 Back
BackBiochemistry Study Guide: Thermodynamics, Metabolism, and Enzyme Function
Study Guide - Smart Notes

Introduction to Biochemistry: Thermodynamics and ATP
Thermodynamics in Biological Systems
Thermodynamics is the study of energy and its transformations, which is fundamental to understanding biochemical reactions and cellular processes.
Energy: The capacity to do work or produce heat. In biochemistry, energy is required for cellular functions and is transferred through metabolic pathways.
Kinetic vs. Potential Energy:
Kinetic Energy: Energy of motion (e.g., movement of molecules).
Potential Energy: Stored energy (e.g., chemical bonds in molecules).
First Law of Thermodynamics: Energy cannot be created or destroyed, only transformed.
Second Law of Thermodynamics: Entropy (disorder) increases in spontaneous processes.
Free Energy and Biological Reactions
Free energy determines whether a reaction will occur spontaneously. In biochemistry, the change in free energy (ΔG) is crucial for understanding metabolic pathways.
Gibbs Free Energy (ΔG): The energy available to do work in a system.
Formula:
Exergonic Reactions: Release energy (ΔG < 0), spontaneous.
Endergonic Reactions: Require energy input (ΔG > 0), non-spontaneous.
ATP: Adenosine triphosphate, the primary energy carrier in cells.
Metabolism and ATP
Adenosine Triphosphate (ATP)
ATP is a nucleotide that stores and transfers energy within cells. It consists of adenine, ribose, and three phosphate groups.
Structure: Contains high-energy phosphate bonds.
Hydrolysis of ATP: Releases energy for cellular processes.
ATP Cycle: ATP is regenerated from ADP and inorganic phosphate.
Types of Metabolic Pathways
Metabolic pathways are categorized based on their function in the cell.
Catabolic Pathways: Breakdown molecules to release energy (e.g., glycolysis).
Anabolic Pathways: Build complex molecules from simpler ones, requiring energy (e.g., protein synthesis).
Enzymes: Structure, Function, and Regulation
Enzyme Basics
Enzymes are biological catalysts that speed up chemical reactions without being consumed. They are essential for metabolic processes.
Active Site: Region on the enzyme where substrate binds.
Substrate: The molecule upon which an enzyme acts.
Enzyme Specificity: Enzymes are specific to their substrates due to the shape and chemical properties of the active site.

Enzyme Catalysis
Enzymes lower the activation energy required for reactions, increasing the rate of reaction.
Mechanism: Enzymes stabilize the transition state, facilitate substrate binding, and promote product formation.
Lock-and-Key Model: Substrate fits precisely into the enzyme's active site.
Induced Fit Model: Enzyme changes shape to accommodate the substrate.
Enzyme Regulation
Enzyme activity is regulated to meet the needs of the cell.
Allosteric Regulation: Binding of molecules at sites other than the active site alters enzyme activity.
Feedback Inhibition: End product of a pathway inhibits an earlier enzyme.
Positive Regulation: Activators increase enzyme activity.
Enzyme Kinetics
Michaelis-Menten Kinetics
Michaelis-Menten kinetics describe the relationship between substrate concentration and reaction rate for many enzymes.
Michaelis-Menten Equation:
Vmax: Maximum reaction rate.
Km: Substrate concentration at which the reaction rate is half of Vmax; indicates enzyme affinity for substrate.

Enzyme Inhibition
Enzyme inhibitors decrease enzyme activity and are classified based on their mechanism.
Competitive Inhibition: Inhibitor binds to the active site, preventing substrate binding.
Non-Competitive Inhibition: Inhibitor binds elsewhere, changing enzyme shape and reducing activity.
Uncompetitive Inhibition: Inhibitor binds only to the enzyme-substrate complex.
Cooperativity and Temperature Effects
Cooperativity
Cooperativity occurs when the binding of a substrate to one active site affects the binding of additional substrates to other active sites, often seen in multimeric enzymes.
Positive Cooperativity: Binding of one substrate increases affinity for others.
Negative Cooperativity: Binding of one substrate decreases affinity for others.
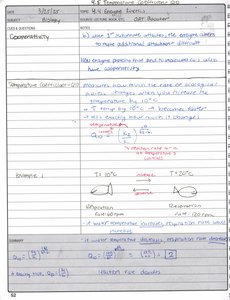
Temperature Coefficient (Q10)
The Q10 coefficient measures the rate of change of a biological or chemical system as a consequence of increasing the temperature by 10°C.
Formula: , where R2 and R1 are reaction rates at two different temperatures.
Application: Used to compare metabolic rates in organisms at different temperatures.
Enzyme Nomenclature
Classification of Enzymes
Enzymes are classified based on the type of reaction they catalyze.
Oxidoreductases: Catalyze oxidation-reduction reactions.
Transferases: Transfer functional groups between molecules.
Hydrolases: Catalyze hydrolysis reactions.
Lyases: Break bonds without water.
Isomerases: Catalyze isomerization reactions.
Ligases: Join two molecules together.
Kinases and Phosphatases
Kinases add phosphate groups to molecules, while phosphatases remove them. These enzymes are crucial for regulating cellular activities.
Kinase: Transfers phosphate from ATP to a substrate.
Phosphatase: Removes phosphate groups from substrates.
Summary Table: Enzyme Classes and Functions
Enzyme Class | Function | Example |
|---|---|---|
Oxidoreductase | Oxidation-reduction | Lactate dehydrogenase |
Transferase | Group transfer | Hexokinase |
Hydrolase | Hydrolysis | Protease |
Lyase | Bond breaking (no water) | Aldolase |
Isomerase | Isomerization | Phosphoglucoisomerase |
Ligase | Bond formation | DNA ligase |
Additional info: These notes cover foundational biochemistry topics including thermodynamics, metabolism, ATP, enzyme structure and function, enzyme kinetics, inhibition, regulation, cooperativity, temperature effects, and enzyme nomenclature. All content is directly relevant to the biochemistry college course outline.
