 Back
BackGas Exchange and Transport in Human Physiology
Study Guide - Smart Notes

Gas Exchange in the Respiratory System
Partial Pressures and Gas Diffusion
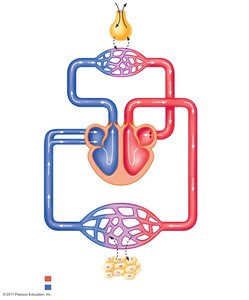
Gas exchange in the lungs and tissues is driven by differences in partial pressures of oxygen (O2) and carbon dioxide (CO2). These gradients facilitate the movement of gases between alveolar air, blood, and tissues.
Partial Pressure (P): The pressure exerted by an individual gas in a mixture. It determines the direction and rate of diffusion.
Alveolar Gas Exchange: O2 diffuses from alveoli (PO2 ≈ 100 mmHg) into pulmonary capillaries (PO2 ≈ 40 mmHg), while CO2 diffuses from blood (PCO2 ≈ 46 mmHg) into alveoli (PCO2 ≈ 40 mmHg).
Tissue Gas Exchange: O2 diffuses from systemic capillaries (PO2 ≈ 100 mmHg) into tissues (PO2 ≤ 40 mmHg), and CO2 moves from tissues (PCO2 ≥ 46 mmHg) into blood (PCO2 ≈ 40 mmHg).
Factors Affecting Diffusion: Thickness and surface area of the respiratory membrane, solubility, and partial pressure gradients of gases.
Example: At high altitude, atmospheric pressure is lower, reducing alveolar PO2 and thus the gradient for O2 diffusion into blood.
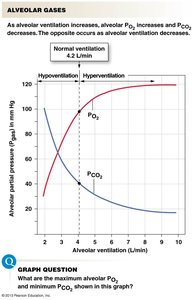
Determinants of Alveolar PO2 and PCO2
Alveolar partial pressures are influenced by:
PO2 and PCO2 of inspired air
Minute alveolar ventilation
Rates of O2 consumption and CO2 production by tissues
The most critical factor is the rate of alveolar ventilation relative to metabolic demand.
Equation: Partial Pressure = Total Pressure × Fraction of Gas
Example: At sea level (760 mmHg), PO2 = 760 × 0.2093 ≈ 159 mmHg. At the summit of Everest (250 mmHg), PO2 = 250 × 0.2093 ≈ 52.3 mmHg.
Respiratory Terminology
Key terms used in respiratory physiology describe various breathing patterns and gas imbalances:
Term | Definition |
|---|---|
Hyperpnea | An increase in ventilation to meet an increase in the metabolic demands of the body |
Dyspnea | Labored or difficult breathing |
Apnea | Temporary cessation of breathing |
Tachypnea | Rapid, shallow breathing |
Hyperventilation | Ventilation exceeds metabolic demands |
Hypoventilation | Ventilation is insufficient to meet metabolic demands |
Hypoxia | Deficiency of oxygen in the tissues |
Hypoxemia | Deficiency of oxygen in the blood |
Hypercapnia | Excess of carbon dioxide in the blood |
Hypocapnia | Deficiency of carbon dioxide in the blood |

Oxygen Transport in Blood
Mechanisms of Oxygen Transport
Oxygen is transported in blood in two forms:
Dissolved in Plasma: Only about 1.5% of O2 is transported this way due to low solubility.
Bound to Hemoglobin (Hb): The majority (98.5%) of O2 is carried bound to hemoglobin within red blood cells.
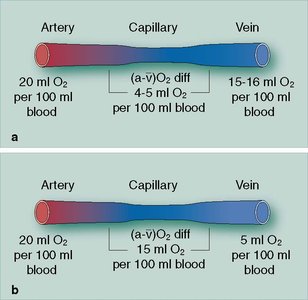
Oxygen Carrying Capacity: 1 L of blood can carry about 200 mL of O2 (20 mL per 100 mL blood).
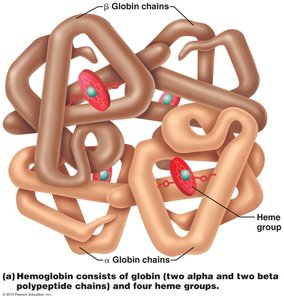
Hemoglobin Structure: Hemoglobin consists of four polypeptide chains (two alpha, two beta) and four heme groups, each capable of binding one O2 molecule.
