Back
BackBiochemistry Foundations: Water, Carbohydrates, Lipids, Proteins, Nucleic Acids, and ATP
Study Guide - Smart Notes

Biochemistry: The Chemical Basis of Life
Introduction to Biochemistry
Biochemistry is the study of the chemical composition and reactions that occur in living matter. It distinguishes between inorganic compounds (such as water, salts, acids, and bases, which generally do not contain carbon) and organic compounds (such as carbohydrates, lipids, proteins, and nucleic acids, which contain carbon and are typically large and covalently bonded). Both types of compounds are essential for life.
Inorganic Compounds
Water
Water is the most abundant inorganic compound in living cells, accounting for 60–80% of cell volume. Its unique properties make it vital for life:
High heat capacity: Absorbs and releases heat with minimal temperature change, preventing sudden temperature shifts.
High heat of vaporization: Requires significant energy to evaporate, providing an effective cooling mechanism.
Polar solvent properties: Dissolves and dissociates ionic substances, forms hydration layers around charged molecules, and serves as the body's major transport medium.
Reactivity: Participates in hydrolysis and dehydration synthesis reactions.
Cushioning: Protects organs from physical trauma (e.g., cerebrospinal fluid cushions the nervous system).
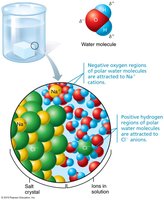
Salts
Salts are ionic compounds that dissociate into cations and anions in water (excluding H+ and OH–). All ions are electrolytes because they conduct electrical currents in solution. Key ions such as sodium, potassium, calcium, and iron play specialized roles in physiological functions. Maintaining ionic balance is crucial for homeostasis.
Acids and Bases
Acids and bases are electrolytes that ionize and dissociate in water:
Acids: Proton donors that release H+ ions (e.g., HCl, acetic acid, carbonic acid).
Bases: Proton acceptors that pick up H+ ions or release OH– (e.g., bicarbonate, ammonia).
pH Scale
The pH scale measures the concentration of hydrogen ions [H+] in a solution, ranging from 0 (most acidic) to 14 (most basic). Each unit represents a tenfold difference in [H+].
Acidic solutions: pH 0–6.99, high [H+]
Neutral solutions: pH 7, equal [H+] and [OH–]
Alkaline (basic) solutions: pH 7.01–14, low [H+]

Neutralization and Buffers
Neutralization: Mixing acids and bases forms water and a salt.
Buffers: Resist abrupt changes in pH by releasing or binding H+ ions. The carbonic acid–bicarbonate system is a key buffer in blood.
Clinical Note: Enzymes function within a narrow pH range; significant deviations can be life-threatening.
Organic Compounds: Synthesis and Hydrolysis
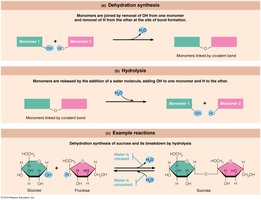
Polymerization and Breakdown
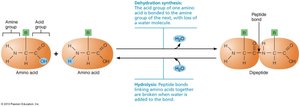
Many organic compounds are polymers—chains of similar units called monomers. They are synthesized by dehydration synthesis (removal of water to form bonds) and broken down by hydrolysis (addition of water to break bonds).
Carbohydrates
Classification and Structure
Carbohydrates are sugars and starches containing C, H, and O (with H:O ratio of 2:1). They are classified as:
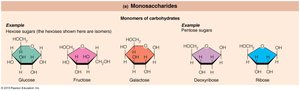
Monosaccharides: Simple sugars (3–7 carbons), e.g., glucose, fructose, galactose, ribose, deoxyribose.
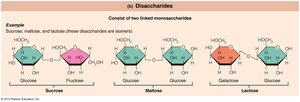
Disaccharides: Double sugars, formed by dehydration synthesis of two monosaccharides (e.g., sucrose, maltose, lactose).
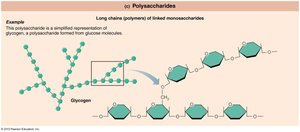
Polysaccharides: Long chains of monosaccharides (e.g., starch in plants, glycogen in animals).
