 Back
BackFluid, Electrolyte, and Acid-Base Balance: ANP Study Notes
Study Guide - Smart Notes

Fluid, Electrolyte, and Acid-Base Balance
Introduction
Fluid, electrolyte, and acid-base balance are essential for maintaining homeostasis in the human body. These processes regulate the distribution and composition of body fluids, the concentration of electrolytes, and the pH of body fluids, all of which are critical for normal cellular function and overall health.
Body Fluid Compartments
Body Water Content
Infants: ~73% or more water (low body fat, low bone mass)
Adult males: ~60% water
Adult females: ~50% water (higher fat content, less skeletal muscle mass)
Adipose tissue: least hydrated of all tissues
Total body water in adults: ~40 L
Water content declines to ~45% in old age
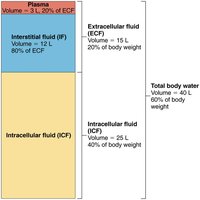
Main Fluid Compartments
Intracellular fluid (ICF): Fluid inside cells; accounts for 2/3 of total body fluid (~25 L)
Extracellular fluid (ECF): Fluid outside cells; accounts for 1/3 of total body fluid (~15 L)
Plasma: 3 L (20% of ECF)
Interstitial fluid (IF): 12 L (80% of ECF)
Other ECF: lymph, cerebrospinal fluid, synovial fluid, serous fluid, gastrointestinal secretions
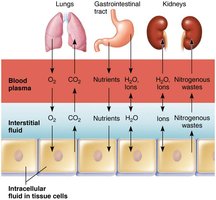
Exchange Between Compartments
Osmotic and hydrostatic pressures regulate continuous exchange and mixing of fluids.
Water moves freely along osmotic gradients; all body fluid osmolality is almost always equal.
Exchanges between plasma and IF occur across capillary walls; exchanges between IF and ICF occur across cell membranes.
Composition of Body Fluids
Electrolytes and Nonelectrolytes
Nonelectrolytes: Most are organic molecules (e.g., glucose, lipids, creatinine, urea); do not dissociate in water; no charged particles created.
Electrolytes: Dissociate into ions in water (e.g., inorganic salts, acids, bases, some proteins); ions conduct electrical current and have greater osmotic power than nonelectrolytes.
Electrolyte concentrations are expressed in milliequivalents per liter (mEq/L).
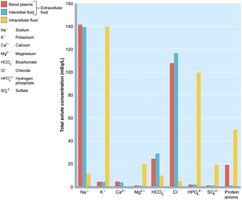
Electrolyte Distribution
ECF: Major cation is Na+; major anion is Cl–.
ICF: Major cation is K+; major anion is HPO42–.
Electrolytes are most abundant solutes in body fluids and determine most chemical and physical reactions.
Bulk of dissolved solutes consists of proteins, phospholipids, cholesterol, and triglycerides.
Water Balance and ECF Osmolality
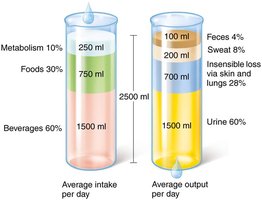
Water Intake and Output
Water intake must equal water output (~2500 ml/day).
Intake: Ingested foods and beverages, metabolic water (from cellular metabolism).
Output: Urine (60%), insensible water loss (skin and lungs), perspiration, feces.
Regulation of Water Intake
Thirst mechanism is the driving force for water intake, governed by the hypothalamic thirst center.
Stimuli: Increased plasma osmolality (1–2%), dry mouth, decreased blood volume/pressure, angiotensin II, baroreceptor input.
Drinking water inhibits the thirst center via relief of dry mouth and activation of stretch receptors in the stomach and intestine.
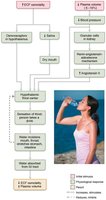
Regulation of Water Output
Obligatory water losses: insensible water loss (lungs, skin), sensible water loss (urine, sweat, feces).
