 Back
BackThe Thyroid Gland: Structure, Function, and Clinical Significance
Study Guide - Smart Notes

The Thyroid Gland
Location and Structure
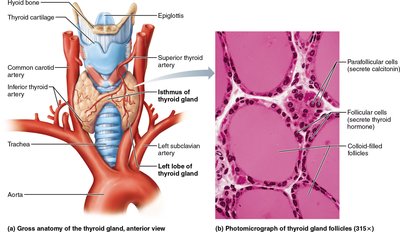
The thyroid gland is a butterfly-shaped organ located in the anterior neck, just below the larynx and on the trachea. It is the largest pure endocrine gland in the body and is highly vascularized, making surgical procedures complex. The gland consists of two lateral lobes connected by a median tissue mass called the isthmus.
Follicular cells: Cuboidal or squamous epithelial cells that produce the glycoprotein thyroglobulin.
Colloid: The central cavity of each follicle stores colloid, a sticky material containing iodinated thyroglobulin.
Parafollicular cells: These cells produce calcitonin and are located in the follicular epithelium.
Thyroid Hormone (TH)
Thyroid hormone is the body's major metabolic hormone and consists of two iodine-containing amine hormones:
Thyroxine (T4): The primary hormone secreted by the thyroid follicles, containing four iodine atoms.
Triiodothyronine (T3): Formed mainly at target tissues by conversion of T4 to T3, containing three iodine atoms.
Both hormones are constructed from two linked tyrosine amino acids. TH affects nearly every cell in the body by entering target cells, binding to nuclear receptors, and initiating transcription of mRNA for protein synthesis.
Calorigenic effect: Increases basal metabolic rate and body heat production by turning on genes for glucose oxidation.
Growth and development: Essential for normal skeletal and nervous system development and reproductive capabilities.
Blood pressure: Maintains blood pressure by increasing adrenergic receptors in blood vessels.
Physiological Effects and Clinical Manifestations
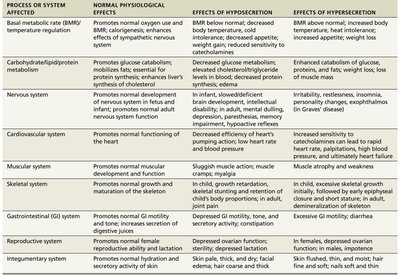
Thyroid hormone influences multiple body systems. The table below summarizes its effects, as well as the consequences of hypo- and hypersecretion:
Process or System Affected | Normal Physiological Effects | Effects of Hyposecretion | Effects of Hypersecretion |
|---|---|---|---|
Basal metabolic rate (BMR)/temperature regulation | Promotes normal oxygen use and BMR; calorigenic effect; enhances sympathetic nervous system effects | BMR below normal; decreased body temperature; cold intolerance; weight gain; reduced appetite | BMR above normal; increased body temperature; heat intolerance; weight loss; increased appetite |
Carbohydrate/lipid/protein metabolism | Promotes glucose catabolism; mobilizes fats; essential for protein synthesis; enhances liver's synthesis of cholesterol | Decreased glucose metabolism; elevated cholesterol; decreased protein synthesis | Enhanced catabolism of glucose, fats, and proteins; decreased cholesterol |
Nervous system | Promotes normal development and function | In infant: mental retardation; in adult: mental sluggishness, depression | Hyperactivity, irritability, insomnia |
Cardiovascular system | Promotes normal functioning of the heart | Decreased efficiency of heart; low blood pressure | Increased heart rate; high blood pressure |
Muscular system | Promotes normal muscular development and function | Sluggish muscle action; muscle weakness | Muscle atrophy and weakness |
Skeletal system | Promotes normal growth and maturation of the skeleton | Impaired growth; joint pain | Premature closure of epiphyseal plates |
Gastrointestinal (GI) system | Promotes normal GI motility and tone | Decreased GI motility; constipation | Diarrhea |
Reproductive system | Promotes normal reproductive function | In females: decreased ovarian function; in males: decreased sperm production | In females: menstrual irregularities |
Integumentary system | Promotes normal hydration and secretion by skin | Pale, thick, dry skin; hair loss | Flushed, thin skin; hair loss |
Synthesis of Thyroid Hormone
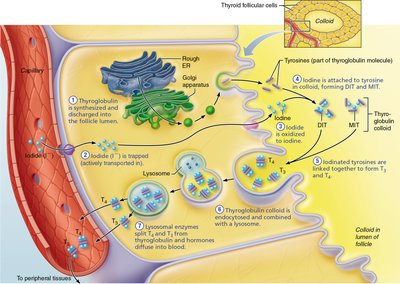
The thyroid gland is unique in its ability to store hormone extracellularly in large quantities. The synthesis of thyroid hormone involves several steps:
Thyroglobulin synthesis: Produced in the rough ER, processed in the Golgi, and discharged into the follicle lumen.
