 Back
BackChatper 10
Study Guide - Smart Notes

10. Lipids
Introduction to Lipids
Lipids are a structurally diverse class of organic molecules characterized by their low solubility in water, making them relatively hydrophobic. They play essential roles in energy storage, membrane structure, insulation, signaling, and more.
Hydrophobicity: Lipids are insoluble or only sparingly soluble in water due to their long hydrocarbon chains or rings.
Structural Diversity: Lipids include fatty acids, triacylglycerols, phospholipids, sphingolipids, sterols, and more.

Biological Functions of Lipids
Energy Storage: Lipids, especially triacylglycerols, store energy efficiently due to their highly reduced state and hydrophobic nature, allowing dense packing in lipid droplets. Beta-oxidation of fatty acids generates acetyl-CoA for ATP production.
Insulation: Lipids provide thermal insulation and mechanical protection, absorbing shocks and maintaining body temperature.
Water Repellency: The hydrophobic nature of lipids prevents excessive wetting and water loss in organisms (e.g., bird feathers, plant leaves).
Buoyancy and Acoustics: In marine mammals, lipids contribute to buoyancy and may play roles in sound transmission.
Membrane Structure: Lipids are the main structural components of biological membranes.
Cofactors and Signaling: Lipids serve as enzyme cofactors (e.g., vitamin K, coenzyme Q), hormones (steroids), and signaling molecules (e.g., paracrine hormones, vitamins A and D).
Pigments and Antioxidants: Some lipids function as pigments (carotenoids) and antioxidants (vitamin E).
Lipids as Pigments
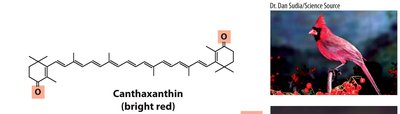
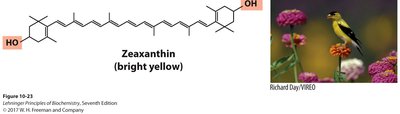
Compounds with long conjugated systems, such as carotenoids, absorb visible light and provide pigmentation in plants and animals. Birds acquire pigments like canthaxanthin (red) and zeaxanthin (yellow) from their diet, leading to sexual dimorphism in coloration due to differences in uptake and processing.
Classification of Lipids
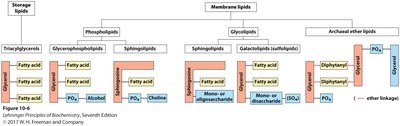
Lipids are classified based on the presence or absence of fatty acids and their biological roles:
Lipids containing fatty acids (complex lipids): Includes storage lipids (e.g., triacylglycerols) and membrane lipids (e.g., phospholipids, glycolipids).
Lipids not containing fatty acids: Includes cholesterol, vitamins, and pigments.
Fatty Acids
Structure and Properties
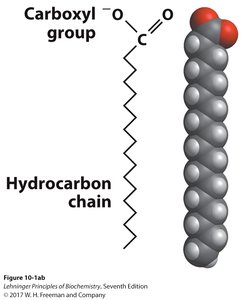
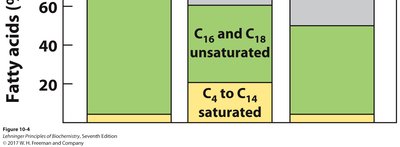
Fatty acids are carboxylic acids with hydrocarbon chains (typically 4–36 carbons). Most natural fatty acids have even numbers of carbons and are unbranched.
Saturated fatty acids: No double bonds in the hydrocarbon chain.
Monounsaturated fatty acids: One double bond in the chain.
Polyunsaturated fatty acids: More than one double bond.
Cis and Trans Double Bonds
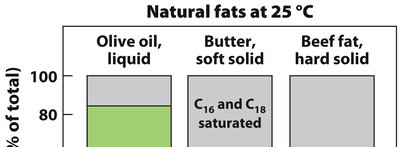
Natural unsaturated fatty acids usually have cis double bonds, introducing kinks and reducing packing efficiency. Trans fatty acids, often produced industrially, allow tighter packing and have higher melting points, but are associated with health risks.
