 Back
BackFluid, Electrolyte, and Acid-Base Balance: Buffer Systems and Clinical Acid-Base Disorders
Study Guide - Smart Notes

Chapter 26: Fluid, Electrolyte, and Acid-Base Balance
Acids and Bases
This section introduces the fundamental concepts of acids and bases, which are crucial for understanding physiological pH regulation in the human body.
Acids: Proton donors; they dissociate in aqueous solution to liberate free hydrogen ions (H+).
Strong acids: Completely dissociate in solution (e.g., HCl, HNO3).
Weak acids: Only partially dissociate (e.g., HCO3-, H2CO3, H3PO4, NH4+).
Bases: Proton acceptors; they dissociate in aqueous solution and bind free hydrogen ions.
Strong bases: Completely dissociate in solution (e.g., NaOH).
Weak bases: Only partially dissociate (e.g., NH3).
Some bases liberate hydroxide ions, while others accept hydrogen ions.
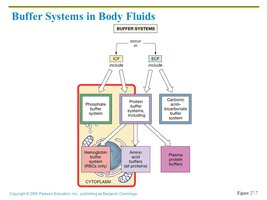
Chemical Buffer Systems
Buffer systems are essential for resisting changes in the pH of body fluids. They typically consist of a weak acid and its conjugate base, and they function by releasing or binding H+ as needed.
Three major buffer systems in the body:
Bicarbonate buffer system: Buffers extracellular fluid.
Phosphate buffer system: Buffers intracellular fluid.
Protein buffer system: Buffers both intracellular and some extracellular fluid (e.g., hemoglobin, albumin).
Phosphate Buffer System
The phosphate buffer system is important in intracellular fluid and renal tubular fluid. It operates similarly to the bicarbonate system but uses phosphate ions.
Components: NaH2PO4 (weak acid) and Na2HPO4 (weak base).
When a strong acid is added, the reaction shifts left, forming NaH2PO4 and NaCl (strong acid replaced by weak acid).
When a strong base is added, the reaction shifts right, forming Na2HPO4 and H2O (strong base replaced by weak base).
Protein Buffer System
Proteins are the most abundant and powerful buffers in the body, especially in intracellular fluid and blood plasma. Their amino acid side chains can act as acids or bases.
Carboxyl (COOH) groups can release H+.
Amino (NH3) groups can bind H+.
Amphoteric proteins can act as both weak acids and weak bases (e.g., hemoglobin).
Bicarbonate Buffer System
The bicarbonate buffer system is the primary buffer in blood plasma. It consists of carbonic acid (H2CO3) and bicarbonate (HCO3-).
When a strong acid is added, the reaction shifts left, forming H2CO3 and NaCl.
When a strong base is added, the reaction shifts right, forming NaHCO3 and H2O.
This system is biased toward buffering excess acid and is tightly regulated by the kidneys.
Physiological Buffering System
The bicarbonate buffering system is clinically significant and is governed by the law of mass action. The reaction is reversible and is regulated by both the lungs and kidneys.
Equation:
Lungs control CO2 levels; kidneys control HCO3- levels.
Respiratory Regulation
The lungs regulate acid-base balance by adjusting the rate and depth of ventilation, which alters CO2 excretion and thus blood pH.
Doubling respiratory rate increases blood pH by 0.2 units.
Hypoventilation causes acidosis; hyperventilation causes alkalosis.
Renal Regulation
The kidneys provide a permanent solution to acid-base imbalances by excreting fixed acids, renewing chemical buffers, and regulating HCO3- and H+ levels.
Key mechanisms: HCO3- conservation, generation of new HCO3-, and HCO3- secretion.
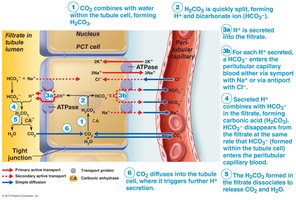
HCO3- Conservation
Occurs in proximal convoluted tubule (PCT) cells via an indirect mechanism involving carbonic anhydrase. HCO3- is reabsorbed into the blood, often coupled with Na+ or exchanged for Cl-.
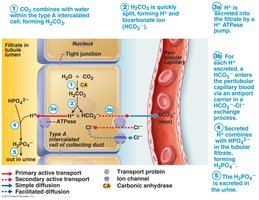
Generating New HCO3-
Type A intercalated cells in the distal convoluted tubule (DCT) and collecting ducts generate new HCO3- and excrete H+ into the filtrate, buffered by HPO42- ions.
