 Back
Back(2/19_A+P) Pulmonary Ventilation and Gas Exchange: Mechanics, Laws, and Clinical Relevance
Study Guide - Smart Notes

The Respiratory System: Pulmonary Ventilation and Gas Exchange
Introduction
The respiratory system is essential for gas exchange, supplying oxygen to the body and removing carbon dioxide. This section focuses on the mechanics of breathing, the physical laws governing ventilation, and the clinical implications of pulmonary function.
Mechanics of Breathing
Pressure Relationships and Boyle’s Law
Breathing depends on pressure differences between the lungs and the atmosphere. According to Boyle’s Law, at a constant temperature, the pressure of a gas is inversely proportional to its volume:
Increased volume → Decreased pressure
Decreased volume → Increased pressure
The mathematical expression is:
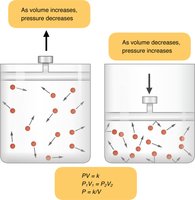
When the thoracic cavity expands, lung volume increases and pressure decreases, causing air to flow in. When the cavity contracts, volume decreases and pressure increases, pushing air out.
Pressure Relationships in the Thoracic Cavity
Atmospheric Pressure (Patm): Pressure exerted by air around the body (760 mm Hg at sea level).
Intra-alveolar Pressure (Palv): Pressure within the alveoli; equalizes with atmospheric pressure.
Intrapleural Pressure (Pip): Always negative relative to Palv and Patm; prevents lung collapse.
Transpulmonary Pressure: Difference between Palv and Pip; keeps lungs inflated.
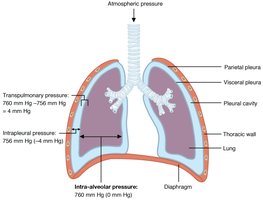
Pneumothorax
Pneumothorax occurs when air enters the pleural cavity, causing Pip to equalize with Patm. This eliminates the pressure gradient, leading to lung collapse.
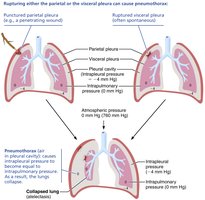
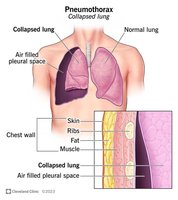
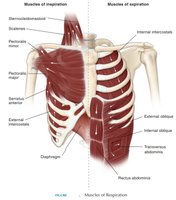
Pulmonary Ventilation: Inspiration and Expiration
Airflow is driven by pressure differences:
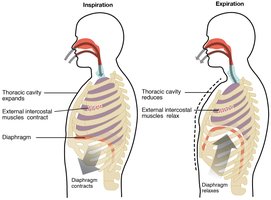
Inspiration: Diaphragm contracts and moves downward, external intercostals lift ribs, thoracic volume increases, Palv drops below Patm, air flows in.
Expiration: Diaphragm and intercostals relax, thoracic volume decreases, Palv rises above Patm, air flows out.
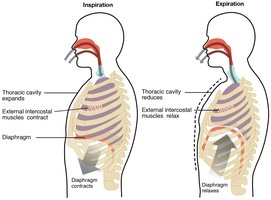
Respiratory Cycle
One complete sequence of inspiration and expiration.
Normal expiration is passive, relying on lung elasticity.
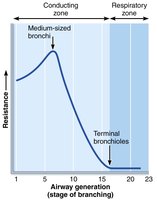
Airway Resistance and Flow
Airway diameter affects resistance:
Smaller airways = higher resistance.
Greatest resistance occurs in medium-sized bronchi.
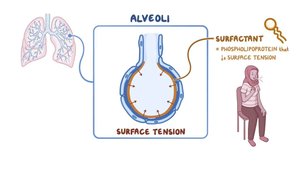
Physical Factors Affecting Ventilation
Surface Tension: Alveolar fluid creates surface tension, resisting expansion.
Surfactant: Reduces surface tension, preventing alveolar collapse.
Thoracic Wall Compliance: High compliance makes lung expansion easier; low compliance increases effort required for breathing.
Modes of Breathing
Quiet Breathing (Eupnea): Automatic, at rest; diaphragm and external intercostals contract for inspiration.
Forced Breathing (Hyperpnea): Active, during exercise or singing; involves additional muscles (scalenes, abdominals, internal intercostals).
Respiratory Volumes and Capacities
Respiratory Volumes
Tidal Volume (TV): Air inhaled/exhaled during normal breathing (~500 mL).
Inspiratory Reserve Volume (IRV): Additional air inhaled after normal inspiration.
Expiratory Reserve Volume (ERV): Additional air exhaled after normal expiration.
Residual Volume (RV): Air remaining in lungs after forced expiration.

Respiratory Capacities
Total Lung Capacity (TLC): Maximum air in lungs after maximal inspiration.
Forced Vital Capacity (FVC): Maximum air exhaled after maximal inspiration.
Inspiratory Capacity (IC): Maximum air inspired after normal expiration.
Functional Residual Capacity (FRC): Air remaining after normal expiration.

