 Back
BackMechanism of Force Generation in Skeletal Muscle
Study Guide - Smart Notes

Mechanism of Force Generation in Muscle
Sliding-Filament Model of Muscle Contraction
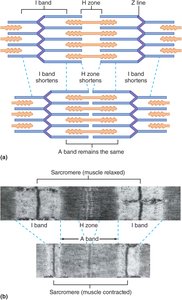
The sliding-filament model explains how skeletal muscle contracts by describing the interaction between thick and thin filaments within the sarcomere. The arrangement of these filaments is crucial for muscle function, and their movement leads to muscle shortening.
Sarcomere Structure: The sarcomere is the basic contractile unit of muscle, bordered by Z lines. It contains thick filaments (myosin) and thin filaments (actin).
Band Changes During Contraction: During contraction, the A band (length of thick filaments) remains unchanged, while the I bands and H zone shorten as thin filaments slide past thick filaments.
Result: The Z lines move closer together, shortening the sarcomere, myofibrils, muscle fibers, and ultimately the whole muscle.
Key Principle: Muscle contraction is due to the sliding of filaments, not their shortening.
Example: The shortening of the I band and H zone during contraction is visible in muscle striation patterns.
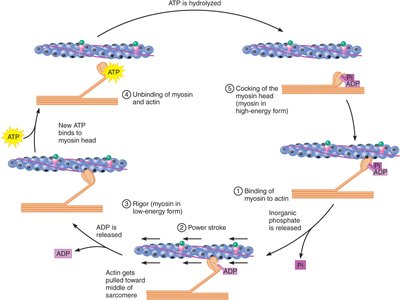
The Crossbridge Cycle: How Muscles Generate Force
The crossbridge cycle is the molecular mechanism that drives the sliding of filaments, powered by ATP hydrolysis. Myosin heads undergo conformational changes, binding and unbinding to actin, resulting in force generation.
Step 1: Binding of Myosin to Actin – Myosin in its energized (high-energy) form binds to actin, facilitated by the presence of calcium.
Step 2: Power Stroke – Myosin head pivots, pulling actin toward the center of the sarcomere, releasing inorganic phosphate (Pi).
Step 3: Rigor – Myosin and actin are tightly bound; ADP is released, and myosin enters its low-energy form. This step is associated with rigor mortis after death.
Step 4: Unbinding of Myosin and Actin – ATP binds to myosin, causing it to detach from actin.
Step 5: Cocking of the Myosin Head – ATP is hydrolyzed, energizing myosin and preparing it for another cycle.
Cycle Continuity: Multiple crossbridges operate out of phase, ensuring continuous force generation during contraction.
Efficiency: Each myosin head completes about five cycles per second, but hundreds of heads per filament allow rapid muscle shortening.
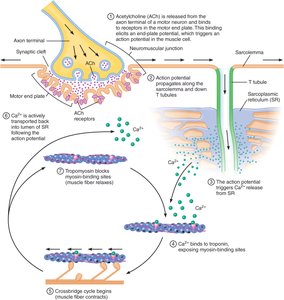
Excitation-Contraction Coupling: Turning Muscle Contractions On and Off
Excitation-contraction coupling links the electrical signal from a motor neuron to muscle contraction. This process involves the neuromuscular junction, action potential propagation, and calcium release.
Neuromuscular Junction: The synapse between a motor neuron and muscle fiber. Acetylcholine (ACh) is released, binds to receptors on the motor end plate, and triggers an action potential in the muscle cell.
Action Potential Propagation: The action potential travels along the sarcolemma and down T tubules, leading to calcium release from the sarcoplasmic reticulum (SR).
Calcium Release: Calcium serves as the signal to initiate the crossbridge cycle and muscle contraction.
End-Plate Potential: The depolarization at the motor end plate is always above threshold, ensuring muscle activation.
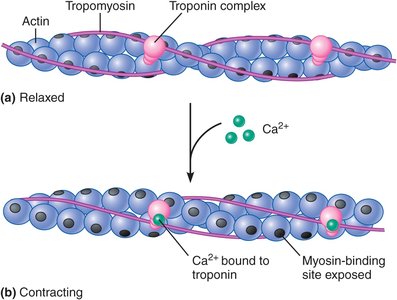
The Roles of Calcium, Troponin, and Tropomyosin in Excitation-Contraction Coupling
Calcium, troponin, and tropomyosin regulate the crossbridge cycle by controlling the exposure of myosin-binding sites on actin.
Resting Muscle: Low cytosolic calcium; troponin and tropomyosin block myosin-binding sites on actin, preventing contraction.
Calcium Pumps: The SR actively transports calcium into its lumen, keeping cytosolic levels low.
Calcium Release: Action potentials open voltage-gated calcium channels, increasing cytosolic calcium.
Troponin-Tropomyosin Regulation: Calcium binds to troponin, causing tropomyosin to shift and expose myosin-binding sites, allowing contraction.
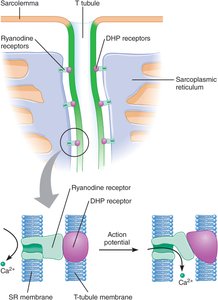
Gating of Sarcoplasmic Reticulum Calcium Channels
The release of calcium from the SR is controlled by DHP and ryanodine receptors, which are physically linked between the T tubule and SR membranes.
DHP Receptors: Located in the T tubule membrane, act as voltage sensors.
Ryanodine Receptors: Located in the SR membrane, function as calcium channels gated by DHP receptors.
Calcium-Induced Calcium Release: Initial calcium release triggers further release, amplifying the signal.
Termination of Muscle Contraction
Muscle contraction ends when motor neuron input ceases, action potentials stop, and calcium is actively transported back into the SR.
Ryanodine Receptor Closure: Channels close as membrane potential normalizes.
Negative Feedback: High cytosolic calcium causes channels to close, stopping further release.
Calcium Removal: Active transport clears calcium from the cytosol, troponin and tropomyosin revert, and contraction ends.
Summary Table: Key Events in Muscle Contraction
Event | Key Molecules/Structures | Function |
|---|---|---|
Sliding Filament | Actin, Myosin | Muscle shortening |
Crossbridge Cycle | Myosin, Actin, ATP | Force generation |
Excitation-Contraction Coupling | Acetylcholine, Calcium | Signal transmission |
Regulation | Troponin, Tropomyosin | Control of contraction |
Calcium Channel Gating | DHP, Ryanodine Receptors | Calcium release |
Termination | Calcium Pumps | Relaxation |
Sample Questions for Review
Which of the following shortens during a muscle contraction: thick filaments, thin filaments, A bands, I bands, H zones, sarcomeres?
When ATP is hydrolyzed, do myosin crossbridges enter the high-energy or low-energy form?
What triggers the power stroke of the crossbridge cycle?
What is the function of the sarcoplasmic reticulum in skeletal muscle?
How does an increase or decrease in cytosolic calcium level cause the crossbridge cycle to turn on or off?
Additional info:
Muscle cramps may result from excessive calcium release, impaired calcium reuptake, or ATP depletion, leading to sustained contraction.
Acetylcholinesterase is the enzyme that breaks down acetylcholine after it is released at the neuromuscular junction.