Oxygen Loading and Unloading
Oxygen binds to hemoglobin in the lungs (forming oxyhemoglobin, HbO2) and is released in the tissues (forming deoxyhemoglobin, Hb).
Law of Mass Action: The amount of O2 bound to Hb depends on PO2 and Hb affinity for O2.
Hemoglobin Saturation: The percentage of Hb binding sites occupied by O2. 100% saturation means all sites are occupied.
Example: In arterial blood (PO2 = 100 mmHg), Hb is ~98.5% saturated; in venous blood (PO2 = 40 mmHg), Hb is ~75% saturated.
The Oxyhemoglobin Dissociation Curve
This curve shows the relationship between PO2 and hemoglobin saturation. It is sigmoidal due to cooperative binding of O2 to Hb.
Right Shift: Decreased affinity (more O2 unloading) due to increased temperature, decreased pH (Bohr effect), increased CO2 (carbamino effect), or increased 2,3-DPG.
Left Shift: Increased affinity (less O2 unloading) due to decreased temperature, increased pH, or decreased CO2.
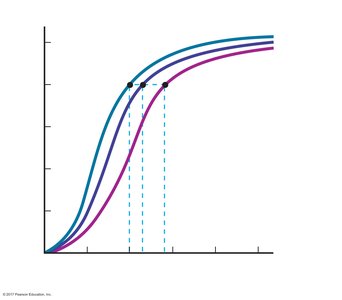
Bohr Effect: Lower pH (higher H+) decreases Hb affinity for O2, enhancing O2 unloading in active tissues.
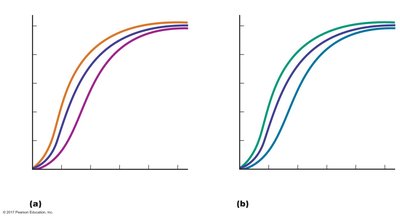
Carbamino Effect: CO2 binding to Hb (forming carbaminohemoglobin) decreases O2 affinity, promoting O2 release in metabolically active tissues.
2,3-DPG: Produced in RBCs under low O2 conditions (e.g., anemia, high altitude), decreases Hb affinity for O2.
Carbon Monoxide: Binds to Hb with higher affinity than O2, preventing O2 transport.
Carbon Dioxide Transport in Blood
Mechanisms of CO2 Transport
CO2 is transported in blood in three forms:
Dissolved in Plasma: 5–6% of total CO2.
Bound to Hemoglobin: 5–8% as carbaminohemoglobin (HbCO2).
As Bicarbonate (HCO3-): 86–90% is converted to bicarbonate in RBCs and transported in plasma.
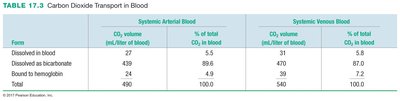
Form | Systemic Arterial Blood (mL/L) | % of Total CO2 | Systemic Venous Blood (mL/L) | % of Total CO2 |
|---|---|---|---|---|
Dissolved in blood | 27 | 5.5 | 31 | 5.8 |
Dissolved as bicarbonate | 429 | 88.6 | 470 | 87.0 |
Bound to hemoglobin | 24 | 4.9 | 39 | 7.2 |
Total | 480 | 100.0 | 540 | 100.0 |
Conversion of CO2 to Bicarbonate
CO2 is converted to carbonic acid (H2CO3) by the enzyme carbonic anhydrase in red blood cells, which then dissociates into bicarbonate (HCO3-) and hydrogen ions (H+).
Equation:
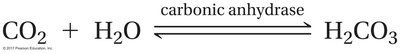
Law of Mass Action: An increase in CO2 drives the reaction toward more bicarbonate and H+ production.
Summary and Learning Outcomes
Understand the role of partial pressures in gas exchange at the lungs and tissues.
Describe the mechanisms of O2 and CO2 transport in blood, including the role of hemoglobin.
Identify physiological factors that affect hemoglobin's affinity for O2 and interpret the oxyhemoglobin dissociation curve.