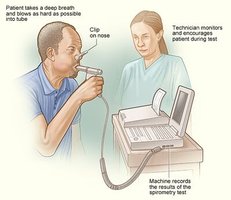
Clinical Pulmonary Function Tests
Pulmonary function tests (e.g., spirometry) measure lung volumes and capacities to assess respiratory health.
Gas Laws and Gas Exchange
Dalton’s Law of Partial Pressures
Atmospheric air is a mixture of gases. Each gas exerts a partial pressure proportional to its concentration. Dalton’s Law states:
Partial Pressure (Px): Pressure exerted by a single gas in a mixture.

Gas | Percent of total composition | Partial pressure (mm Hg) |
|---|---|---|
Nitrogen (N₂) | 78.6 | 597.4 |
Oxygen (O₂) | 20.9 | 158.8 |
Water (H₂O) | 0.4 | 3.0 |
Carbon dioxide (CO₂) | 0.04 | 0.3 |
Others | 0.06 | 0.5 |
Total | 100% | 760.0 |
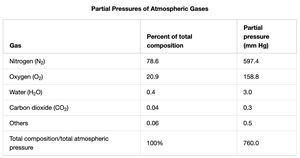
At higher altitudes, total atmospheric pressure and partial pressures decrease proportionally.
Henry’s Law
Henry’s Law states that the amount of gas dissolved in a liquid is proportional to its partial pressure and solubility in that liquid:
Higher partial pressure = more gas dissolves.
Solubility varies by gas (CO₂ is more soluble than O₂; N₂ is least soluble).
Example: In scuba diving, increased pressure causes more nitrogen to dissolve in blood, which can be dangerous if not managed properly.
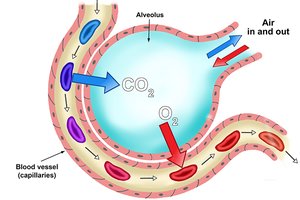
Gas Exchange: Pulmonary and Tissue Levels
Sites and Mechanism of Gas Exchange
External Respiration: Gas exchange between alveoli and blood (O₂ in, CO₂ out).
Internal Respiration: Gas exchange between blood and tissues (O₂ out, CO₂ in).
Occurs by simple diffusion down partial pressure gradients.
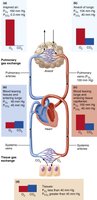
Factors Influencing Pulmonary Gas Exchange
Partial Pressure Gradients and Gas Solubilities: Large PO₂ gradient drives O₂ into blood; smaller PCO₂ gradient, but CO₂ diffuses rapidly due to higher solubility.
Thickness and Surface Area of Respiratory Membrane: Thin membrane and large surface area facilitate rapid diffusion.
Ventilation-Perfusion Coupling: Matching airflow (ventilation) and blood flow (perfusion) optimizes gas exchange.
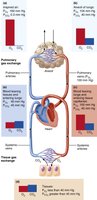
Thickness and Surface Area of the Respiratory Membrane
Normal membrane: 0.5–1 micron thick; large surface area (~60x skin surface).
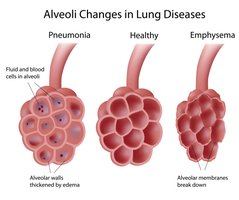
Pulmonary edema increases thickness, slowing O₂ diffusion (hypoxia).
Emphysema reduces surface area, decreasing gas exchange efficiency.
Ventilation-Perfusion Coupling
Efficient gas exchange requires matching of ventilation (V) and perfusion (Q).
Low ventilation: Pulmonary vasoconstriction redirects blood to better-ventilated alveoli.
High ventilation: Pulmonary vasodilation increases blood flow to match increased airflow.

Summary Table: Key Respiratory Volumes and Capacities
Volume/Capacity | Definition |
|---|---|
Tidal Volume (TV) | Air inhaled/exhaled during normal breathing (~500 mL) |
Inspiratory Reserve Volume (IRV) | Additional air inhaled after normal inspiration |
Expiratory Reserve Volume (ERV) | Additional air exhaled after normal expiration |
Residual Volume (RV) | Air remaining in lungs after forced expiration |
Total Lung Capacity (TLC) | Maximum air in lungs after maximal inspiration |
Forced Vital Capacity (FVC) | Maximum air exhaled after maximal inspiration |
Inspiratory Capacity (IC) | Maximum air inspired after normal expiration |
Functional Residual Capacity (FRC) | Air remaining after normal expiration |
Key Takeaways
Breathing is governed by pressure-volume relationships (Boyle’s Law).
Gas exchange depends on partial pressure gradients (Dalton’s Law) and solubility (Henry’s Law).
Efficient gas exchange requires thin membranes, large surface area, and matched ventilation and perfusion.
Clinical conditions like pneumothorax, pulmonary edema, and emphysema disrupt normal respiratory function.
