 Back
BackBiochemical Interactions in Aqueous Environments: Noncovalent Forces and the Role of Water
Study Guide - Smart Notes

Biochemical Interactions in Aqueous Environments
Importance of Noncovalent Interactions in Biochemistry
Noncovalent interactions are fundamental to the structure and function of biomolecules. Although individually weak, their collective effect provides both stability and dynamic flexibility to macromolecules, allowing essential biological processes such as molecular recognition, folding, and assembly.
Weakness and reversibility: Noncovalent bonds can be broken and re-formed continuously, enabling dynamic biological processes.
Summation of energies: In macromolecules, the sum of many weak interactions results in sufficient stability.
Types: Includes charge-charge, dipole-dipole, van der Waals, and hydrogen bonds.
Types of Noncovalent Interactions
Noncovalent interactions are fundamentally electrostatic and can be classified based on the nature of the interacting species.
Charge-charge: Interaction between two ions.
Charge-dipole: Interaction between an ion and a polar molecule.
Dipole-dipole: Interaction between two polar molecules.
Charge-induced dipole: Interaction between an ion and a nonpolar molecule.
Dipole-induced dipole: Interaction between a polar and a nonpolar molecule.
Dispersion (van der Waals): Interaction between nonpolar molecules due to temporary dipoles.
Hydrogen bond: Special interaction involving a hydrogen atom bonded to an electronegative atom and another electronegative atom.
Energy of Noncovalent Interactions
The energies of noncovalent interactions are much lower than those of covalent bonds, but are sufficient to stabilize biomolecular structures.
Type of Interaction | Approximate Energy (kJ/mol) |
|---|---|
Charge–charge | 13 to 17 |
Hydrogen bond | 2 to 21 |
van der Waals | 0.4 to 0.8 |
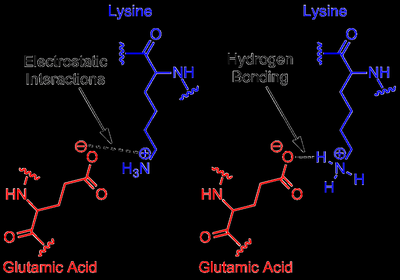
Charge-Charge Interactions
Coulomb’s Law and Biological Context
Charge-charge interactions are governed by Coulomb’s law, which describes the energy between two point charges. In biological environments, the dielectric constant (relative permittivity) of water significantly reduces the strength of these interactions.
Coulomb’s Law:
Dielectric constant: Water’s high dielectric constant weakens electrostatic interactions, facilitating solubility of ions.
Example Calculation: Ionic Bond Strength
Comparing ionic bond strengths in crystals of KCl and CaCl2 using ionic radii and Coulomb’s law:
KCl:
CaCl2:
Result: CaCl2 has stronger ionic bonds due to higher charge and shorter distance.
Dipole and Induced-Dipole Interactions
Dipole Moment
A dipole moment is a measure of the separation of positive and negative charges in a molecule. It is defined as:
Polarizable molecules: Molecules in which a dipole can be induced by an external electric field.
Examples of Dipole Moments
Some molecules, such as water, have a net dipole moment, while others, like carbon dioxide, do not.
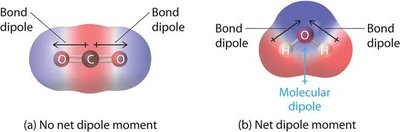
Types of Dipole Interactions
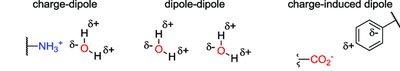
Charge-dipole: Ion interacting with a polar molecule.
Dipole-dipole: Two polar molecules interacting.
Charge-induced dipole: Ion induces a dipole in a nonpolar molecule.
Dipole-induced dipole: Polar molecule induces a dipole in a nonpolar molecule.