Monosaccharides
Monomers of carbohydrates, important for energy metabolism and as building blocks for nucleic acids.
Disaccharides
Formed by joining two monosaccharides; too large to pass through cell membranes without hydrolysis.
Polysaccharides
Polymers of monosaccharides, used for energy storage and structural support. Starch is used by plants, glycogen by animals.
Lipids
Types and Functions
Lipids are hydrophobic molecules containing C, H, O (less O than carbohydrates), and sometimes P. Main types include:
Triglycerides: Three fatty acids bonded to glycerol; function in energy storage, insulation, and protection.
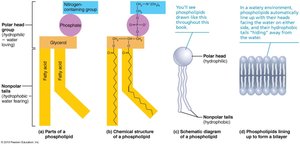
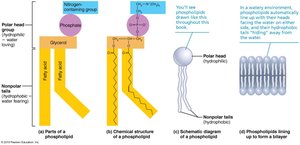
Phospholipids: Glycerol, two fatty acids, and a phosphate group; major component of cell membranes.
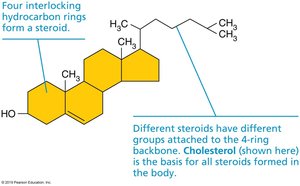
Steroids: Four interlocking hydrocarbon rings; cholesterol is the most important, serving as a precursor for hormones and vitamin D.
Eicosanoids: Derived from arachidonic acid; include prostaglandins, which regulate inflammation and other processes.
Triglycerides
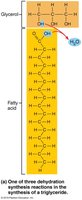
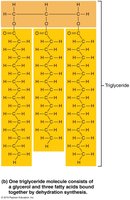

Saturated vs. Unsaturated Fatty Acids
Saturated fatty acids: Only single bonds between carbons; solid at room temperature (e.g., butter).
Unsaturated fatty acids: One or more double bonds; liquid at room temperature (e.g., olive oil).
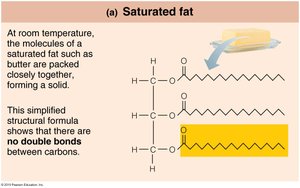
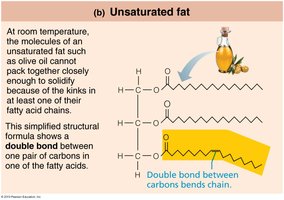
Phospholipids
Phospholipids have a hydrophilic (water-attracting) head and hydrophobic (water-repelling) tails, making them essential for forming biological membranes.
Steroids
Steroids have a characteristic four-ring structure. Cholesterol is a key steroid, forming the basis for other steroids in the body.
Proteins
Structure and Function
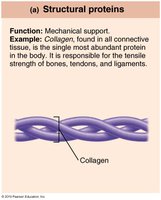
Proteins are polymers of amino acids joined by peptide bonds. They contain C, H, O, N, and sometimes S and P. Proteins perform a vast array of functions, including structural support, catalysis, transport, movement, communication, and defense.
Examples of Protein Functions
Type | Function | Example |
|---|---|---|
Structural | Support | Collagen |
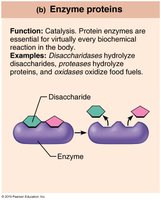
Enzyme | Catalysis | Disaccharidases, proteases |
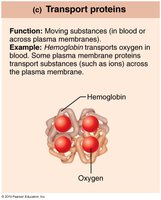
Transport | Move substances | Hemoglobin |
Contractile | Movement | Actin, myosin |
Communication | Signals | Insulin |
Defensive | Protection | Antibodies |
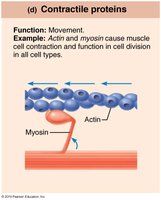
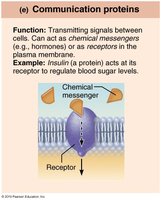
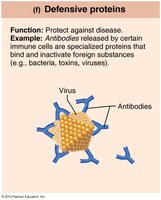
Amino Acids and Peptide Bonds
All proteins are made from 20 amino acids, each with an amine group, acid group, and a unique "R group." Peptide bonds form between the acid group of one amino acid and the amine group of another.
