 Back
BackFluid, Electrolyte, and Acid-Base Balance: Regulation and Compensation
Study Guide - Smart Notes

Fluid and Acid-Base Balance
Factors Affecting Blood pH
The pH of blood is tightly regulated to maintain homeostasis. Several physiological and external factors influence blood pH:
Carbon dioxide (CO2) and bicarbonate (HCO3-) balance: The ratio of these two determines the acidity or alkalinity of blood.
Respiratory function: The lungs regulate CO2 levels, directly impacting pH.
Metabolic processes: Cellular metabolism produces acids (e.g., lactic acid, ketone bodies) and bases, affecting pH.
Renal (kidney) function: Kidneys control the reabsorption of bicarbonate and excretion of hydrogen ions (H+).
External factors: Diet and medications can alter acid-base balance, though their effect is usually minor compared to metabolic and respiratory influences.
Sources of Acids and Bases in the Body
Acids and bases originate from both metabolic activities and dietary intake:
Metabolic acids:
CO2 (volatile acid, eliminated by lungs and kidneys)
Lactic acid, uric acid, citric acid intermediates, ketone bodies (fixed acids, removed by kidneys)
Dietary sources:
Amino acids (from proteins), fatty acids (from fats), citric acid (from fruits), bicarbonate (from fruits/vegetables)
Additional info: While some advocate for alkaline diets, their impact on systemic acid-base balance is limited due to robust physiological regulation.
Buffer Systems in the Body
Major Buffer Systems
Buffer systems are the first line of defense against pH changes in the body:
Bicarbonate buffer system: The main buffer in blood, involving carbonic acid (H2CO3) and bicarbonate ions (HCO3-).
Phosphate buffer system: Important in intracellular fluid and the kidneys.
Protein buffers: Proteins (e.g., hemoglobin) can bind or release H+ ions, contributing to pH regulation.
These systems work together to neutralize excess acids or bases, maintaining blood pH within a narrow range (7.35–7.45).
Physiological Regulation of Acid-Base Balance
Respiratory Regulation of pH
The respiratory system helps maintain acid-base balance by regulating CO2 levels:
CO2 removal: Increased respiration eliminates more CO2, reducing acidity.
CO2 retention: Decreased respiration retains CO2, increasing acidity.
Interaction with buffers: Works with chemical buffers to stabilize pH.
Renal Regulation of pH
The kidneys are essential for long-term acid-base balance:
Bicarbonate reabsorption: Conserves HCO3- to buffer acids.
Hydrogen ion excretion: Removes H+ from the body.
Generation of new bicarbonate: Produces additional HCO3- as needed.
Mechanisms in the Renal Tubule
In the nephron, several processes regulate acid-base balance:
Hydrogen ion secretion and bicarbonate reabsorption (proximal tubule):
H+ is secreted into the filtrate, where it binds HCO3- to form H2CO3.
Carbonic anhydrase catalyzes the conversion of H2CO3 to CO2 and H2O, which diffuse into the tubule cell.
Inside the cell, CO2 and H2O reform H2CO3, which dissociates into HCO3- (reabsorbed into blood) and H+ (recycled for secretion).
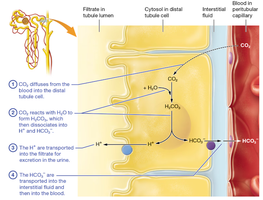
Additional info: This process is crucial for reclaiming filtered bicarbonate and preventing acidosis.
Compensation Mechanisms
Compensation for Acid-Base Disorders
When acid-base imbalances occur, the body uses compensatory mechanisms:
Respiratory compensation: Adjusts ventilation to change CO2 levels (fast response).
Renal compensation: Alters H+ excretion and HCO3- reabsorption (slower, but more powerful).
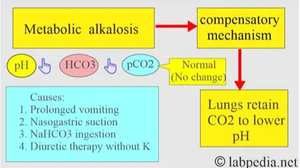
Compensation in Metabolic Alkalosis
Causes: Prolonged vomiting, nasogastric suction, NaHCO3 ingestion, diuretic therapy without potassium.
Compensatory mechanism: Lungs retain CO2 (hypoventilation) to lower pH.
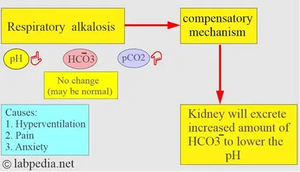
Compensation in Respiratory Alkalosis
Causes: Hyperventilation, pain, anxiety.
Compensatory mechanism: Kidneys excrete more HCO3- to lower pH.
Examples of Compensation
Metabolic acidosis: Compensated by increased respiratory rate (hyperventilation) to expel CO2.
Respiratory acidosis: Compensated by increased renal excretion of H+ and reabsorption of HCO3-.
Key Questions and Applications
How can carbon dioxide become an acid? It reacts with water to produce carbonic acid (H2CO3).
Hyperventilation effect: Leads to an increase in the pH of extracellular fluid (alkalosis).
Compensation for metabolic acidosis: Increase the respiratory rate to expel CO2 and decrease acidity.
Clinical scenario (Mr. Wong): Obstructive lung disease (e.g., pneumonia, asthma) leads to respiratory acidosis (low pH, high PCO2, low HCO3-).
Summary Table: Acid-Base Disorders and Compensation
Disorder | Primary Change | Compensation |
|---|---|---|
Metabolic Acidosis | ↓ HCO3- | ↑ Ventilation (↓ CO2) |
Metabolic Alkalosis | ↑ HCO3- | ↓ Ventilation (↑ CO2) |
Respiratory Acidosis | ↑ CO2 | ↑ Renal HCO3- reabsorption |
Respiratory Alkalosis | ↓ CO2 | ↑ Renal HCO3- excretion |
Key Equations
Bicarbonate buffer equation:
Henderson-Hasselbalch equation:
Additional info: These equations are fundamental for understanding acid-base physiology and interpreting arterial blood gases (ABGs).
