Back
BackThe Urinary System: Structure, Function, and Physiology
Study Guide - Smart Notes

The Urinary System
Overview and Anatomy
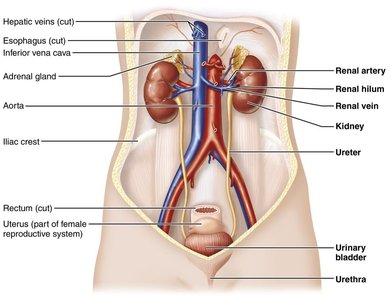
The urinary system is essential for maintaining homeostasis by regulating the composition and volume of blood, removing metabolic wastes, and balancing electrolytes and pH. The main organs include the kidneys, ureters, urinary bladder, and urethra.
Kidneys: Filter blood, remove wastes, and regulate fluid and electrolyte balance.
Ureters: Transport urine from the kidneys to the bladder.
Urinary bladder: Stores urine until excretion.
Urethra: Conducts urine out of the body.
Functions of the Kidneys
Regulation of blood ion concentration and plasma osmolarity
Regulation of blood pH by excreting hydrogen ions and reabsorbing bicarbonate
Regulation of blood volume and pressure through water reabsorption and hormone secretion
Hormone production: Erythropoietin (stimulates RBC production) and renin (regulates blood pressure)
Regulation of blood glucose level via gluconeogenesis
Excretion of metabolic wastes and foreign substances
Waste Products and Nitrogenous Wastes
Types of Wastes
Wastes are substances that are either useless to the body or present in excess. The kidneys are responsible for excreting metabolic wastes, especially nitrogenous wastes.
Metabolic waste: Produced by normal metabolic processes.
Nitrogenous wastes: By-products of protein and nucleic acid metabolism.
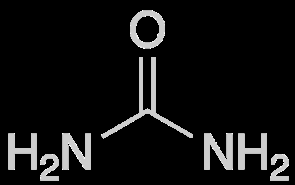
Urea: Main nitrogenous waste (about 50%), produced from protein catabolism.
Creatinine: From creatine phosphate metabolism.
Uric acid: From nucleic acid catabolism.
BUN (Blood Urea Nitrogen): Clinical measure of nitrogenous waste in blood.
Kidney Structure and Blood Supply
Gross and Internal Anatomy
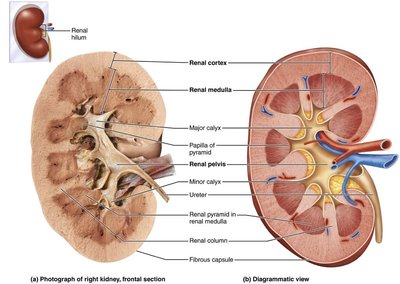
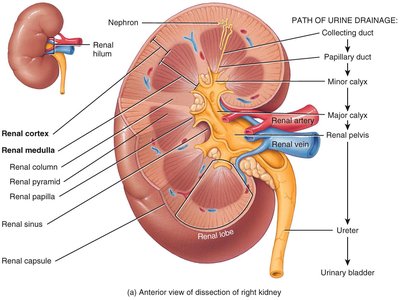
The kidney is divided into an outer cortex and an inner medulla. The cortex is the site of filtration, while the medulla collects and excretes urine. The renal hilum is the entry/exit site for vessels and the ureter.
Renal cortex: Site of filtration and initial urine formation.
Renal medulla: Contains renal pyramids, which collect and channel urine.
Renal pelvis: Funnel-shaped structure that collects urine and channels it to the ureter.
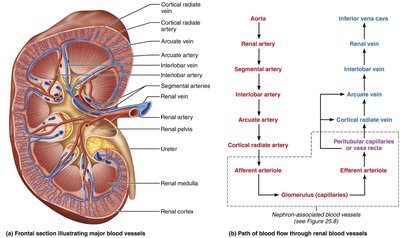
Renal Blood Flow
The kidneys receive about 25% of resting cardiac output through the renal arteries. Blood flows through a series of arteries and capillaries, allowing for filtration and reabsorption.