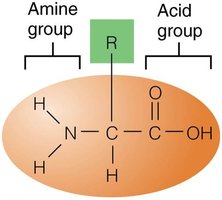
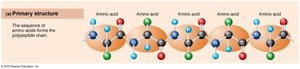
Levels of Protein Structure
Primary: Linear sequence of amino acids.
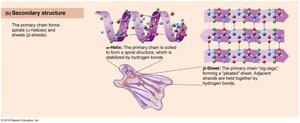
Secondary: Alpha helices and beta sheets formed by hydrogen bonding.
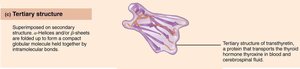
Tertiary: 3D folding due to interactions among R groups.
Quaternary: Association of two or more polypeptide chains.
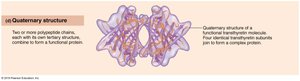
Protein Denaturation
Denaturation is the loss of a protein's functional 3D shape due to changes in pH or temperature. This process is usually reversible unless the changes are extreme.
Enzymes and Enzyme Kinetics
Enzyme Structure and Function
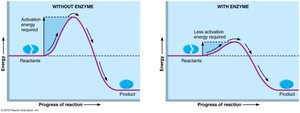
Enzymes are globular proteins that act as biological catalysts, increasing the speed of chemical reactions by lowering activation energy. Most enzymes are holoenzymes, consisting of an apoenzyme (protein) and a cofactor (metal ion or coenzyme).
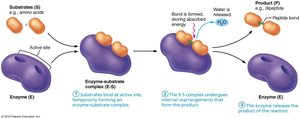
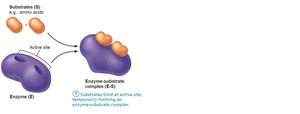
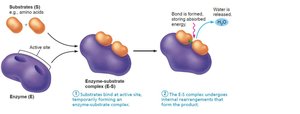
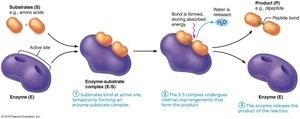
Mechanism of Enzyme Action
Substrate binds to the enzyme's active site, forming an enzyme-substrate complex.
The complex undergoes rearrangement, forming the product.
The product is released, and the enzyme is free to catalyze another reaction.
Nucleic Acids
Structure and Function
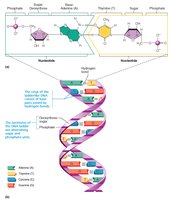
Nucleic acids (DNA and RNA) are polymers of nucleotides, each composed of a nitrogenous base, a pentose sugar, and a phosphate group. They store and transmit genetic information.
DNA: Double-stranded helix, stores genetic blueprint, bases are adenine (A), guanine (G), cytosine (C), and thymine (T).
RNA: Single-stranded, involved in protein synthesis, bases are adenine (A), guanine (G), cytosine (C), and uracil (U).
ATP (Adenosine Triphosphate)
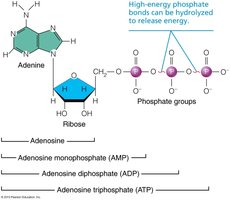
Structure and Role
ATP is the primary energy carrier in cells. It consists of an adenine-containing RNA nucleotide with two additional phosphate groups. Energy is released when the terminal phosphate bond is broken, converting ATP to ADP or AMP.
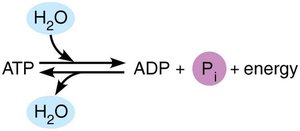
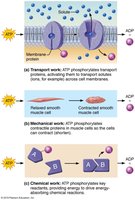
Cellular Work Driven by ATP
Transport work: ATP phosphorylates transport proteins to move substances across membranes.
Mechanical work: ATP phosphorylates contractile proteins for muscle contraction.
Chemical work: ATP provides energy for chemical reactions.