 Back
BackBiological Macromolecules: Structure, Function, and Cellular Context
Study Guide - Smart Notes

Biological Macromolecules
Introduction to Biological Macromolecules
Living systems are primarily composed of a limited set of chemical elements, with carbon (C), hydrogen (H), oxygen (O), and nitrogen (N) being the most abundant. Other essential elements include sulfur (S), phosphorus (P), and various ions such as sodium (Na+), potassium (K+), magnesium (Mg2+), calcium (Ca2+), and chloride (Cl-). These elements form the basis of four major classes of biological macromolecules: nucleic acids, proteins, polysaccharides, and lipids. Each class is built from smaller organic subunits called monomers, which are linked by specific types of covalent bonds through dehydration synthesis (condensation reactions).
Nucleic Acids
Structure and Function of Nucleic Acids
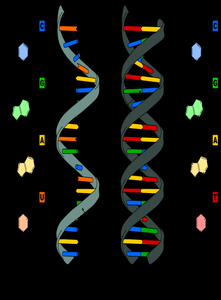
Nucleic acids, including DNA and RNA, are polymers known as polynucleotides. Their primary function is the storage, transmission, and expression of genetic information. DNA serves as the genetic blueprint within the nucleus, while RNA is involved in the readout and translation of this information.
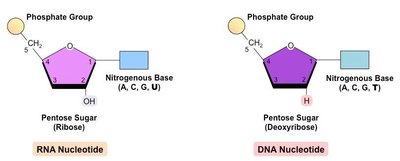
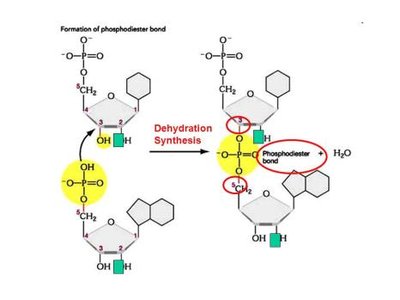
Monomer: Nucleotide (composed of a pentose sugar, a phosphate group, and a nitrogenous base)
Linkage: Phosphodiester bond (between the 3' carbon of one sugar and the 5' carbon of the next)
Key Functions: Information storage (DNA), information transfer and expression (RNA)
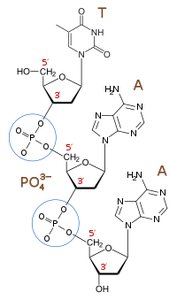
Example: The human genome contains billions of nucleotides organized into chromosomes, with DNA molecules reaching molecular masses of up to 20 billion Daltons.
DNA and RNA: Structural Differences
DNA: Double-stranded helix, deoxyribose sugar, bases A, T, G, C
RNA: Single-stranded, ribose sugar, bases A, U, G, C
Proteins
Structure and Function of Proteins
Proteins are polymers of amino acids linked by peptide (amide) bonds. They exhibit remarkable structural diversity and perform a wide range of biological functions, including catalysis (enzymes), structural support, transport, signaling, and immune defense.
Monomer: Amino acid
Linkage: Peptide bond (amide bond)
Key Functions: Enzymatic catalysis, structural support, transport, signaling, immune response
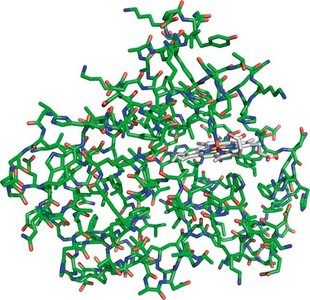
Example: Myoglobin, a globular protein, binds and stores oxygen in muscle tissue. Proteins can range from 10,000 to 1,000,000 Da in size and are the most abundant macromolecules in cells like E. coli.
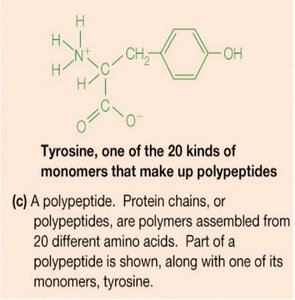
Amino Acids: Structure and Properties
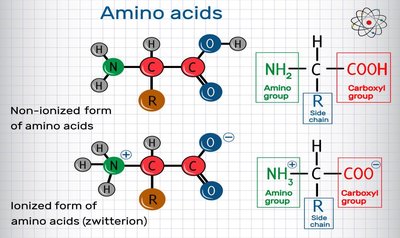
All amino acids have a central (α) carbon, an amino group, a carboxyl group, a hydrogen atom, and a variable side chain (R group).
At physiological pH (~7), amino acids exist as zwitterions (NH3+ and COO-).
There are 20 common amino acids, classified by the properties of their side chains (nonpolar, polar, acidic, basic, aromatic).
Peptide Bond Formation
Amino acids are joined by peptide bonds through a condensation reaction, releasing water. The peptide bond is planar due to partial double-bond character, restricting rotation and influencing protein structure.
Polysaccharides (Carbohydrates)
Structure and Function of Polysaccharides
Polysaccharides are long chains of monosaccharide units linked by glycosidic bonds. They serve as energy storage (e.g., starch, glycogen) and structural components (e.g., cellulose in plants, chitin in fungi and arthropods).
