 Back
BackGas Exchange and Breathing: Pulmonary Ventilation, Gas Transport, and Regulation
Study Guide - Smart Notes

Gas Exchange and Breathing
Overview of Pulmonary Ventilation and Gas Exchange
Gas exchange and breathing are essential physiological processes that ensure the delivery of oxygen to tissues and the removal of carbon dioxide from the body. These processes involve the coordinated function of the respiratory and cardiovascular systems, including the movement of gases across the respiratory membrane and their transport in the blood.
Pulmonary and Systemic Circulation
Blood Flow and Gas Transport
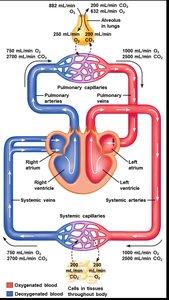
Pulmonary circulation carries deoxygenated blood from the right ventricle to the lungs for gas exchange and returns oxygenated blood to the left atrium.
Systemic circulation delivers oxygenated blood from the left ventricle to body tissues and returns deoxygenated blood to the right atrium.
Oxygen and carbon dioxide are exchanged between alveoli and blood, and between blood and tissues, following their respective partial pressure gradients.
Gas Exchange at the Respiratory Membrane
Structure and Function
The respiratory membrane consists of the alveolar epithelium, fused basement membranes, and capillary endothelium.
Gas exchange occurs by diffusion across this thin barrier, allowing O2 to enter the blood and CO2 to exit.

Partial Pressures and Gas Diffusion
Dalton’s Law of Partial Pressures
Dalton’s Law states that the total pressure exerted by a mixture of gases is equal to the sum of the pressures exerted by each individual gas.
The partial pressure of a gas is calculated as the product of its fractional concentration and the total pressure of the mixture.
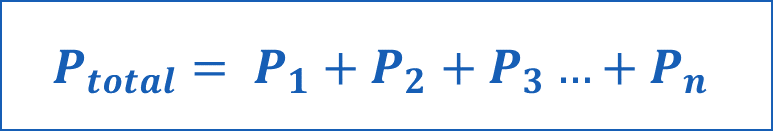
Example: For atmospheric air at sea level (760 mmHg):
Oxygen (O2): 20.93% → PO2 = 0.2093 × 760 mmHg = 159 mmHg
Nitrogen (N2): 79.04% → PN2 = 0.7904 × 760 mmHg = 601 mmHg
Carbon Dioxide (CO2): 0.03% → PCO2 = 0.0003 × 760 mmHg = 0.23 mmHg
Effect of Humidity
Water vapor contributes to the total pressure in humid air, reducing the partial pressures of other gases.
Solubility of Gases in Liquids
Gas Dissolution and Equilibration
Gases dissolve in liquids in proportion to their partial pressures and solubility coefficients.
CO2 is about 20 times more soluble in water than O2.
At equilibrium, the partial pressure of a gas in the liquid equals that in the air, but concentrations differ due to solubility.
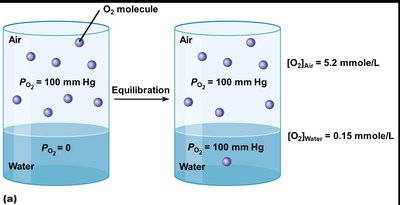
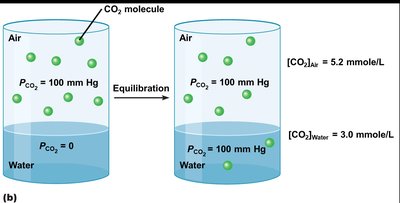
Example: At PO2 = 100 mmHg, [O2]air = 5.2 mmol/L, [O2]water = 0.15 mmol/L; [CO2]air = 5.2 mmol/L, [CO2]water = 3.0 mmol/L.
Partial Pressures at Different Body Sites
Typical Values
The partial pressures of O2 and CO2 vary at different sites in the body, reflecting ongoing gas exchange.
Site | Oxygen (mm Hg) | Carbon Dioxide (mm Hg) |
|---|---|---|
Atmospheric air | 160 | 0.3 |
Alveolar air | 100 | 40 |
Pulmonary veins | 100 | 40 |
Systemic arteries | 100 | 40 |
Cells | ≤40 | ≥46 |
Systemic veins | 40 | 46 |
Pulmonary arteries | 40 | 46 |