Volume of urine excreted and solute concentration depend on fluid intake, diet, and water loss via other avenues.
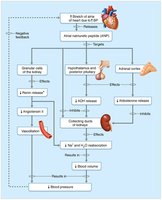
Influence of Antidiuretic Hormone (ADH)
Water reabsorption in collecting ducts is proportional to ADH release.
Decreased ADH: dilute urine, drop in body fluid volume.
Increased ADH: concentrated urine, increased body fluid volume.
Other triggers: large changes in blood volume or pressure, intense sweating, vomiting, diarrhea, severe blood loss, burns, fever.
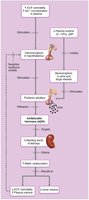
Disorders of Water Balance
Dehydration
ECF water loss due to hemorrhage, burns, vomiting, diarrhea, sweating, water deprivation, diuretic abuse, endocrine disturbances.
Symptoms: dry mouth, thirst, dry flushed skin, oliguria; may lead to weight loss, fever, confusion, hypovolemic shock, electrolyte loss.

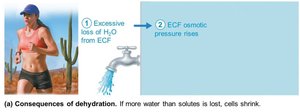
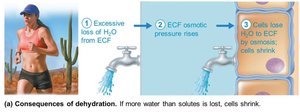
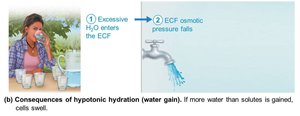
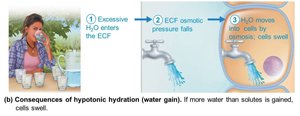
Hypotonic Hydration (Water Intoxication)
Occurs with renal insufficiency or rapid excess water ingestion.
ECF osmolality decreases, causing hyponatremia; water enters tissue cells, causing swelling.
Symptoms: nausea, vomiting, cramping, cerebral edema, possible death; treated with hypertonic saline.

Edema
Atypical accumulation of IF, resulting in tissue swelling (not cell swelling).
Can impair tissue function by increasing diffusion distance for oxygen and nutrients.
Caused by increased fluid flow out of blood or decreased return of fluid to blood.
Electrolyte Balance
Overview
Electrolyte balance usually refers to salt balance (mainly NaCl), though electrolytes include acids, bases, and some proteins.
Salts control fluid movements, provide minerals for excitability, secretory activity, and membrane permeability.
Salts enter by ingestion/metabolism and are lost via perspiration, feces, urine, vomit.
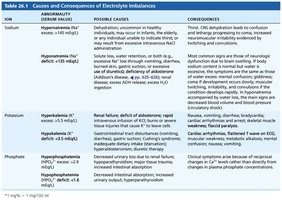
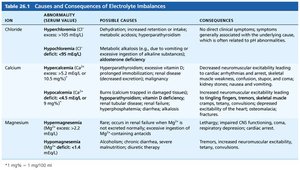
Causes and Consequences of Electrolyte Imbalances
The following tables summarize the main electrolyte imbalances, their causes, and consequences:
Ion | Abnormality | Possible Causes | Consequences |
|---|---|---|---|
Sodium | Hypernatremia, Hyponatremia | Dehydration, excessive water loss, SIADH, etc. | Thirst, confusion, neuromuscular irritability, CNS symptoms |
Potassium | Hyperkalemia, Hypokalemia | Renal failure, aldosterone deficit, GI disturbances | Bradycardia, arrhythmias, muscle weakness, paralysis |
Phosphate | Hyperphosphatemia, Hypophosphatemia | Renal failure, hypoparathyroidism, malnutrition | Neuromuscular excitability, muscle weakness |
Central Role of Sodium
Sodium is the most abundant cation in ECF; only cation exerting significant osmotic pressure.
Controls ECF volume and water distribution; changes in Na+ affect plasma volume, blood pressure, and ECF/IF volumes.
Na+ concentration determines ECF osmolality and influences excitability of neurons and muscles.