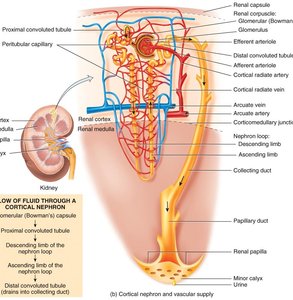
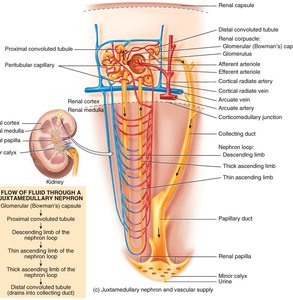
Renal artery → Segmental artery → Interlobar artery → Arcuate artery → Cortical radiate artery → Afferent arteriole → Glomerulus → Efferent arteriole → Peritubular capillaries/vasa recta → Cortical radiate vein → Arcuate vein → Interlobar vein → Renal vein
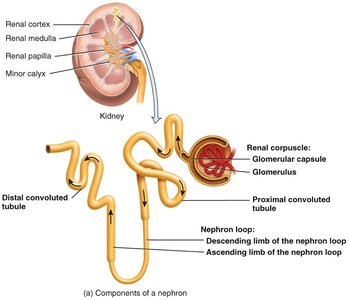
The Nephron: Structure and Types
Nephron Structure
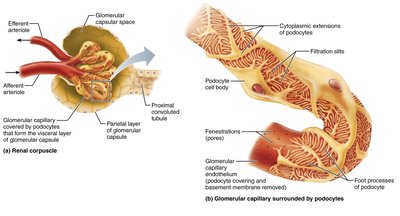
The nephron is the functional unit of the kidney, responsible for filtering blood and forming urine. Each nephron consists of a renal corpuscle and renal tubules.
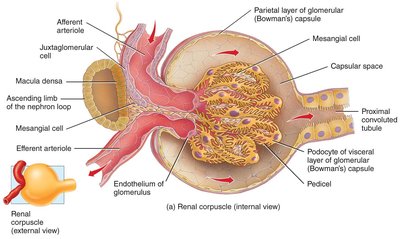
Renal corpuscle: Includes the glomerulus (capillary network) and Bowman's (glomerular) capsule; site of filtration.
Renal tubule: Includes the proximal convoluted tubule (PCT), nephron loop (loop of Henle), distal convoluted tubule (DCT), and collecting duct.
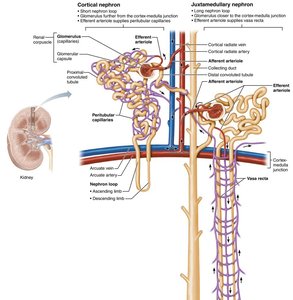
Types of Nephrons
Cortical nephrons: 80-85% of nephrons; located mainly in the cortex; short loops of Henle; peritubular capillaries supply blood.
Juxtamedullary nephrons: Located deep in the cortex; long loops of Henle extend into the medulla; vasa recta supply blood; crucial for urine concentration/dilution.
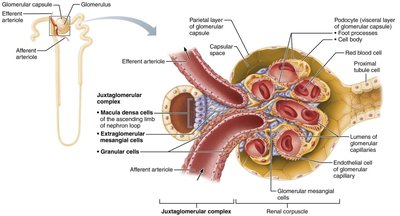
Juxtaglomerular Apparatus (JGA)
The JGA is a specialized structure that regulates blood pressure and GFR. It consists of the macula densa (in the ascending limb of the nephron loop) and juxtaglomerular cells (in the afferent arteriole wall). The JGA secretes renin in response to low blood pressure.
Renal Physiology: Filtration, Reabsorption, and Secretion
Nephron Functions
Glomerular filtration: Pressure forces water and solutes from blood into the glomerular capsule, forming filtrate.
Tubular reabsorption: Movement of useful substances from filtrate back into the blood.
Tubular secretion: Transfer of additional wastes from blood into the filtrate for excretion.
Glomerular Filtration
Filtration occurs in the renal corpuscle, where protein-free plasma is filtered into the glomerular capsule. The filtration membrane allows molecules smaller than 3 nm to pass (water, electrolytes, glucose, amino acids, nitrogenous wastes).
Filtration fraction: 16-20% of plasma entering the glomerulus becomes filtrate; 99% of filtrate is reabsorbed.