Iodide trapping: Follicular cells actively transport iodide from the blood into the cell, then into the follicle lumen.
Iodide oxidation: Iodide is oxidized to iodine at the follicular cell-colloid interface.
Iodination: Iodine attaches to tyrosine residues in thyroglobulin, forming monoiodotyrosine (MIT) and diiodotyrosine (DIT).
Coupling: Enzymes link MIT and DIT to form T3 and T4.
Endocytosis: Follicular cells reclaim iodinated thyroglobulin by endocytosis and combine it with lysosomes.
Release: Lysosomal enzymes split T3 and T4 from thyroglobulin, and the hormones diffuse into the blood.
Transport and Regulation
Most T3 and T4 released into the blood bind to thyroxine-binding globulins (TBGs) and other transport proteins produced by the liver. T3 binds more tightly to target tissue receptors and is more active than T4. Peripheral tissues convert T4 to T3 by removing one iodine atom.
Negative feedback loop: Falling TH levels trigger TSH release, stimulating more TH production. Rising TH levels inhibit the hypothalamic–anterior pituitary axis, reducing TSH release.
Cold exposure: In infants, cold stimulates the hypothalamus to release TRH, increasing TSH and TH production.
Inhibitors: GHIH, dopamine, glucocorticoids, and high blood iodide concentrations inhibit TSH release.
Clinical Disorders: Homeostatic Imbalance
Hypothyroidism: May result from thyroid defects, inadequate TSH/TRH, surgical removal, or iodine deficiency.
Myxedema: Characterized by low metabolic rate, cold intolerance, constipation, dry skin, edema, lethargy, and mental sluggishness.
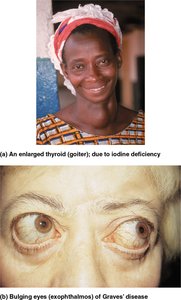
Goiter: Enlarged thyroid due to iodine deficiency; colloid accumulates but cannot be converted to functional hormone.
Congenital hypothyroidism: Poor thyroid development in infants; early treatment with hormone replacement is crucial.
Hyperthyroidism: Most commonly caused by Graves' disease, an autoimmune disorder where antibodies mimic TSH and overstimulate TH release.
Symptoms: Elevated metabolic rate, sweating, rapid heartbeat, nervousness, weight loss, and exophthalmos (bulging eyes).
Treatment: Surgical removal or radioactive iodine therapy.
Calcitonin
Calcitonin is a polypeptide hormone released by parafollicular cells in response to elevated blood calcium levels. It does not have a known physiological role in humans, and is not replaced in patients without a thyroid gland. At pharmacological doses, calcitonin has a bone-sparing effect and is used therapeutically for Paget's disease and osteoporosis.
Inhibits osteoclast activity: Reduces bone resorption and release of calcium from bone matrix.
Stimulates calcium uptake: Promotes incorporation of calcium into bone matrix.
Check Your Understanding
What is the difference between T3 and T4? Which one is referred to as thyroxine? T4 (thyroxine) has four iodine atoms and is the main hormone secreted by the thyroid. T3 has three iodine atoms and is more active at target tissues.
Why does TH require plasma proteins to move through blood? Where are the receptors for TSH located? TH is lipid-soluble and requires plasma proteins for transport in the aqueous environment of blood. TSH receptors are located on the follicular cells of the thyroid gland.
Additional info: The thyroid gland is essential for metabolic regulation, growth, and development. Disorders of thyroid function can have widespread effects on multiple organ systems.
