 Back
BackHeart Failure: Systolic and Diastolic Dysfunction, HFpEF, HCM, and RCM
Study Guide - Smart Notes

Heart Failure: Systolic and Diastolic Dysfunction
Systolic Dysfunction
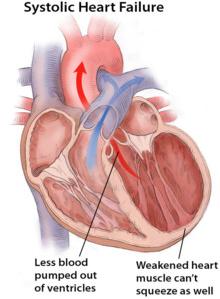
Systolic dysfunction is a form of heart failure characterized by the heart's inability to contract effectively, resulting in reduced ejection fraction and ventricular dilation.
Ejection Fraction (EF) < 50%: Indicates reduced pumping ability of the left ventricle.
Ventricular Dilation: The left ventricle becomes enlarged as it tries to compensate for poor contractility.
Symptoms: Shortness of breath, exercise intolerance, and peripheral edema are common clinical features.
Pathophysiology: Weakened heart muscle cannot generate enough force to eject blood efficiently.
Key Equation: Ejection fraction is calculated as: where EDV is end-diastolic volume and ESV is end-systolic volume.

Diastolic Dysfunction
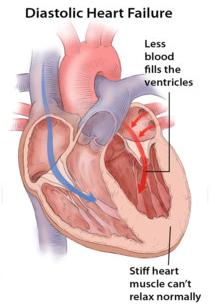
Diastolic dysfunction occurs when the heart muscle becomes stiff and cannot relax properly, leading to impaired ventricular filling despite a preserved ejection fraction.
Ejection Fraction (EF) ≥ 50%: The heart's pumping function is preserved, but filling is impaired.
No Ventricular Dilation: The ventricle size remains normal or may be slightly increased due to hypertrophy, not dilation.
Symptoms: Similar to systolic dysfunction—shortness of breath, exercise intolerance, and edema.
Pathophysiology: Stiff ventricular walls prevent adequate relaxation and filling during diastole, leading to increased filling pressures.
Heart Failure with Preserved Ejection Fraction (HFpEF)
Definition and Pathophysiology
HFpEF is a clinical syndrome where patients exhibit signs and symptoms of heart failure, but the left ventricular ejection fraction remains normal or near-normal (≥ 50%). The primary abnormality is diastolic dysfunction.
Impaired Relaxation: The myocardium cannot relax efficiently, often due to increased cytosolic calcium or changes in the extracellular matrix and cytoskeleton.
Elevated Filling Pressures: Because the ventricle is stiff, higher pressures are needed to fill it, leading to symptoms of congestion.
Common Causes: Hypertension, aging, obesity, diabetes, and systemic inflammation contribute to HFpEF.
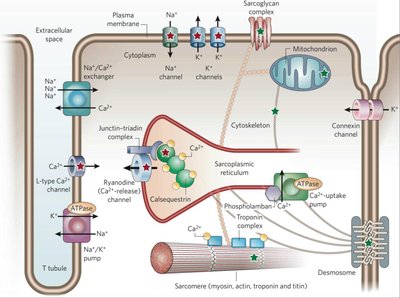
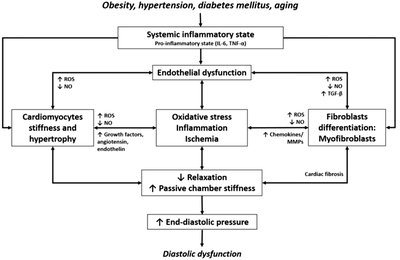
Phases of Ventricular Diastole
Ventricular diastole consists of several phases, each contributing to ventricular filling:
Isovolumetric Relaxation: The ventricle relaxes but all valves are closed, so volume does not change.
Early Rapid Filling (E wave): The mitral valve opens and blood flows rapidly into the ventricle.
Diastasis: Slow filling phase as pressures equilibrate.
Atrial Systole (A wave): The atrium contracts, pushing additional blood into the ventricle.