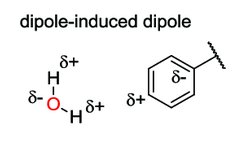
Van der Waals Interactions
Dispersion Forces and Molecular Packing
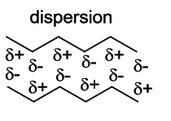
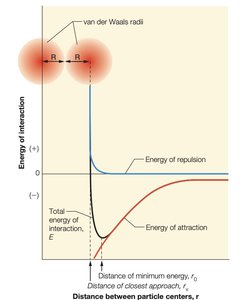
Van der Waals interactions arise from temporary fluctuations in electron distribution, leading to induced dipoles. These forces are significant only at very short range and are important for molecular packing and stability.
Dispersion forces: Occur between molecules with no net charge or permanent dipole.
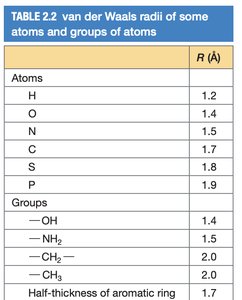
Van der Waals radius: The effective radius for closest molecular packing.
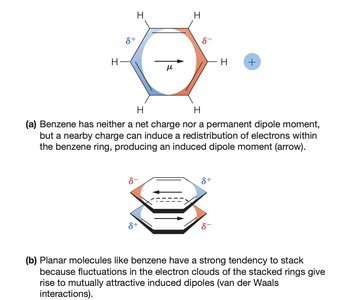
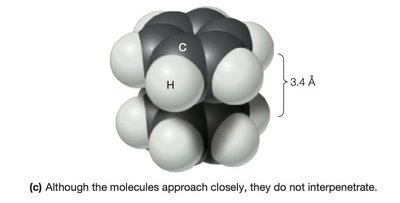
Van der Waals Radii
Atoms | R (Å) |
|---|---|
H | 1.2 |
O | 1.4 |
N | 1.5 |
C | 1.7 |
S | 1.8 |
P | 1.9 |
Hydrogen Bonds
Nature and Importance of Hydrogen Bonds
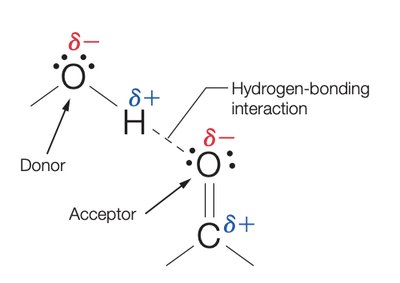
Hydrogen bonds are crucial for organizing biochemical structures, such as DNA, proteins, and water. They occur between a hydrogen atom covalently bonded to an electronegative atom and another electronegative atom with lone pairs.
Donor: Atom to which hydrogen is covalently bonded (e.g., O or N).
Acceptor: Atom with lone pairs (e.g., O or N).
Strength: Hydrogen bonds are strongest when the donor-acceptor angle is 180°.
Role of Water in Biological Processes
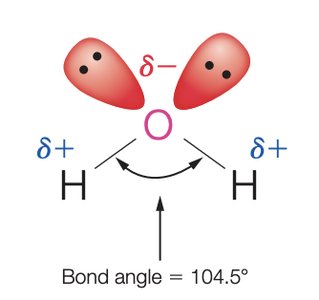
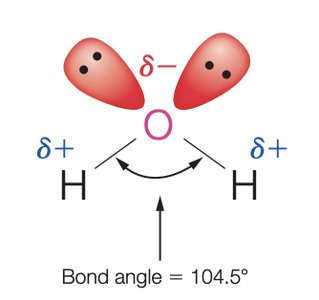
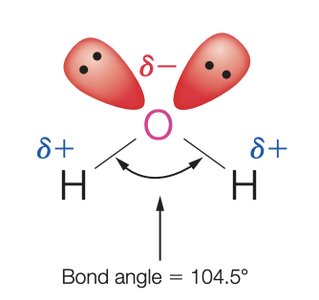
Structure and Properties of Water
Water is a polar molecule with a bent geometry and a bond angle of 104.5°. Its polarity and ability to form hydrogen bonds make it an excellent solvent for ionic and polar compounds.
High dielectric constant: Reduces electrostatic forces between ions.