Determinants of Alveolar Gas Pressures
Factors Affecting Alveolar PO2 and PCO2
Partial pressures of inspired air
Alveolar ventilation rate
Rate of tissue O2 consumption and CO2 production
The most important determinant is the ratio of alveolar ventilation to oxygen consumption/carbon dioxide production.
Respiratory Physiology Terms
Key Definitions
Term | Definition |
|---|---|
Hyperpnea | Increased ventilation to meet metabolic demands |
Dyspnea | Labored or difficult breathing |
Apnea | Temporary cessation of breathing |
Tachypnea | Rapid, shallow breathing |
Hyperventilation | Ventilation exceeds metabolic demands |
Hypoventilation | Ventilation insufficient for metabolic demands |
Hypoxia | Deficiency of oxygen in tissues |
Hypoxemia | Deficiency of oxygen in blood |
Hypercapnia | Excess carbon dioxide in blood |
Hypocapnia | Deficiency of carbon dioxide in blood |

Transport of Gases in the Blood
Oxygen Transport
Oxygen is transported in two forms: dissolved in plasma (3 mL/L) and bound to hemoglobin (197 mL/L).
Hemoglobin saturation depends on the partial pressure of oxygen (PO2).
Oxyhemoglobin (HbO2) forms at high PO2; deoxyhemoglobin predominates at low PO2.

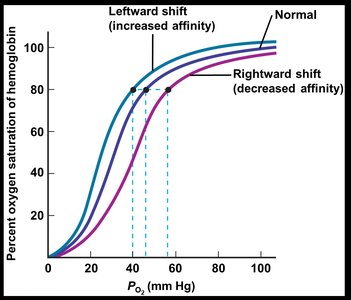
Oxyhemoglobin Dissociation Curve
Describes the relationship between PO2 and hemoglobin saturation.
At PO2 of 100 mmHg (arterial), Hb is ~98.5% saturated; at 40 mmHg (venous), ~75% saturated.
The curve is sigmoidal due to cooperative binding of O2 to hemoglobin.

Factors Affecting Hemoglobin Affinity for O2
Increased temperature, decreased pH, increased PCO2, and increased 2,3-BPG shift the curve right (decreased affinity).
Decreased temperature, increased pH, decreased PCO2, and decreased 2,3-BPG shift the curve left (increased affinity).

Carbon Dioxide Transport
CO2 is transported dissolved in plasma (5%), bound to hemoglobin as carbaminohemoglobin (5%), and as bicarbonate ions (90%).
The enzyme carbonic anhydrase catalyzes the conversion of CO2 and H2O to H2CO3, which dissociates to H+ and HCO3-.
Regulation of Ventilation
Control by Chemoreceptors
Central chemoreceptors (medulla) and peripheral chemoreceptors (carotid bodies) detect changes in arterial PO2 and PCO2.
Increased PCO2 or decreased PO2 stimulates increased ventilation.
Negative feedback mechanisms maintain homeostasis of blood gases.
Acid-Base Homeostasis
pH Regulation and Buffer Systems
Normal blood pH is 7.38–7.42; deviations can be life-threatening.
Acidosis: pH < 7.35 (CNS depression); Alkalosis: pH > 7.45 (CNS excitation).
Hemoglobin and bicarbonate ions act as buffers to resist changes in pH.
The Henderson-Hasselbalch equation describes the relationship between pH, bicarbonate, and CO2:
Respiratory and renal compensation mechanisms restore acid-base balance.
Summary
Gas exchange and transport are tightly regulated to maintain homeostasis.
Partial pressures, solubility, and hemoglobin affinity are key determinants of O2 and CO2 movement.
Ventilation is controlled by chemoreceptor feedback to match metabolic demands and maintain acid-base balance.
