 Back
BackBiochemistry Foundations: Functional Groups, Macromolecules, and Chemical Interactions
Study Guide - Smart Notes

Common Functional Groups in Biomolecules
Organic Compounds and Functional Groups
Biomolecules are composed of specific functional groups that determine their chemical properties and biological functions. Understanding these groups is fundamental to biochemistry.
Alcohols: Contain a hydroxyl group (–OH) attached to a carbon atom.
Aldehydes: Have a carbonyl group (C=O) bonded to at least one hydrogen.
Ketones: Feature a carbonyl group bonded to two carbon atoms.
Carboxylic Acids: Possess a carboxyl group (–COOH), often ionized at physiological pH.
Thiols (Sulfhydryls): Contain a –SH group, important in protein structure.
Amines: Classified as primary, secondary, or tertiary based on the number of alkyl groups attached to the nitrogen. At physiological pH, amines are often protonated (as ammonium ions).
Phosphates and Phosphoryl Groups: Key in energy transfer and signaling.
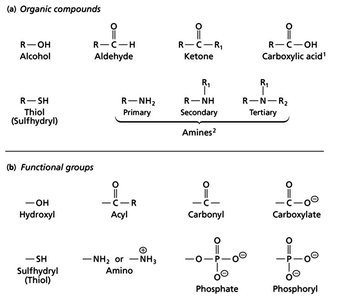
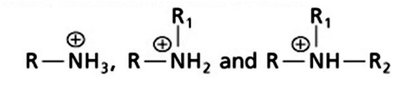
Additional info: Functional groups are the reactive parts of molecules and are essential for the structure and function of biomolecules.
Classes of Biological Macromolecules
Major Macromolecule Types
Biological macromolecules are classified into four main types, each with unique structures and functions:
Proteins: Polymers of amino acids, responsible for catalysis, structure, and regulation.
Carbohydrates: Polymers of sugars, serve as energy sources and structural components.
Nucleic Acids: DNA and RNA, polymers of nucleotides, store and transmit genetic information.
Lipids: Fatty acids and derivatives, form membranes and act as energy storage.
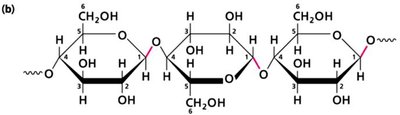
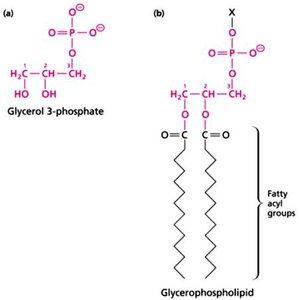
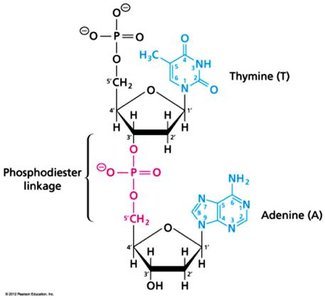
Additional info: Each macromolecule class exhibits both covalent and non-covalent interactions, crucial for their biological roles.
Covalent and Non-Covalent Interactions in Biomolecules
Types and Strengths of Chemical Bonds
Biomolecules are stabilized by both covalent and non-covalent interactions. Covalent bonds involve electron sharing, while non-covalent interactions include:
Charge–charge interactions: Electrostatic attractions between charged groups.
Hydrogen bonds: Formed between H donors and acceptors, critical in water and biomolecular structure.
van der Waals interactions: Weak attractions due to transient dipoles in non-polar molecules.
Hydrophobic interactions: Entropy-driven association of non-polar surfaces in aqueous environments.
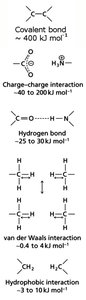
Example: Hydrophobic interactions are key in protein folding and membrane formation.
