 Back
BackBuffer Systems and pH Regulation in Biochemistry
Study Guide - Smart Notes

Buffer Systems and pH Regulation in Biochemistry
The Zwitterionic Nature of Amino Acids
Amino acids, the building blocks of proteins, can exist in different ionic forms depending on the pH of their environment. At a specific pH unique to each amino acid, they exist as zwitterions—molecules with both positive and negative charges but an overall neutral charge.
Zwitterion: A molecule with both a positively charged (protonated amine) and a negatively charged (deprotonated carboxyl) group.
pH Dependence: At low pH, amino acids are fully protonated and positively charged; at high pH, they are deprotonated and negatively charged.
Isoelectric Point (pI): The pH at which the amino acid has no net charge.
Protonation/Deprotonation: The process by which amino acids gain or lose protons, altering their charge state.
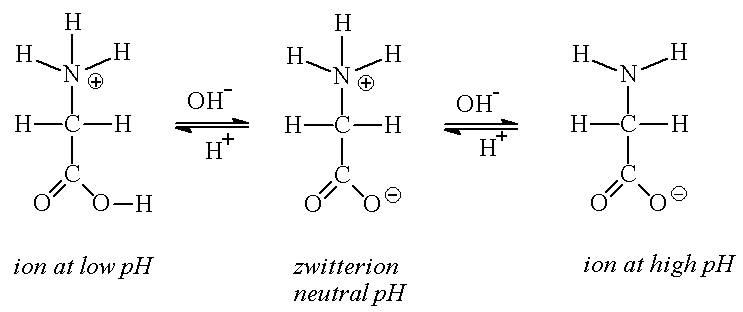
Example: Glycine at low pH is fully protonated (positive charge), at neutral pH is a zwitterion (net zero charge), and at high pH is deprotonated (negative charge).
Molecular Charge and pH: The Isoelectric Point
The overall charge of a protein or biomolecule depends on the pH of its environment. The isoelectric point (pI) is the pH at which the molecule carries no net electrical charge. This property is crucial for protein solubility and function.
Below pI: The molecule is positively charged.
Above pI: The molecule is negatively charged.
At pI: The molecule has a net charge of zero.

Example: Proteins in blood have a pI around 6.8; at physiological pH (~7.4), they are slightly negatively charged.
Importance of Blood pH Regulation
Maintaining blood pH within a narrow range (7.35–7.45) is essential for proper cellular function. Deviations can denature enzymes, disrupt metabolic processes, and lead to severe health consequences.
Enzyme Denaturation: Extreme pH can unfold proteins, disabling their catalytic activity.
Homeostasis: The body uses buffer systems to resist drastic changes in pH.
The Bicarbonate Buffer System in Blood
The primary buffer system in human blood is the bicarbonate buffer, which involves carbonic acid (H2CO3) and bicarbonate ion (HCO3-). This system helps maintain blood pH by neutralizing excess acids or bases.
CO2 Transport: CO2 from cellular respiration is converted to carbonic acid in red blood cells, which then dissociates to bicarbonate and hydrogen ions.
Buffer Reaction: H2CO3 ↔ HCO3- + H+
pH Regulation: The buffer system resists changes in pH when acids or bases are introduced.
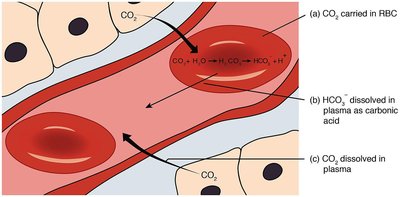
Mechanism of Buffer Action in Blood
Buffers in blood neutralize added acids or bases, preventing significant pH changes. When an acid is added, bicarbonate neutralizes it; when a base is added, carbonic acid neutralizes it.
Acid Addition: HCO3- + H+ → H2CO3
Base Addition: H2CO3 + OH- → HCO3- + H2O
Result: Hydronium or hydroxide ions are removed, stabilizing pH.

Buffer Systems: Definitions and Principles
A buffer is a solution that resists changes in pH when small amounts of acid or base are added. Buffers typically consist of a weak acid and its conjugate base, or a weak base and its conjugate acid.
Weak Acid Buffer: HA ↔ H+ + A-
Weak Base Buffer: B + H+ ↔ HB+
Buffer Capacity: The amount of acid or base a buffer can neutralize before pH changes significantly.
The Henderson–Hasselbalch Equation
The Henderson–Hasselbalch equation relates the pH of a buffer solution to the concentration of acid and its conjugate base. It is essential for calculating the pH of buffer systems in biochemistry.
Equation:
Application: Used to determine the pH of blood and other biological fluids.
When [A-] = [HA]: pH = pKa
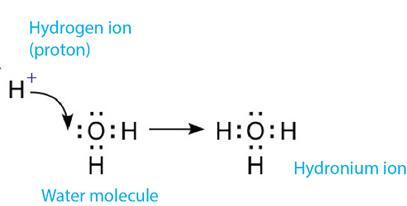
Acid Dissociation and Hydronium Ion Formation
Acids dissociate in water to produce hydronium ions (H3O+), which are central to pH calculations and buffer action.
Acid Dissociation: HA ↔ H+ + A-
Hydronium Ion: H2O + H+ → H3O+
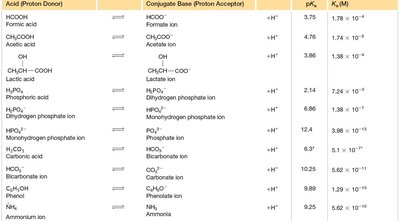
Common Biological Buffer Systems
Several weak acids and their conjugate bases serve as important buffers in biological systems. Their effectiveness depends on their pKa values, which should be close to the desired pH.
Acid (Proton Donor) | Conjugate Base (Proton Accepter) | pKa | Ka (M) |
|---|---|---|---|
HCOOH (Formic acid) | HCOO- (Formate ion) | 3.75 | 1.78 × 10-4 |
CH3COOH (Acetic acid) | CH3COO- (Acetate ion) | 4.76 | 1.74 × 10-5 |
H2CO3 (Carbonic acid) | HCO3- (Bicarbonate ion) | 6.35 | 4.45 × 10-7 |
NH4+ (Ammonium ion) | NH3 (Ammonia) | 9.25 | 5.62 × 10-10 |
Example: The bicarbonate buffer system is effective in blood because its pKa is close to physiological pH.
Practice Problems: Buffer Calculations
Understanding buffer systems requires the ability to calculate pH changes upon addition of acids or bases. The following are typical problems encountered in biochemistry:
Binding of Ca2+ to Carboxyl Groups: Predict at which pH Ca2+ binds most tightly to a carboxylic acid group with pKa 4.2.
pH of Ammonium Chloride Solution: Calculate the pH of 1.0 M NH4Cl solution.
Buffer Response to Strong Acid/Base: Determine the new pH after adding HCl or NaOH to an acetic acid/acetate buffer.
Buffer Mixtures: Calculate the pH of mixtures such as acetic acid with sodium acetate, or phosphoric acid with monopotassium phosphate.
Key Formula:
Example: For a buffer with equal concentrations of acetic acid and acetate, pH = pKa = 4.76.
Additional info: For all calculations, use the Henderson–Hasselbalch equation and consider the effect of dilution and addition of strong acids or bases on buffer capacity.
