 Back
BackThe Reproductive System: Hormonal Regulation and Gametogenesis in Mammals
Study Guide - Smart Notes

Reproductive System Overview
Introduction to the HPG Axis
The reproductive system in mammals is regulated by the hypothalamus-pituitary-gonad (HPG) axis, which integrates neural and endocrine signals to control reproductive timing and function. Environmental factors such as light, temperature, and tactile stimuli can influence reproductive cycles through their effects on this axis.
HPG Axis: Involves the hypothalamus (releasing GnRH), pituitary (releasing LH and FSH), and gonads (producing steroid hormones).
Feedback Regulation: Gonadal steroids exert negative feedback on GnRH release, except for the positive feedback of estrogens on the female surge center.
Environmental Influence: External cues modulate reproductive timing, especially in seasonally breeding species.
Hormonal Control of Reproduction
Gonadotropins and Steroid Hormones
Reproductive events are orchestrated by the pulsatile release of gonadotropin-releasing hormone (GnRH), which stimulates the anterior pituitary to secrete luteinizing hormone (LH) and follicle-stimulating hormone (FSH). These hormones regulate gametogenesis and steroidogenesis in both sexes.
FSH: Stimulates estrogen production in females and spermatogenesis in males.
LH: Triggers ovulation in females and androgen synthesis in males.
Sexual Differentiation: Gonadal steroids maintain secondary sexual characteristics (e.g., uterus, mammary glands, muscle development).
Male Reproductive Physiology
Spermatogenesis and Testicular Function
Spermatogenesis is the process of sperm cell development, regulated by FSH, LH, and local paracrine factors. FSH acts on Sertoli cells, which support and nourish developing sperm, while LH stimulates Leydig cells to produce testosterone.
Sertoli Cells: Mediate the effects of FSH and secrete paracrine factors and inhibin.
Leydig Cells: Produce testosterone in response to LH.
Testosterone: Acts locally at high concentrations to promote spermatogenesis, mainly through indirect mechanisms.
Estrogen Receptors: Present on germ cells, facilitating spermatogenesis via locally produced estrogens.
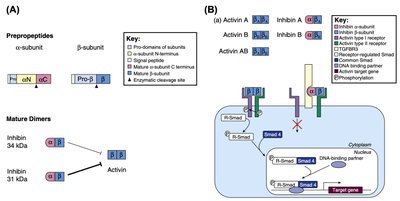
Inhibin: Secreted by Sertoli cells, inhibits FSH release from the pituitary.
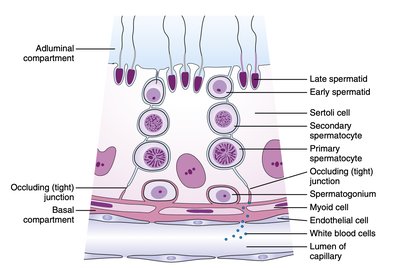
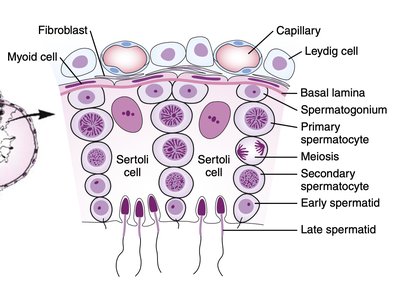
Paracrine and Endocrine Regulation
Testicular function is tightly regulated by a balance of endocrine and paracrine signals. Androgens, estrogens, and inhibins/activins coordinate the proliferation and maturation of germ cells.
GRTH/DDX25: A gene product activated by androgens, essential for meiosis in germ cells.
Estrogen Metabolism: Local conversion of androgens to estrogens is crucial for normal spermatogenesis.
Inhibin and Activin: Regulate FSH secretion and local testicular function.
Female Reproductive Physiology
Ovarian Cycle and Folliculogenesis
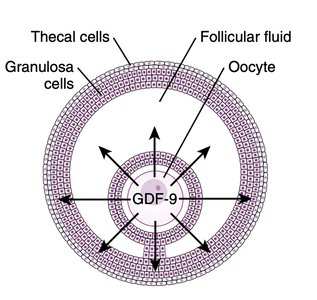
The ovarian cycle consists of the follicular phase, ovulation, and luteal phase. Follicular development is driven by FSH and LH, leading to the selection and maturation of a dominant follicle, ovulation, and corpus luteum formation.
Follicular Phase: Growth of ovarian follicles and estrogen production.
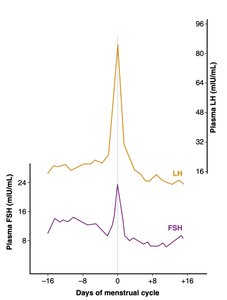
Ovulation: Triggered by an LH surge following a critical rise in estradiol.