Hydrogen Bonds in Biomolecular Interactions
Major Types of Hydrogen Bonds
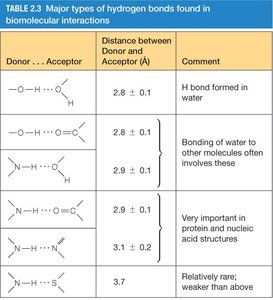
Hydrogen bonds are essential for the structure and function of proteins and nucleic acids. Their strength and geometry depend on the donor and acceptor atoms.
Donor...Acceptor | Distance (Å) | Comment |
|---|---|---|
O–H...O | 2.8 ± 0.1 | H bond formed in water |
O–H...N | 2.8 ± 0.1 | Bonding of water to other molecules |
N–H...O | 2.9 ± 0.1 | Important in protein and nucleic acid structures |
N–H...N | 3.1 ± 0.1 | Less common, weaker |
S–H...O | 3.7 | Rare, weaker |
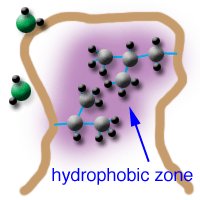
Hydrophobic Effects in Biomolecules
Role and Mechanism
Hydrophobic effects drive the association of non-polar molecules, leading to protein folding, membrane formation, and molecular recognition.
Non-polar molecules cluster together to minimize contact with water.
This increases the entropy of water, making the process thermodynamically favorable.
Chemistry of Carbon in Living Organisms
Properties and Importance
Carbon's ability to form four covalent bonds allows for complex molecular structures. Its neutrality in electronegativity makes pure hydrocarbons hydrophobic, but the addition of O, N, S, and P introduces polarity and functional diversity.
Carbon forms linear, branched, and cyclic structures.
Electronegative atoms increase polarity and potential for hydrogen and ionic bonding.
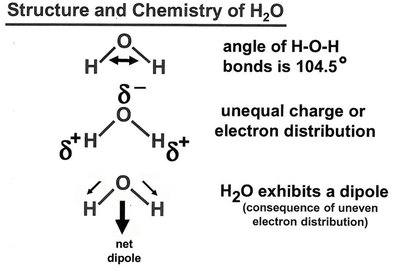
Dipole Moments and Molecular Polarity
Quantitative Determination
The dipole moment () of a molecule is determined by the difference in electronegativity between atoms and the vector sum of individual bond dipoles. It is measured in Debye units (D).
Water ( D), Ammonia ( D), Methanol ( D)
Arrows in molecular diagrams point toward the negative end of the dipole.
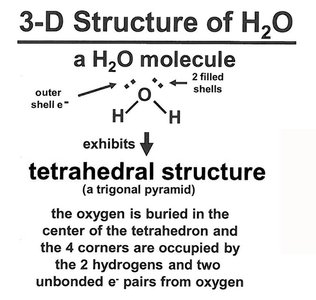
Structure and Properties of Water
Geometry and Hydrogen Bonding
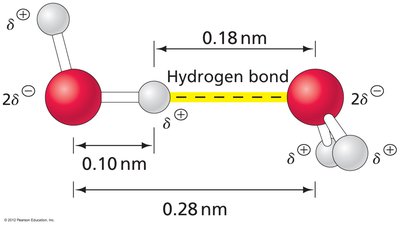
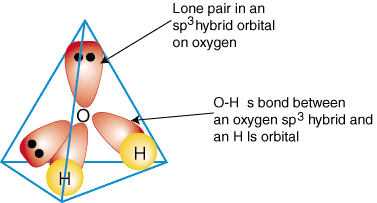
Water has a bent structure with an angle of 104.5°, resulting in a net dipole. Its tetrahedral geometry allows for extensive hydrogen bonding, making it a universal solvent in biological systems.
Each water molecule can form up to four hydrogen bonds.
Hydrogen bonds are nearly linear and contribute to water's high boiling point and solvent properties.