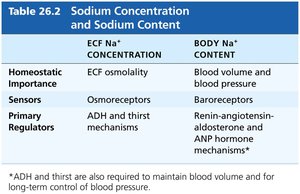
ECF Na+ Concentration | Body Na+ Content | |
|---|---|---|
Homeostatic Importance | ECF osmolality | Blood volume and blood pressure |
Sensors | Osmoreceptors | Baroreceptors |
Primary Regulators | ADH and thirst mechanisms | Renin-angiotensin-aldosterone and ANP hormone mechanisms |
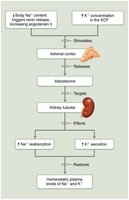
Regulation of Sodium Balance
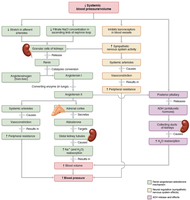
No known receptors monitor Na+ levels directly; regulation is linked to blood pressure and volume control mechanisms.
Aldosterone plays the biggest role in regulation by the kidneys; renin-angiotensin-aldosterone mechanism is the main trigger for aldosterone release.
Other hormones: Atrial natriuretic peptide (ANP) reduces blood pressure and blood volume by inhibiting Na+ and water retention.
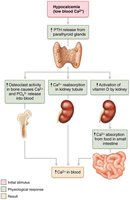
Regulation of Potassium, Calcium, and Phosphate Balance
Potassium: Affects resting membrane potential in neurons and muscle cells; regulated by kidneys, aldosterone, and dietary intake.
Calcium: 99% in bones; regulated by parathyroid hormone (PTH), which increases blood Ca2+ by acting on bones, kidneys, and intestines.
Phosphate: Reabsorbed in the kidneys; regulated by PTH, insulin, and glucagon.
Acid-Base Balance
pH Regulation
Normal arterial blood pH: 7.4; venous blood and IF: 7.35; ICF: 7.0.
Alkalosis: arterial pH > 7.45; Acidosis: arterial pH < 7.35.
Most H+ is produced as a by-product of metabolism.
Buffer Systems
Chemical buffer systems: First line of defense; include bicarbonate, phosphate, and protein buffer systems.
Respiratory system: Eliminates CO2 (an acid); acts within minutes.
Renal mechanisms: Most potent; require hours to days to effect pH changes.
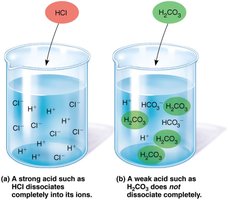

Bicarbonate Buffer System
Mixture of H2CO3 (weak acid) and salts of HCO3– (weak base).
Main ECF buffer; also operates in ICF.
Regulated by the kidneys.
Phosphate Buffer System
Salts of H2PO4– (weak acid) and HPO42– (weak base).
Important buffer in urine and ICF.
Protein Buffer System
Most important buffer in ICF; also in blood plasma.
Proteins are amphoteric (can act as acid or base).
Respiratory Regulation of H+
Respiratory system eliminates CO2 (an acid); reversible equilibrium exists in blood.
Increased PCO2 or H+ stimulates increased respiratory rate and depth, removing CO2 and reducing H+.
Alkalosis depresses respiratory center, decreasing respiratory rate and increasing H+.