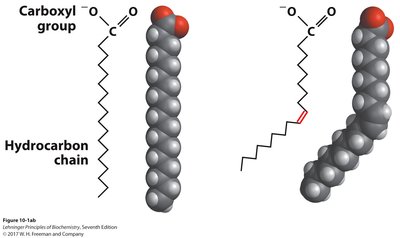
Nomenclature of Fatty Acids
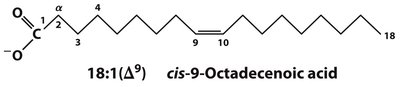
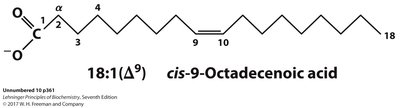
Systematic name: Indicates the configuration and position of double bonds (e.g., cis-9-octadecenoic acid).
Common name: Traditional names (e.g., oleic acid).
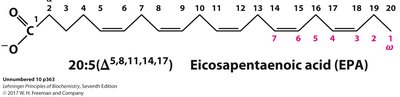
Delta (Δ) numbering: Number of carbons: number of double bonds (Δ position of double bonds from carboxyl end).
Omega (ω) numbering: Position of double bond from the methyl (omega) end.
Physical Properties
Solubility: Decreases with increasing chain length.
Melting Point: Increases with chain length; decreases with more double bonds.
Essential Fatty Acids
Omega-3 fatty acids (e.g., alpha-linolenic acid, eicosapentaenoic acid, docosahexaenoic acid) are essential nutrients for humans, required in the diet as they cannot be synthesized de novo.
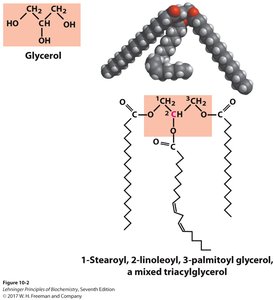
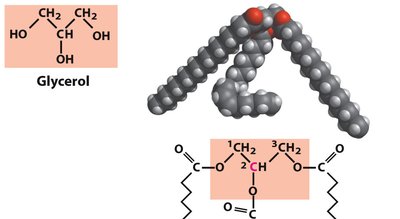
Triacylglycerols (Triglycerides)
Structure and Function
Triacylglycerols are the main storage form of fatty acids in biological systems. They consist of three fatty acids esterified to a glycerol backbone. Fats are solid at room temperature, while oils are liquid.
Energy Storage: Triacylglycerols are highly efficient energy stores, more reduced than carbohydrates and less hydrated, allowing compact storage.
Physical Properties: Less soluble in water than free fatty acids due to esterification; less dense than water.
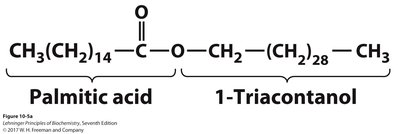
Waxes
Structure and Biological Roles
Waxes are esters of long-chain fatty acids with long-chain alcohols. They are highly hydrophobic, insoluble in water, and have high melting points.
Functions: Energy storage in plankton, waterproofing in plants and animals, protection and pliability for skin and hair, and use in human products (lotions, polishes).

Structural Lipids in Membranes
Membrane Lipid Diversity
Membrane lipids are amphipathic, containing both hydrophilic (polar) head groups and hydrophobic (nonpolar) tails. The main classes include glycerophospholipids, galactolipids, sulfolipids, archaeal ether lipids, sphingolipids, and sterols.
Glycerophospholipids: Two fatty acids and a phosphate group attached to glycerol.
Galactolipids/Sulfolipids: Predominate in plant membranes; contain galactose or sulfonated sugars.
Archaeal Ether Lipids: Found in archaea; ether linkages and long chains confer stability in extreme environments.
Sphingolipids: Contain a sphingosine backbone, fatty acid, and polar head group.
Sterols: Four fused rings; structural role in eukaryotic membranes.

