 Back
BackEar Disorders: Cerumen Impaction and Hearing Aids
Study Guide - Smart Notes

Ear Disorders
Introduction to Ear Disorders
Ear disorders related to cerumen (earwax) and hearing loss are common clinical concerns. Understanding the physiology of cerumen, causes and symptoms of impaction, and the safe use of hearing aids is essential for effective patient care and self-management.
Cerumen (Earwax)
Role and Benefits of Cerumen
Cerumen is a natural substance produced in the ear canal that serves several protective functions:
Protective barrier: Shields the ear canal from dust, microorganisms, and foreign objects.
Traps foreign objects: Prevents debris from reaching the tympanic membrane.
Reduces risk of infection: Possesses antimicrobial properties that help prevent infections.
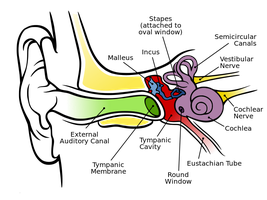
Causes of Excessive or Impacted Cerumen
Excessive or impacted cerumen can result from anatomical, behavioral, or device-related factors:
Excessive cerumen: Abnormally narrow or misshapen ear canals, excessive hair growth, or overactive ceruminous glands.
Impacted cerumen: Atrophy of ceruminous glands, use of hearing aids, ear plugs, airpods/ear buds, or cotton-tipped swabs.
Symptoms of Excessive or Impacted Cerumen
Common symptoms include:
Ear fullness, dull pain, itching, discomfort
Tinnitus (ringing in the ears), dizziness, vertigo, cough
Hearing loss (may lead to impaired cognition in older adults)
Hearing aid malfunction or feedback
Discharge and odor
When to Refer Patients
Referral to a healthcare provider is necessary if the patient presents with:
Signs of infection (pain with ear discharge)
Bleeding, trauma, or ruptured tympanic membrane
Recent ear surgery (within 6 weeks) or tympanostomy tubes
Inability to follow instructions
Children under 12 years of age
Cerumen Removal: Agents and Methods
FDA-Approved Cerumen Softening Agent
The only FDA-approved cerumen softening agent is carbamide peroxide (6.5%). It is available in several over-the-counter (OTC) products.

Mechanism of Action and Directions for Use
Carbamide peroxide works by releasing oxygen, creating a foaming action that softens and loosens earwax, making it easier to remove.
For adults and children over 12 years: Tilt head sideways, place 5–10 drops into the ear, keep drops in for several minutes, use twice daily for up to four days.
Do not insert the applicator tip into the ear canal.
Any remaining wax may be removed by gently flushing the ear with warm water using a soft rubber bulb ear syringe.
Children under 12 years: Consult a doctor.

Role of the Rubber Bulb Ear Syringe
The rubber bulb ear syringe is used to gently wash the ear with warm water after using the earwax removal solution, helping to remove any softened or loosened wax.
Ear Candling: Risks and Dangers
Ear candling is an alternative practice that involves placing a hollow candle in the ear canal and lighting it. This method is dangerous and not recommended due to risks of burns, ear canal obstruction, and tympanic membrane perforation.

Hearing Aids
OTC Hearing Aids
In August 2022, the FDA established a new category of over-the-counter (OTC) hearing aids for adults (18+) with mild to moderate hearing loss. These devices are intended for individuals who experience:
Muffled speech or sounds
Trouble hearing in groups, noisy areas, or on the phone
Need to ask others to repeat themselves or speak louder
Preference for higher volume on TV/radio than others
OTC hearing aids are not the same as personal sound amplification products (PSAPs), which are not FDA-approved medical devices.





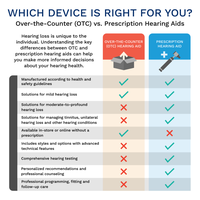
OTC vs Prescription Hearing Aids
The main differences between OTC and prescription hearing aids are summarized below:
Feature | OTC Hearing Aids | Prescription Hearing Aids |
|---|---|---|
Manufactured according to health and safety guidelines | ✔️ | ✔️ |
Solutions for mild hearing loss | ✔️ | ✔️ |
Solutions for moderate-to-profound hearing loss | ❌ | ✔️ |
Individually programmed, customized fit | ❌ | ✔️ |
Available in-store or online without a prescription | ✔️ | ❌ |
Comprehensive hearing testing | ❌ | ✔️ |
Professional recommendations and fitting | ❌ | ✔️ |
Exclusions for OTC Hearing Aids
OTC hearing aids are not appropriate for individuals with:
Fluid, pus, or blood discharge from the ear within the past 6 months
Pain or discomfort in the ear
History of excessive earwax or suspicion of a foreign body in the ear canal
Episodes of vertigo with hearing loss
Sudden or rapidly worsening hearing loss
Hearing loss or tinnitus in only one ear, or a noticeable difference between ears
Consumer Ear Disease Risk Assessment
Patients can use online resources such as the Consumer Ear Disease Risk Assessment (CEDRA) to help determine their risk for ear disease and appropriateness for OTC hearing aids.
Summary Table: Cerumen and Hearing Aid Management
Condition | Symptoms | Self-Care | When to Refer |
|---|---|---|---|
Excessive/Impacted Cerumen | Fullness, pain, itching, hearing loss, tinnitus | Carbamide peroxide drops, ear irrigation (if appropriate) | Infection, trauma, children <12, recent surgery, unable to follow instructions |
Hearing Loss (Mild-Moderate) | Muffled speech, difficulty in noise, increased TV volume | OTC hearing aids (adults >18, no exclusions) | Sudden loss, unilateral symptoms, discharge, pain, vertigo |