Acids, Bases, and Buffer Systems
Definitions and Mathematical Description
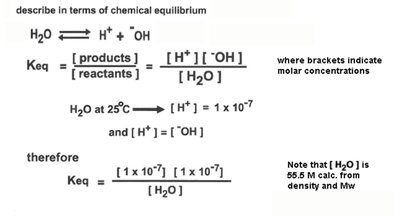
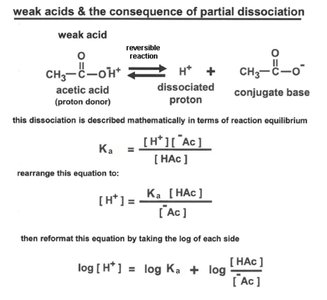
Acids are proton donors, bases are proton acceptors. The strength of acids and bases is characterized by their dissociation constants ( for acids, for bases).
Strong acids: Completely dissociate in water (e.g., HCl).
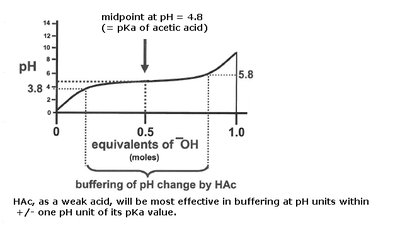
Weak acids: Partially dissociate (e.g., acetic acid).
pKa: The pH at which an acid is half dissociated.
The Henderson-Hasselbalch equation relates pH, pKa, and the ratio of conjugate base to acid:
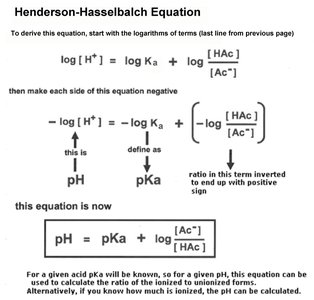
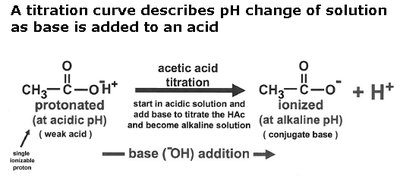
Amino Acids: Structure and Properties
General Structure and Ionization
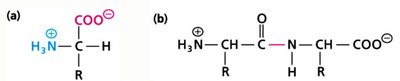
Amino acids are the building blocks of proteins. They contain a central alpha carbon, an amino group, a carboxyl group, and a variable side chain (R group). At physiological pH, amino acids exist as zwitterions.
19 amino acids have a common structure; proline is an exception with a cyclic ring.
Ionizable groups determine the pKa and isoelectric point (pI) of amino acids.
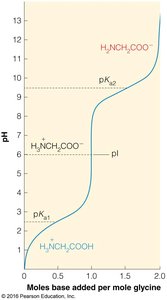
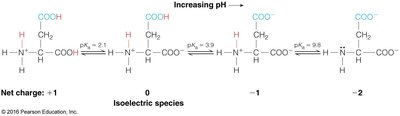
Example: The pI is calculated as the average of the pKa values surrounding the neutral species.
Peptide Bond Formation and Protein Structure
Peptide Bond and Levels of Structure
Peptide bonds are formed by condensation reactions between amino acids, resulting in polypeptides. Proteins have four levels of structure:
Primary: Sequence of amino acids.
Secondary: Local folding (alpha helices, beta sheets) stabilized by hydrogen bonds.
Tertiary: Overall 3D folding of the polypeptide.
Quaternary: Association of multiple polypeptide chains.
Protein Secondary Structure: Alpha Helix and Beta Sheet
Alpha Helix
The alpha helix is a right-handed coil stabilized by hydrogen bonds between the C=O of residue i and the NH of residue i+4. All backbone C=O and NH groups participate in hydrogen bonding except at the ends.
3.6 residues per turn, translation per residue of 1.5 Å.
Helical dipole moment: 3.5 Debye units per residue.
Beta Sheet
Beta sheets are formed by hydrogen bonding between strands. They can be parallel or antiparallel, with side chains alternating above and below the sheet.