Filtration is enhanced by:
Large surface area of glomerular capillaries
High permeability of the filtration membrane
High glomerular capillary hydrostatic pressure
Net Filtration Pressure (NFP) and Starling Forces
NFP determines the amount of filtrate formed. It is calculated as the difference between outward and inward pressures at the glomerulus:
Blood hydrostatic pressure (BHP): 55 mmHg (pushes fluid out)
Capsular hydrostatic pressure (CHP): 15 mmHg (opposes filtration)
Blood colloid osmotic pressure (BCOP): 30 mmHg (opposes filtration)
Equation:
Example: mmHg
Factors Affecting NFP and GFR
High blood pressure: Increases NFP and GFR.
Low plasma proteins (e.g., starvation): Decreases BCOP, increases filtration, may cause edema.
Severe blood loss: Decreases BHP, reduces GFR.
Dehydration: Increases plasma protein concentration, increases BCOP, reduces GFR.
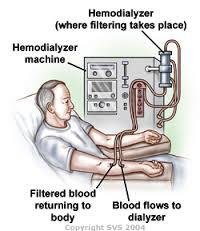
Dialysis
If kidney function is severely impaired, hemodialysis is used to artificially filter blood, removing wastes and excess electrolytes.
Glomerular Filtration Rate (GFR) and Its Regulation
Definition and Importance
GFR is the amount of filtrate formed in all renal corpuscles of both kidneys each minute. Normal GFR is 100-125 mL/min per kidney.
High GFR: Substances pass too quickly, not reabsorbed efficiently, lost in urine.
Low GFR: Nearly all filtrate is reabsorbed, including wastes, leading to inadequate excretion.
Regulation of GFR
Renal autoregulation: Nephrons adjust their own blood flow and GFR without external control (myogenic mechanism and tubuloglomerular feedback).
Neural regulation: Sympathetic nervous system constricts afferent arterioles, reducing GFR during stress or hemorrhage.
Hormonal regulation: Angiotensin II (vasoconstrictor, reduces GFR), Atrial natriuretic peptide (ANP, increases GFR by relaxing mesangial cells).
Tubular Reabsorption and Secretion
Principles of Tubular Reabsorption
Reabsorption returns essential substances (glucose, electrolytes, water, amino acids) to the blood. It occurs via paracellular (between cells) and transcellular (through cells) routes, using active and passive transport mechanisms.
Na+/K+ pump: Maintains low intracellular Na+, drives secondary active transport of other solutes.
Transport maximum (Tm): Maximum rate at which solutes can be reabsorbed; exceeded in conditions like glucosuria.
Water Reabsorption
Obligatory reabsorption: 90% of water reabsorbed in PCT and descending nephron loop via osmosis.
Facultative reabsorption: 10% of water reabsorbed in collecting duct, regulated by ADH.
Segmental Functions
Proximal convoluted tubule (PCT): Reabsorbs 65% of filtered water, Na+, K+, 100% glucose and amino acids, 50% Cl-, 80-90% HCO3-; secretes H+, NH4+, urea.
Nephron loop: Descending limb permeable to water, ascending limb permeable to solutes but not water; establishes osmotic gradient.
Distal convoluted tubule (DCT) and collecting duct: Fine-tune reabsorption and secretion; principal cells reabsorb Na+ and secrete K+ (regulated by aldosterone and ADH); intercalated cells reabsorb K+, HCO3-, secrete H+.
Hormonal Regulation of Renal Function
Key Hormones
Angiotensin II: Vasoconstricts arterioles, stimulates aldosterone and ADH release, increases Na+ and water reabsorption.
Aldosterone: Increases Na+ reabsorption and K+ secretion in principal cells of late DCT and collecting duct.
Antidiuretic hormone (ADH): Increases water reabsorption by inserting aquaporin-2 channels in principal cells.
Atrial natriuretic peptide (ANP): Increases GFR and excretion of Na+ and water.
Parathyroid hormone (PTH): Increases Ca2+ reabsorption in DCT.
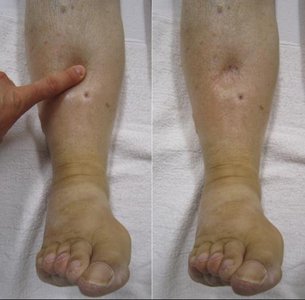
Clinical Application: Edema
Hyperaldosteronism causes excessive Na+ and water reabsorption, leading to increased blood volume and edema (fluid accumulation in tissues).