 Back
BackCarbohydrates: Structure, Stereochemistry, Cyclization, and Biological Roles
Study Guide - Smart Notes

Carbohydrates: Structure and Classification
Definition and General Properties
Carbohydrates are polyhydroxy aldehydes or ketones, with the general formula Cn(H2O)n. They are produced from CO2 and H2O via photosynthesis in plants and serve as energy sources, structural components, and informational molecules in cell signaling. Carbohydrates can be covalently linked to proteins and lipids.
Classification: Based on the number of carbon atoms (triose, tetrose, pentose, hexose, etc.) and the type of carbonyl group (aldose or ketose).
Functional Groups: All carbohydrates initially contain a carbonyl group, either as an aldehyde (aldose) or a ketone (ketose).
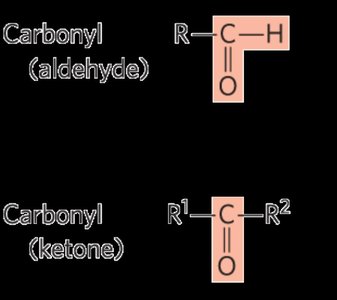
Examples of Simple Carbohydrates
Glyceraldehyde: The simplest aldose (triose).
Dihydroxyacetone: The simplest ketose (triose).
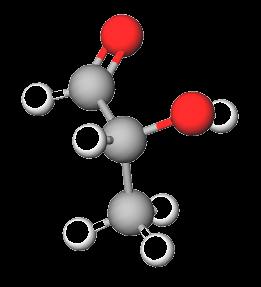
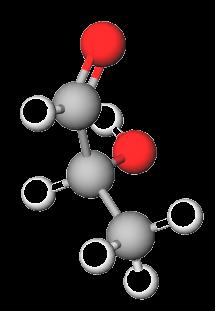
Stereochemistry of Carbohydrates
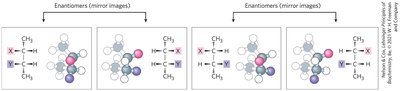
Chirality and Isomerism
Carbohydrates often contain chiral centers, leading to multiple stereoisomers. The D- and L- forms are distinguished by their ability to rotate polarized light: D (dextrorotatory) rotates light clockwise, L (levorotatory) rotates light counterclockwise. Most natural sugars are D-isomers.
Enantiomers: Mirror-image isomers, opposite at all chiral centers.
Diastereomers: Stereoisomers that are not mirror images, differ at some but not all chiral centers.
Epimers: Diastereomers that differ at only one chiral center.
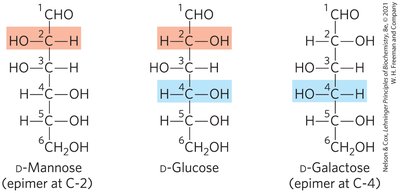
Epimers of Glucose
D-mannose and D-galactose are epimers of D-glucose, differing at C-2 and C-4 respectively. Epimers are not mirror images and have distinct physical properties.
Physical Properties and Reactivity of Carbohydrates
Reducing Properties
Aldehydes and ketones are strong reductants, and thus aldoses and ketoses are generally reducing sugars. However, their reducing ability is less than expected due to their structure. Carbonyl groups absorb UV and IR light, but carbohydrate solutions lack strong absorbance bands in these regions.
Cyclization and Glycosidic Bond Formation
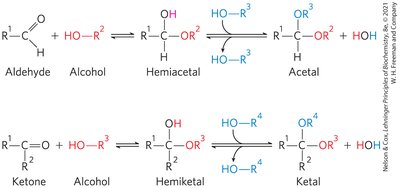
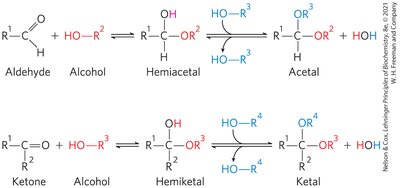
Hemiacetals and Hemiketals
Carbohydrates contain both carbonyl and alcohol groups, allowing intramolecular reactions to form hemiacetals (from aldoses) and hemiketals (from ketoses). This reactivity forms the basis for sugar cyclization.