Glutamine Mechanism
In the proximal convoluted tubule, glutamine is metabolized to produce NH4+ (excreted in urine) and new HCO3- (reabsorbed into blood).
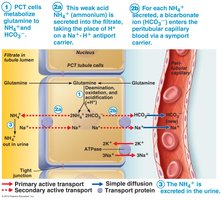
HCO3- Secretion
Type B intercalated cells secrete HCO3- into the filtrate and reabsorb H+ into the blood, which is important for correcting alkalosis.
Effects of Acidosis and Alkalosis
Maintaining blood pH within the narrow range of 7.35–7.45 is critical for life. Deviations can have severe effects on the nervous and muscular systems.
Acidosis (pH < 7.35): CNS depression, coma, death; increased blood Ca++.
Alkalosis (pH > 7.45): Neuromuscular over-excitability, tetany, convulsions, arrhythmias; decreased blood Ca++.
Acid-Base Disturbances
Acid-base disorders are classified by their origin (respiratory or metabolic) and their effect on pH, pCO2, and HCO3- levels.
Respiratory acidosis: ↓ pH, ↑ pCO2
Respiratory alkalosis: ↑ pH, ↓ pCO2
Metabolic acidosis: ↓ pH, ↓ HCO3-
Metabolic alkalosis: ↑ pH, ↑ HCO3-
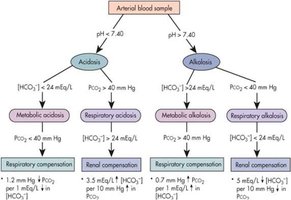
Respiratory and Renal Compensation
When one system (respiratory or renal) is the cause of an acid-base imbalance, the other system compensates to restore pH balance.
Metabolic disorders are compensated by respiratory changes.
Respiratory disorders are compensated by renal changes.
Clinical Diagnosis and Normal Ranges
Diagnosis of acid-base disorders relies on measuring pH, pCO2, and HCO3- in arterial blood.
Normal pH: 7.35–7.45 (mean 7.40)
Normal pCO2: 35–45 mmHg (mean 40)
Normal HCO3-: 22–26 mEq/L (mean 24)
Acid-Base Disorders: Causes and Compensation
Respiratory Acidosis: Caused by hypoventilation (e.g., COPD, drug overdose). Compensation: Renal retention of HCO3-.
Respiratory Alkalosis: Caused by hyperventilation (e.g., anxiety, high altitude). Compensation: Renal excretion of HCO3-.
Metabolic Acidosis: Caused by increased acid or loss of HCO3- (e.g., diarrhea, renal failure). Compensation: Hyperventilation to decrease pCO2.
Metabolic Alkalosis: Caused by loss of acid or excess HCO3- (e.g., vomiting, diuretics). Compensation: Hypoventilation to increase pCO2.
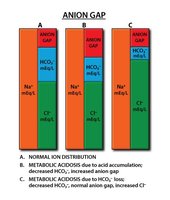
Anion Gap Analysis
The anion gap helps determine the cause of metabolic acidosis by comparing the concentrations of major cations and anions in plasma.
Calculation:
Normal anion gap: ~10–12 mEq/L
High anion gap: >12 mEq/L (e.g., MUDPILES mnemonic for causes)
Normal anion gap: 10–12 mEq/L (e.g., SAD CRASH mnemonic for causes)
Low anion gap: <10 mEq/L (e.g., hypoalbuminemia)
Example Calculation
Given: [Na+] = 140 mEq/L, [Cl-] = 102 mEq/L, [HCO3-] = 24 mEq/L
Calculation: mEq/L
Summary Table: Acid-Base Disorders
Disorder | pH | pCO2 | HCO3- | Compensation |
|---|---|---|---|---|
Respiratory Acidosis | ↓ | ↑ | ↑ | Renal |
Respiratory Alkalosis | ↑ | ↓ | ↓ | Renal |
Metabolic Acidosis | ↓ | ↓ | ↓ | Respiratory |
Metabolic Alkalosis | ↑ | ↑ | ↑ | Respiratory |
Acid-Base Nomogram
The acid-base nomogram is a clinical tool for visualizing and diagnosing acid-base disorders based on pH and pCO2 values.

Normal Ranges (Bonus Cheat Sheet)
pH: 7.35–7.45 (mean 7.40)
pCO2: 35–45 mmHg (mean 40)
HCO3-: 22–26 mEq/L (mean 24)