Hydrogen bonding: Leads to high boiling and melting points, and high heat of vaporization.
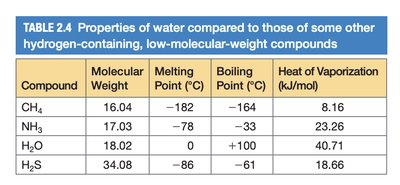
Comparison of Water with Other Compounds
Compound | Molecular Weight | Melting Point (°C) | Boiling Point (°C) | Heat of Vaporization (kJ/mol) |
|---|---|---|---|---|
CH4 | 16.04 | -182 | -164 | 8.16 |
NH3 | 17.03 | -78 | -33 | 23.26 |
H2O | 18.02 | 0 | 100 | 40.71 |
H2S | 34.08 | -86 | -61 | 18.66 |
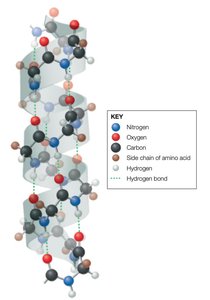
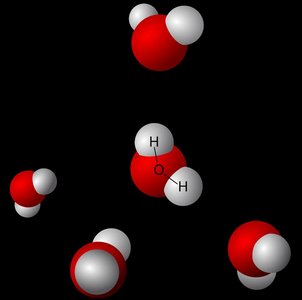
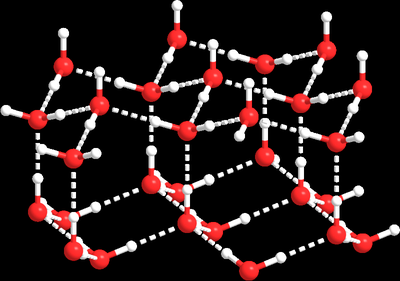
Ice Molecular Lattice
Water forms a crystalline lattice in ice, stabilized by hydrogen bonds, which accounts for its lower density compared to liquid water.
Compounds in Aqueous Solutions
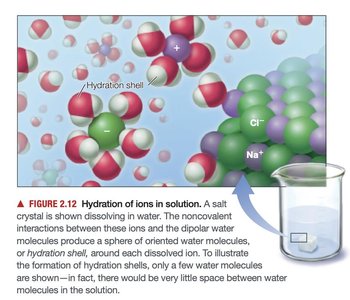
Solubility and Hydration Shells
Water is an excellent solvent for ionic compounds due to its high dielectric constant and ability to form hydration shells around ions, decreasing electrostatic forces and increasing solubility.
Hydration shell: Sphere of oriented water molecules around each dissolved ion.
Energetically favorable: Formation of hydration shells stabilizes ions in solution.
Hydrophilic, Hydrophobic, and Amphipathic Molecules
Hydrophilic vs. Hydrophobic
Hydrophilic molecules interact favorably with water, while hydrophobic molecules avoid water. Amphipathic molecules contain both hydrophilic and hydrophobic regions, enabling them to form structures like micelles and membranes.
Hydrophilic: "Water loving"; polar or charged molecules.
Hydrophobic: "Water fearing"; nonpolar molecules.
Amphipathic: Molecules with both polar and nonpolar regions.
Summary Table: Types of Noncovalent Interactions
Type | Model | Example | Distance Dependence |
|---|---|---|---|
Charge–charge | Ion pairs | Na+ and Cl- | 1/r |
Charge–dipole | Ion and polar molecule | NH3+ and H2O | 1/r2 |
Dipole–dipole | Two polar molecules | H2O and H2O | 1/r3 |
Charge–induced dipole | Ion and nonpolar molecule | CO2- and benzene | 1/r4 |
Dipole–induced dipole | Polar and nonpolar molecule | H2O and benzene | 1/r5 |
Dispersion (van der Waals) | Temporary dipoles | Stacked aromatic rings | 1/r6 |
Hydrogen bond | Donor and acceptor | O–H···O | Fixed bond length |
Additional info: Academic context and expanded explanations were added to ensure completeness and clarity for biochemistry students.