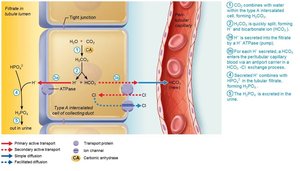
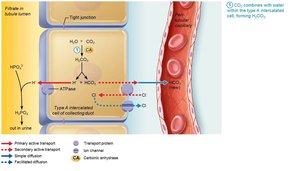
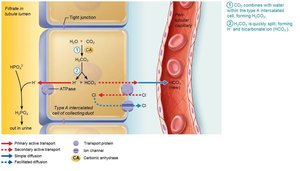
Renal Regulation of Acid-Base Balance
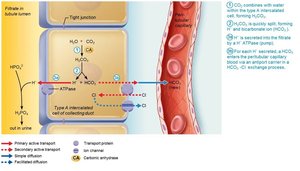
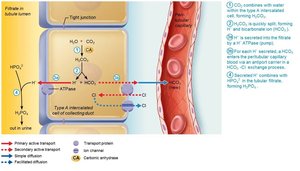
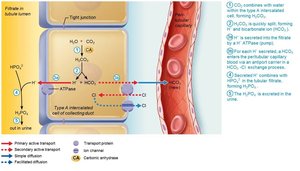
Kidneys regulate acid-base balance by adjusting the amount of bicarbonate in blood.
Conserve (reabsorb) or generate new HCO3–; excrete HCO3– as needed.
Renal regulation depends on the kidney’s ability to secrete or retain H+.
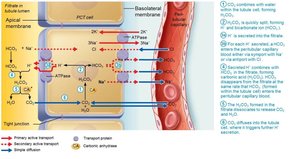
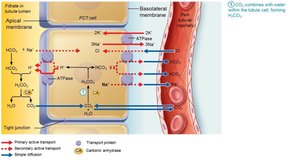
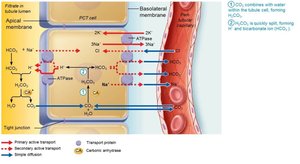
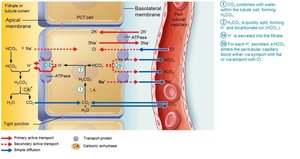
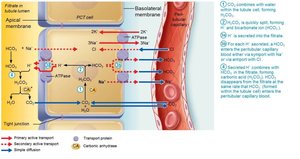
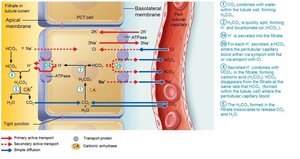
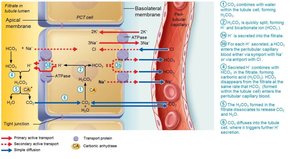
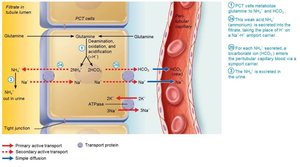
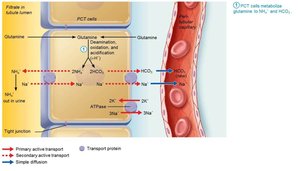
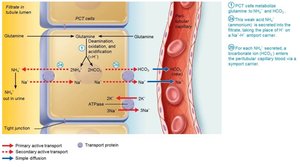
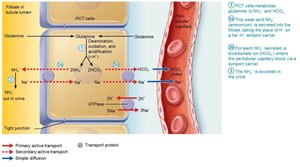
Generating New Bicarbonate Ions
Occurs via excretion of buffered H+ (mainly phosphate buffer system) and via NH4+ excretion (glutamine metabolism).
Abnormalities of Acid-Base Balance
Types of Imbalances
Respiratory acidosis/alkalosis: Caused by failure of the respiratory system to regulate pH (main indicator: blood PCO2).
Metabolic acidosis/alkalosis: All other abnormalities (main indicator: abnormal HCO3– levels).

Compensation Mechanisms
If one physiological buffer system fails, the other compensates (respiratory system compensates for metabolic imbalances, kidneys for respiratory imbalances).
Respiratory compensation: Lungs adjust rate and depth of breathing to correct metabolic pH problems.
Renal compensation: Kidneys adjust bicarbonate levels to correct respiratory pH problems.
Developmental Aspects
Infants have proportionately more ECF than adults until about 2 years of age; higher risk of dehydration and acidosis.
At puberty, males develop greater muscle mass, leading to differences in body water content.
In old age, total body water decreases; homeostatic mechanisms slow down, increasing risk for dehydration and acid-base disturbances.