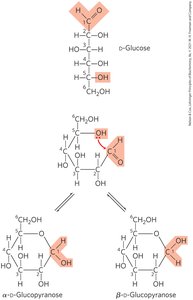
Cyclization of Monosaccharides
Pentoses and hexoses readily cyclize, creating a new chiral center called the anomeric carbon. The position of the hydroxyl group on the anomeric carbon determines the α or β anomer.
α-anomer: Hydroxyl group opposite to the CH2OH moiety.
β-anomer: Hydroxyl group on the same side as the CH2OH moiety.
Mutarotation
When glucose is dissolved in water, the optical rotation changes as equilibrium is reached between α, β, and linear forms. This phenomenon is called mutarotation.
Polymerization and Glycosidic Bonds
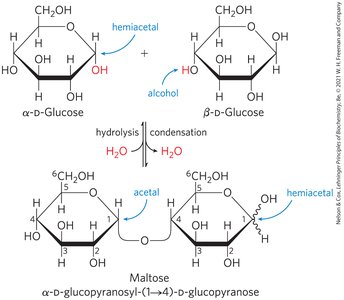
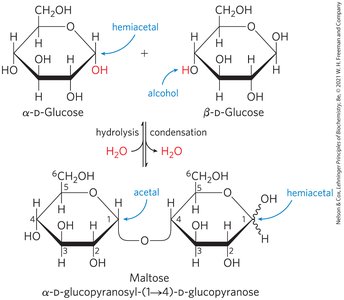
Formation of Glycosidic Bonds
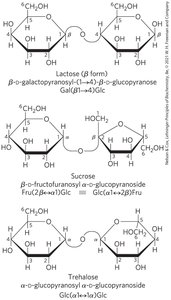
Monosaccharides can react via their hemiacetal/hemiketal and hydroxyl groups to form glycosidic bonds, resulting in disaccharides and polysaccharides. The glycosidic bond is more stable and less reactive than the original hemiacetal/hemiketal.
Reducing disaccharides: One free hemiacetal/hemiketal group remains, retaining some reducing ability.
Non-reducing disaccharides: Both anomeric carbons are involved in the bond, no reducing ends.
Polysaccharides: Structure and Function
Types of Polysaccharides
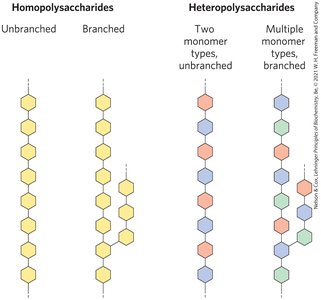
Polysaccharides are natural carbohydrate polymers, classified as homopolysaccharides (one repeating monomer) or heteropolysaccharides (multiple monomers). They can be linear or branched, and their molecular weight is not defined due to dynamic polymerization.
Starch and Glycogen
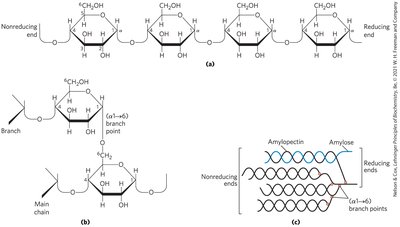
Starch: Main storage polysaccharide in plants, composed of amylose (unbranched, α1→4) and amylopectin (branched, α1→6 every 24–30 residues).
Glycogen: Main storage polysaccharide in animals, branched (α1→6 every 8–12 residues).
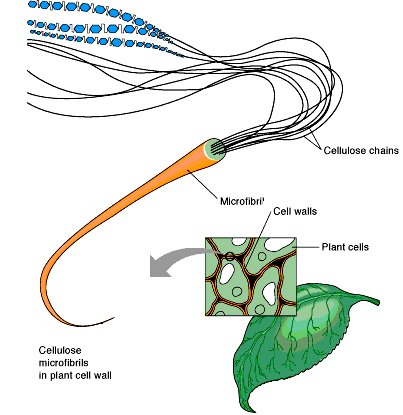
Cellulose
Cellulose is a linear homopolysaccharide of glucose with β1→4 linkages. Hydrogen bonds between monomers and chains provide structural strength and water insolubility, making cellulose the most abundant polysaccharide in nature.
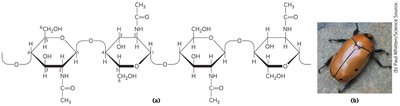
Chitin
Chitin is a linear homopolysaccharide of N-acetylglucosamine, with β1→4 linkages. It forms tough, flexible, water-insoluble fibers found in fungal cell walls and arthropod exoskeletons.