Monomer: Monosaccharide (e.g., glucose)
Linkage: Glycosidic bond (ether linkage)
Key Functions: Energy storage, structural support, cell recognition
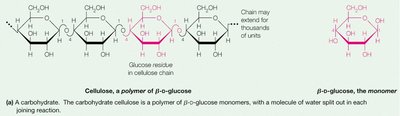
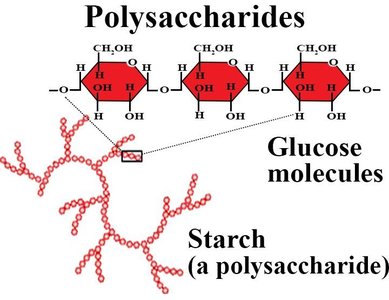
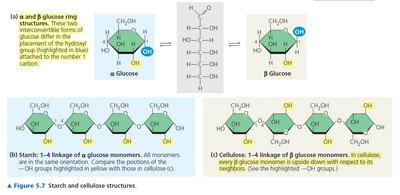
Example: Cellulose is a β(1→4)-linked polymer of glucose, forming rigid plant cell walls. Starch is an α(1→4)-linked polymer, serving as an energy reserve in plants.
Lipids
Structure and Function of Lipids
Lipids are a chemically diverse group of hydrophobic molecules, including fats, phospholipids, and steroids. They are not true polymers but form large complexes. Lipids are essential for energy storage, membrane structure, and signaling.
Monomer: Fatty acid (for many lipids)
Linkage: Ester bond (in triacylglycerols and phospholipids)
Key Functions: Energy storage, membrane structure, signaling molecules
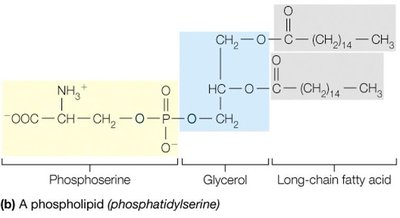
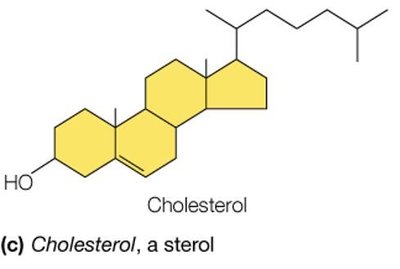
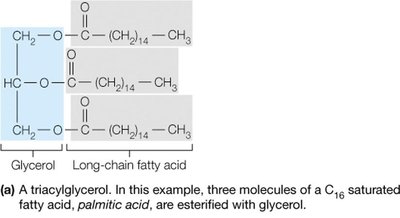
Example: Triacylglycerols (triglycerides) store energy, phospholipids form the basis of cellular membranes, and cholesterol is a membrane component and precursor to steroid hormones.
Biological Membranes and Amphipathic Lipids
Phospholipids are amphipathic, containing both hydrophobic (fatty acid tails) and hydrophilic (phosphate head) regions. In aqueous environments, they spontaneously form bilayers, which are the structural foundation of cellular membranes.
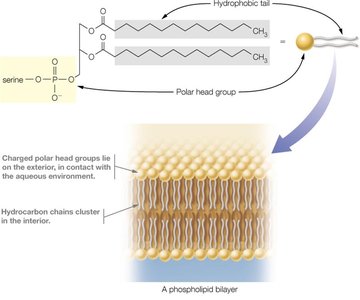
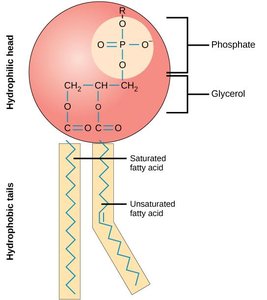
Cellular Organization
Types of Cells
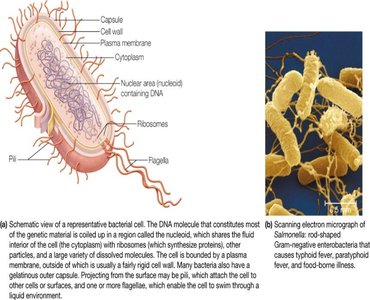
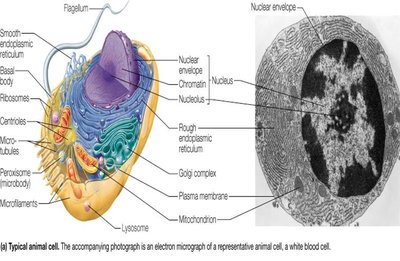
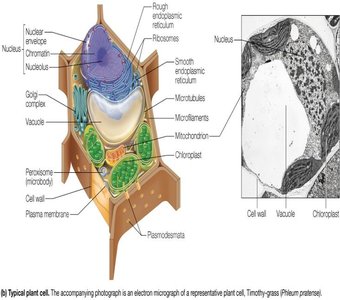
Cells are the universal unit of life and are classified into three domains: Bacteria, Archaea (both prokaryotic), and Eukarya (eukaryotic). Prokaryotic cells lack internal compartmentalization, while eukaryotic cells contain membrane-bound organelles.
Prokaryotic Cells: No nucleus, DNA in cytosol, surrounded by plasma membrane and cell wall, contain ribosomes, may have pili and flagella.
Eukaryotic Cells: Larger, contain nucleus, mitochondria, endoplasmic reticulum, Golgi complex, and other organelles. Animal cells lack cell walls; plant cells have cell walls, chloroplasts, and large vacuoles.
Summary Table: Biological Macromolecules
Macromolecule | Monomer | Linkage |
|---|---|---|
Nucleic acids | Nucleotide | Phosphodiester |
Proteins | Amino acid | Peptide (amide) |
Polysaccharides | Monosaccharide | Glycosidic (ether) |
Lipids | Fatty acid | Ester |
Additional info: The diversity and complexity of biological macromolecules underlie the structure and function of all living cells. Their assembly, properties, and interactions are central to biochemistry and molecular biology.