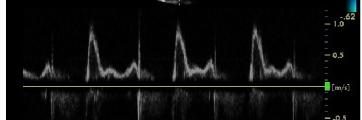
Assessment of Diastolic Function
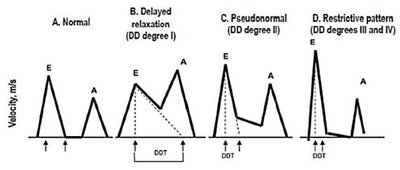
Diastolic function is commonly assessed using Doppler echocardiography, focusing on the E/A ratio and deceleration time (DT):
Normal: E/A > 1, DT 150–200 ms
Impaired Relaxation (Grade I): E/A < 1, prolonged DT
Pseudonormal (Grade II): E/A normalizes, but filling pressures are elevated
Restrictive Filling (Grade III/IV): E/A > 2, short DT, high filling pressures
Hypertrophic Cardiomyopathy (HCM)
Definition and Etiology
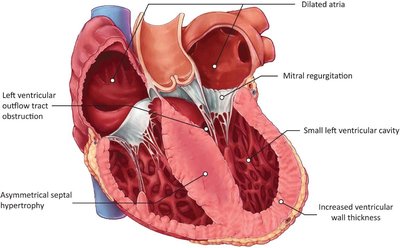
Hypertrophic cardiomyopathy is a genetic disorder characterized by abnormal thickening of the ventricular walls, most commonly the interventricular septum, leading to impaired diastolic filling and sometimes outflow tract obstruction.
Genetic Causes: Mutations in sarcomeric proteins such as β-myosin heavy chain, troponin I/T, and tropomyosin.
Other Causes: Metabolic and neuromuscular diseases (e.g., Anderson-Fabry, Pompe, Friedreich’s ataxia).
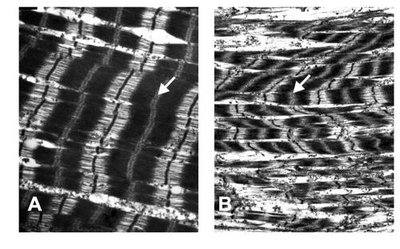
Pathology: Myocyte disarray, fibrosis, and abnormal sarcomere structure.
Clinical Features and Diagnosis
Symptoms: Dyspnea, chest pain, syncope, and risk of sudden cardiac death.
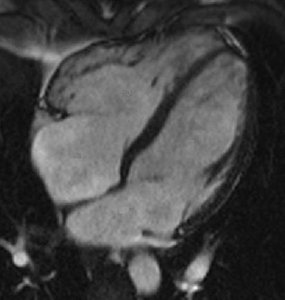
Diagnosis: Echocardiography shows asymmetric septal hypertrophy, small left ventricular cavity, and possible outflow tract obstruction.
Treatment
Beta-blockers: First-line therapy to reduce heart rate and increase diastolic filling time.
Calcium Channel Blockers: Used if beta-blockers are not tolerated; improve relaxation.
Disopyramide: Negative inotrope, added in obstructive HCM, often with beta-blockers.
Mavacamten: Cardiac myosin inhibitor for symptomatic obstructive HCM.
Restrictive Cardiomyopathy (RCM)
Definition and Etiology
Restrictive cardiomyopathy is characterized by stiff ventricular walls that impede diastolic filling, while systolic function is often preserved until late stages.
Noninfiltrative: Sarcomeric protein mutations, systemic sclerosis, idiopathic causes.
Infiltrative: Amyloidosis, sarcoidosis, iron overload.
Metabolic: Danon disease, Pompe disease, Gaucher disease.
Endomyocardial: Fibrosis, metastatic tumor, chemotherapy, radiation therapy.
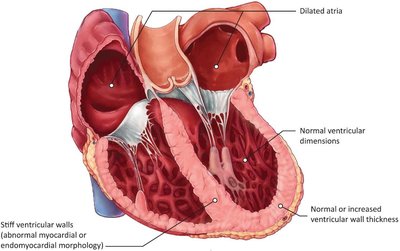
Clinical Features and Diagnosis
Symptoms: Similar to other forms of heart failure—dyspnea, fatigue, edema.
Diagnosis: Echocardiography shows normal or increased wall thickness, normal ventricular dimensions, and dilated atria.
Treatment
Beta-blockers: Used to slow heart rate and increase filling time.
Treat Underlying Cause: Disease-specific therapy for amyloidosis, immunosuppression for sarcoidosis.
Advanced Options: Heart transplant in selected patients.
Comparison of HCM and RCM
Feature | Hypertrophic Cardiomyopathy (HCM) | Restrictive Cardiomyopathy (RCM) |
|---|---|---|
Ventricular Wall Thickness | Increased (asymmetric) | Normal or increased (diffuse) |
Ventricular Cavity Size | Small | Normal |
Diastolic Function | Impaired (due to hypertrophy) | Impaired (due to stiffness) |
Systolic Function | Normal or increased | Normal until late |
Summary and Key Learning Points
Differentiate systolic vs diastolic dysfunction based on ejection fraction, ventricular structure, and filling mechanics.
Explain the pathophysiology of HFpEF, with emphasis on impaired relaxation and elevated filling pressures.
Recognize the structural and functional hallmarks of HCM and RCM.
Compare HCM and RCM in terms of ventricular thickness, cavity size, and diastolic function.