Luteal Phase: Formation of the corpus luteum, which secretes progesterone and estrogens.
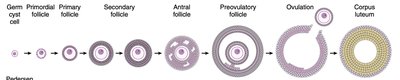
Hormonal Regulation of the Ovarian Cycle
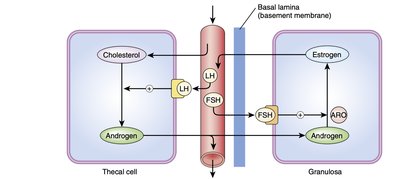
FSH and LH regulate the synthesis of estrogens and androgens in the ovary. Thecal cells produce androgens under LH stimulation, which are converted to estrogens by granulosa cells under FSH stimulation. Inhibin and activin modulate FSH secretion and local aromatase activity.
Two-Cell, Two-Gonadotropin Model: Thecal cells (LH) produce androgens; granulosa cells (FSH) convert androgens to estrogens.
Inhibin: Inhibits FSH secretion and local aromatase activity.
Activin: Enhances aromatase activity in granulosa cells.
Ovulation and Luteinization
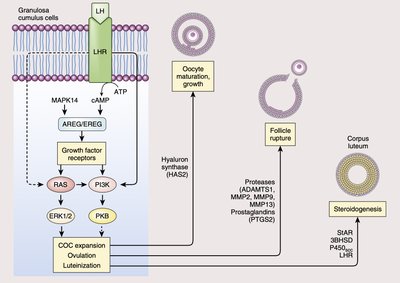
Ovulation is induced by a surge in LH, which activates multiple signaling pathways in granulosa cells, leading to follicle rupture and corpus luteum formation. The corpus luteum functions as a temporary endocrine gland, secreting progesterone and estrogens to maintain the luteal phase and inhibit further ovulation.
LH Surge: Causes ovulation and initiates luteinization of granulosa and thecal cells.
Signaling Pathways: Involvement of PKA, RAS, and ERK1/2 pathways in gene regulation for ovulation and luteinization.
Corpus Luteum: Secretes hormones to prevent additional ovulation and support early pregnancy if fertilization occurs.
Luteolysis and Maternal Recognition of Pregnancy
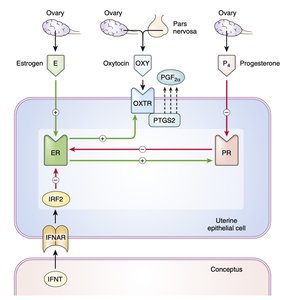
Luteolysis is the degeneration of the corpus luteum, triggered by prostaglandin F2α (PGF2α) secretion from the uterus in the absence of pregnancy. Progesterone and estrogen regulate the expression of receptors and enzymes involved in this process. Signals from the conceptus (e.g., interferon tau) can block luteolysis, maintaining the corpus luteum and supporting pregnancy.
Estradiol: Increases expression of estrogen, progesterone, and oxytocin receptors in uterine cells.
Progesterone: Downregulates its own receptor, enhancing ER and OXTR expression.
Oxytocin: Stimulates PGF2α release, leading to luteolysis.
Conceptus Signals: Interferon tau (IFNT) blocks the luteolytic pathway, prolonging corpus luteum function.
Summary Table: Key Hormones and Their Functions in Mammalian Reproduction
Hormone | Source | Main Target(s) | Primary Function(s) |
|---|---|---|---|
GnRH | Hypothalamus | Pituitary | Stimulates LH and FSH release |
FSH | Pituitary | Ovary (granulosa cells), Testis (Sertoli cells) | Follicle growth, estrogen synthesis, spermatogenesis |
LH | Pituitary | Ovary (thecal cells), Testis (Leydig cells) | Ovulation, androgen synthesis |
Estrogen | Ovary (granulosa cells) | Uterus, hypothalamus, pituitary | Endometrial growth, feedback regulation |
Progesterone | Corpus luteum | Uterus, hypothalamus | Maintains luteal phase, inhibits GnRH |
Testosterone | Testis (Leydig cells) | Various tissues | Secondary sexual characteristics, spermatogenesis |
Inhibin | Sertoli cells, granulosa cells | Pituitary | Inhibits FSH secretion |
Activin | Gonads | Gonads, pituitary | Stimulates FSH secretion, enhances aromatase activity |
PGF2α | Uterus | Corpus luteum | Induces luteolysis |
IFNT | Conceptus | Uterus | Blocks luteolysis, maintains pregnancy |