Agar and Agarose
Agar is a branched heteropolysaccharide from seaweeds, used in bacterial culture media. Agarose, a component of agar, forms gels for DNA electrophoresis. Agarose consists of D-galactose and 3,6-anhydro-L-galactose units joined by alternating β1→4 and α1→3 bonds.
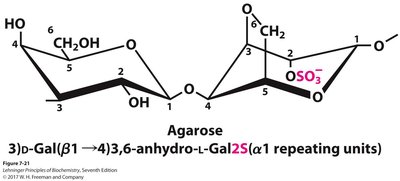
Table: Structures and Roles of Some Polysaccharides
Polysaccharide | Type | Repeating Unit | Size | Role/Significance |
|---|---|---|---|---|
Starch (Amylose/Amylopectin) | Homo | α1→4 Glc, α1→6 branches | 50–5,000 / up to 106 | Energy storage in plants |
Glycogen | Homo | α1→4 Glc, α1→6 branches | Up to 50,000 | Energy storage in animals |
Cellulose | Homo | β1→4 Glc | Up to 15,000 | Structural in plants |
Chitin | Homo | β1→4 GlcNAc | Very large | Structural in exoskeletons |
Dextran | Homo | α1→6 Glc, α1→3 branches | Wide range | Structural in bacteria |
Peptidoglycan | Hetero | Mur2Ac(β1→4)GlcNAc | Very large | Structural in bacteria |
Agarose | Hetero | D-Gal(β1→4)3,6-anhydro-L-Gal(α1) | 1,000 | Structural in algae |
Hyaluronan | Hetero | GlcA(β1→3)GlcNAc(β1) | Up to 100,000 | Structural in vertebrates |
Monosaccharide Derivatives and Glycoconjugates
Monosaccharide Derivatives
Carbohydrates are highly functionalized due to their hydroxyl groups. Important derivatives include deoxyribose (DNA), glucosamine (dietary supplement), and various hexose derivatives involved in biological processes.
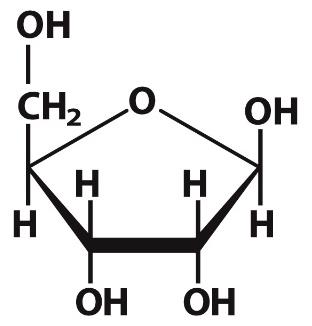
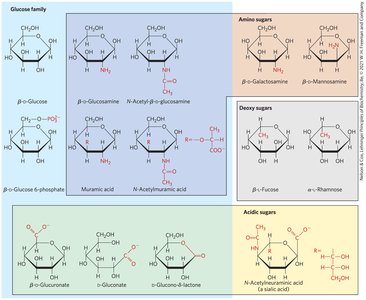
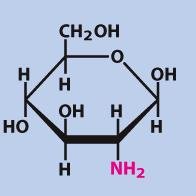
Glucosamine
Glucosamine is a functionalized form of glucose, with an amine substituting a hydroxyl group. It is commonly used as a dietary supplement and forms linkers between carbohydrates and proteins/lipids.

Glycoconjugates: Glycolipids and Glycoproteins
Glycolipids are lipids with covalently bound oligosaccharides, important in cell signaling and recognition. Glycoproteins are proteins with attached oligosaccharides, playing roles in protein-protein recognition and immune evasion by viruses.
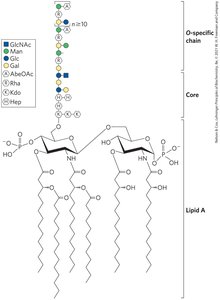
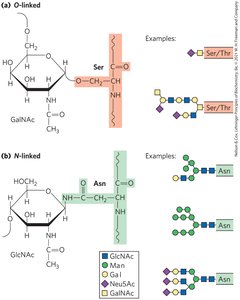
Extracellular Matrix (ECM)
Structure and Function
The ECM is a structural material outside cells, providing strength, elasticity, and a physical barrier in tissues. Its main components are proteoglycan aggregates, collagen fibers, and elastin. ECM acts as a barrier to tumor cell invasion.
Summary
Structures and properties of important monosaccharides
Monosaccharide cyclization and mutarotation
Glycosidic bond formation and types of disaccharides
Structures and biological roles of key polysaccharides and sugar derivatives
