into various functional groups, increasing polarity and water solubility.
CYP450 enzymes: Contain a heme-iron complex and are classified by family, subfamily, and gene (e.g., CYP2D6).
Major families for drug metabolism: CYP1, CYP2, CYP3.
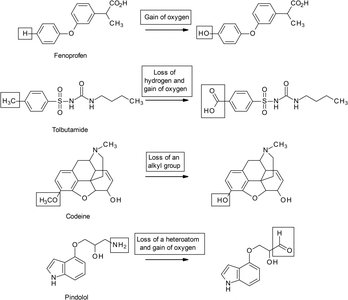
Oxidation can involve gain of oxygen, loss of hydrogen, loss of alkyl group, or loss of heteroatom.
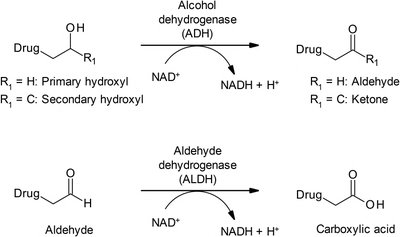
Other Oxidative Enzymes
Flavin monooxygenase (FMO): Oxidizes nitrogen and sulfur atoms in drugs.
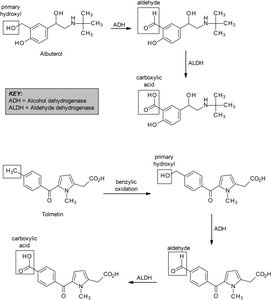
Alcohol dehydrogenase (ADH): Oxidizes alcohols to aldehydes/ketones.
Aldehyde dehydrogenase (ALDH): Oxidizes aldehydes to carboxylic acids.
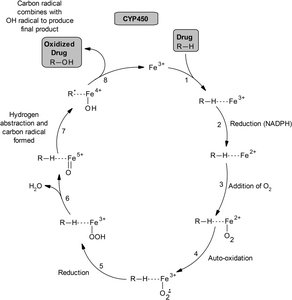
Mechanism of CYP450 Oxidation
The CYP450 catalytic cycle involves substrate binding, reduction, oxygen activation, and transfer of an oxygen atom to the drug.
Oxidation of Hydrocarbon Rings and Chains
Aromatic hydroxylation: Occurs mainly at the para position of unsubstituted phenyl rings; electron-withdrawing groups decrease reactivity.
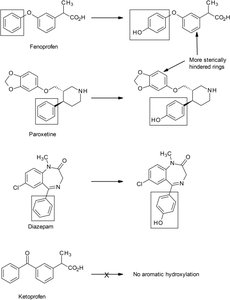
Ortho hydroxylation: Can occur adjacent to existing phenolic groups, forming catechols.

Alkene oxidation: Requires a hydrogen on the alkene carbon; forms epoxides, diols, or peroxides.
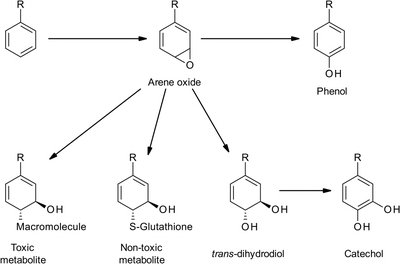
Arene oxide intermediates: Can rearrange to phenols, react with glutathione, or form toxic/carcinogenic metabolites.
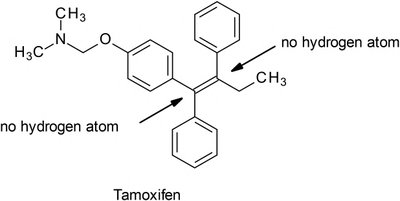
Oxidation at Benzylic, Allylic, and Adjacent Carbons
Benzylic oxidation: Occurs at aliphatic carbons attached to aromatic rings, favoring least hindered positions.
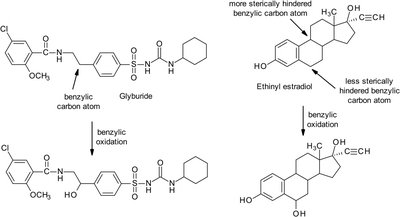
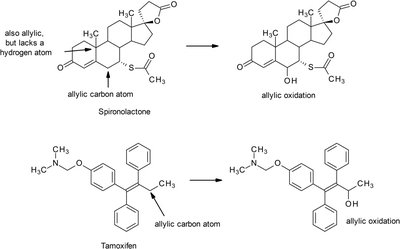
Allylic oxidation: Occurs at carbons adjacent to double bonds, requiring an available hydrogen.
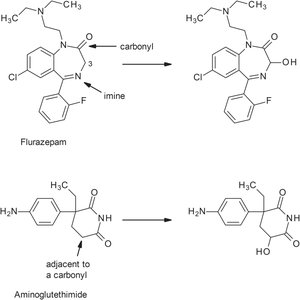
Oxidation adjacent to imines/carbonyls: Adds hydroxyl groups, often at prochiral centers, generating new chiral centers.