Protein Folding and Free Energy
Thermodynamics of Folding
Protein folding is driven by a balance of enthalpy and entropy, resulting in a modest negative free energy change (). Hydrophobic interactions and internal bonding contribute to stability.
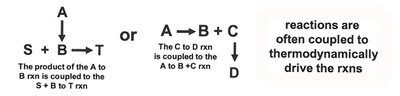
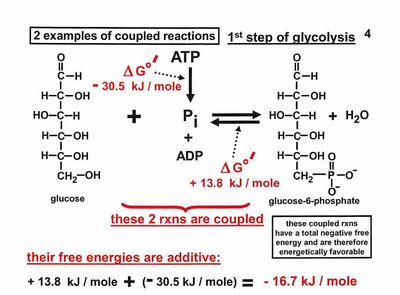
Coupling of Biochemical Reactions
Energetically Unfavorable Reactions
Biochemical reactions can be coupled to drive unfavorable processes. The total free energy change is the sum of individual reactions.
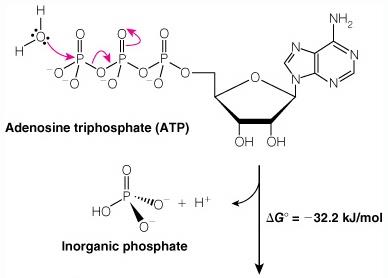
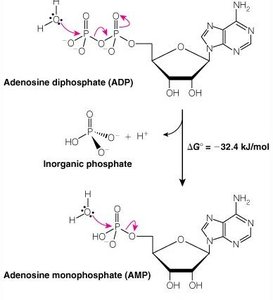
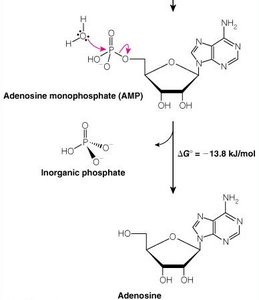
ATP and Phosphoryl Group Transfers
ATP Hydrolysis
ATP is the universal energy currency in cells. Hydrolysis of ATP releases energy by breaking high-energy phosphoanhydride bonds.
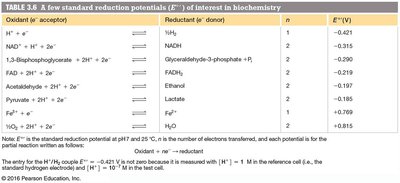
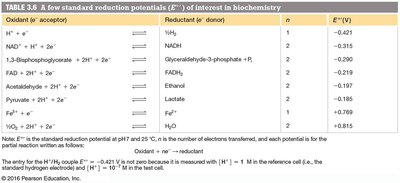
Oxidation-Reduction Reactions in Biochemistry
Redox Reactions and Standard Reduction Potentials
Redox reactions involve electron transfer. The direction is determined by standard reduction potentials (), which can be used to calculate free energy changes:
n = number of electrons transferred
F = Faraday constant (96.48 kJ V-1 mol-1)
Amino Acid Structure and Properties
Absorption Spectra and Disulfide Bonds
Aromatic amino acids (phenylalanine, tyrosine, tryptophan) absorb in the near-UV region, useful for protein quantification. Cysteine can form disulfide bonds, important for protein stability.
Protein Structure Levels and Folding
Primary, Secondary, Tertiary, and Quaternary Structure
Proteins exhibit hierarchical structure, from sequence to complex assemblies. Folding is stabilized by internal interactions and hydrophobic effects.

Ligand Binding and Allostery in Proteins
Myoglobin and Hemoglobin
Myoglobin and hemoglobin bind oxygen, with hemoglobin showing cooperative binding and allosteric regulation. The binding curve for hemoglobin is sigmoidal, reflecting changes in quaternary structure.
Allosteric effectors (e.g., 2,3-BPG) modulate hemoglobin activity.
Bohr effect: Lower pH decreases O2 affinity, promoting release.
Additional info: Ligand binding equations and cooperative effects are central to understanding protein function in biochemistry.