Oxidation of Aliphatic and Alicyclic Carbons
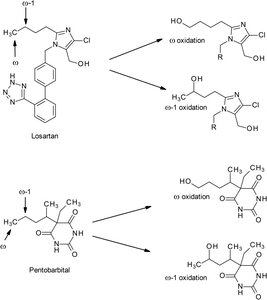
ω and ω-1 oxidation: Occurs at terminal and penultimate carbons of alkyl chains.
Nonaromatic ring oxidation: Occurs at least hindered positions (e.g., C3 or C4 of cyclohexane).
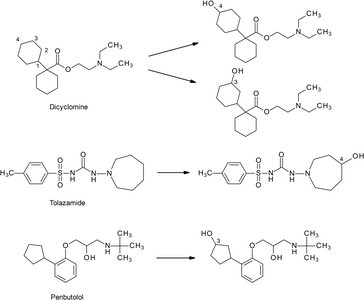
Oxidation of Amines and Amides
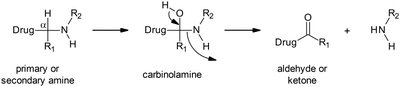
Oxidative deamination: Removes amine groups, forming aldehydes or ketones.
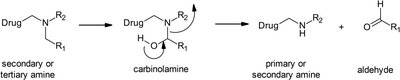
Oxidative N-dealkylation: Removes alkyl groups from amines/amides, forming smaller amines and aldehydes/ketones.
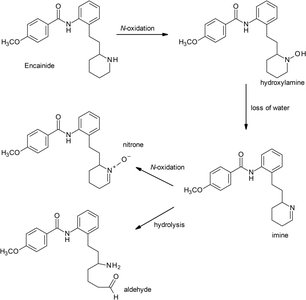
N-oxidation: Direct oxidation of nitrogen atoms, forming N-oxides, hydroxylamines, or nitroso groups.
Oxidation of Functional Groups with Oxygen or Sulfur
Alcohols: Primary alcohols oxidized to aldehydes, then carboxylic acids; secondary alcohols to ketones.
Ethers: Undergo O-dealkylation, forming alcohols and aldehydes/ketones.
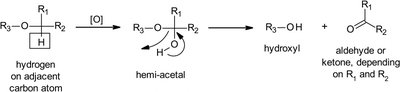
Thioethers: Undergo S-dealkylation or S-oxidation to sulfoxides/sulfones.
Phase I Metabolism: Reduction and Hydrolysis
Reduction: Less common; involves gain of hydrogen, catalyzed by reductases (e.g., nitroreductase, azoreductase).
Hydrolysis: Cleavage of esters, amides, lactones, and lactams by addition of water, forming more polar metabolites.
Phase II Metabolism: Conjugation
Phase II reactions attach endogenous molecules to functional groups, greatly enhancing water solubility and excretion. The main conjugation pathways are:
Glucuronic acid conjugation: Most common; catalyzed by UDP-glucuronyltransferase (UGT).
Sulfate conjugation: Catalyzed by sulfotransferase (SULT); mainly for phenols.
Amino acid conjugation: Glycine or glutamine added to carboxylic acids.
Glutathione conjugation: Detoxifies electrophilic intermediates; catalyzed by glutathione S-transferase.
Acetylation: Acetyl CoA donates acetyl group to amines/hydrazines; catalyzed by N-acetyltransferase.
Methylation: S-adenosylmethionine (SAM) donates methyl group; catalyzed by methyltransferases.
Factors Affecting Drug Metabolism
Genetic polymorphisms: Variations in CYP450 and other enzymes affect drug response and toxicity.
Age, disease, nutrition: Liver function, age, and nutritional status can alter metabolism rates.
Dose and drug interactions: High doses can saturate pathways; co-administered drugs can induce or inhibit enzymes.
Summary Table: Major CYP450 Families and Example Drugs
CYP Family | Example Isoforms | Example Substrates |
|---|---|---|
CYP1 | CYP1A1, CYP1A2 | Polycyclic aromatic hydrocarbons, haloperidol |
CYP2 | CYP2A6, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6, CYP2E1 | Clozapine, amitriptyline, paclitaxel, diazepam, esomeprazole, codeine, ethanol |
CYP3 | CYP3A4, CYP3A5, CYP3A7 | Erythromycin, clopidogrel, retinoic acid |
Key Points
Drug metabolism enhances elimination by increasing water solubility.
Phase I reactions introduce or expose functional groups; Phase II reactions conjugate endogenous molecules.
Metabolic pathways are influenced by drug structure, enzyme accessibility, and genetic/physiological factors.
Some drugs are excreted unchanged; others require multiple metabolic steps.