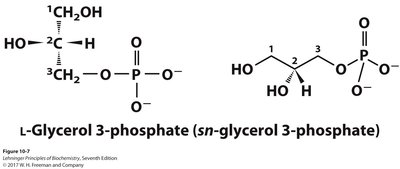
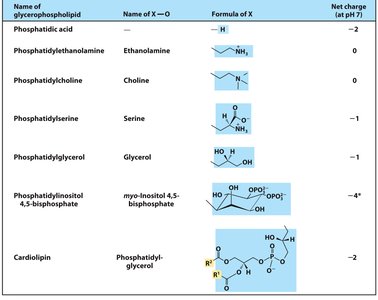
Glycerophospholipids
These are the primary constituents of cell membranes. They have two fatty acids esterified to L-glycerol-3-phosphate, with a polar phosphate head group that may be further modified by an alcohol (the head group).
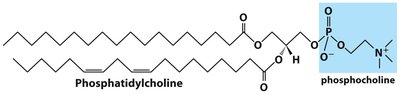
Phosphatidylcholine
Phosphatidylcholine is the most abundant phospholipid in eukaryotic cell membranes. Many prokaryotes lack this lipid.
Sphingolipids
Sphingolipids have a sphingosine backbone, a fatty acid attached via an amide linkage, and a polar head group. Glycosphingolipids are important in cell recognition and signaling, especially in the outer leaflet of plasma membranes.
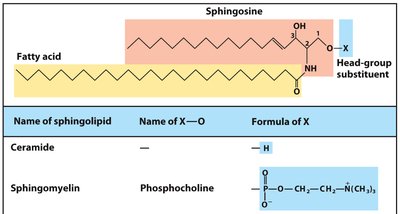
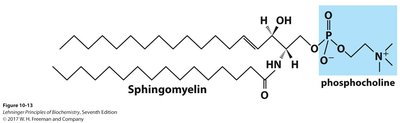
Sphingomyelin
Sphingomyelin is a type of sphingolipid with a phosphocholine head group, abundant in the myelin sheath of nerve cells.
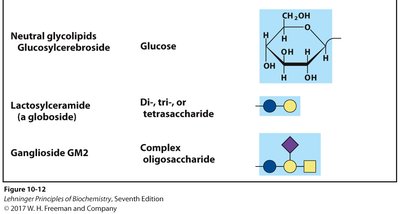
Glycosphingolipids and Blood Groups
Blood group antigens are determined by the specific sugars present on glycosphingolipids in the plasma membrane. The expression of different glycosyltransferases leads to the O, A, or B blood group antigens.
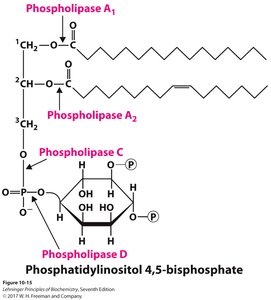
Lipid Degradation in Lysosomes
Membrane lipids are continually degraded and replaced. Phospholipases cleave specific bonds in phospholipids, while gangliosides are degraded by sequential enzymatic cleavage. Defects in these pathways can lead to lysosomal storage disorders.
Sterols and Cholesterol
Structure and Function
Sterols, such as cholesterol, have a characteristic structure of four fused rings (steroid nucleus), a polar hydroxyl group, and a nonpolar side chain. Cholesterol modulates membrane fluidity and serves as a precursor for steroid hormones and bile acids.
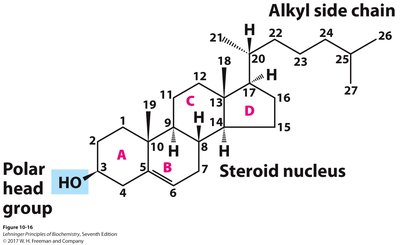
Physiological Role of Sterols
Membrane Component: Cholesterol is present in most eukaryotic membranes, affecting fluidity and permeability.
Precursor: Cholesterol is the precursor for steroid hormones, vitamin D, and bile acids.
Transport: Cholesterol is transported in the blood bound to proteins (lipoproteins); excess can lead to atherosclerosis.
Steroid Hormones
Steroid hormones are oxidized derivatives of sterols, lacking the alkyl side chain of cholesterol and being more polar. They are synthesized in the gonads and adrenal glands and transported in the bloodstream bound to carrier proteins. Examples include testosterone, cortisol, and estradiol.
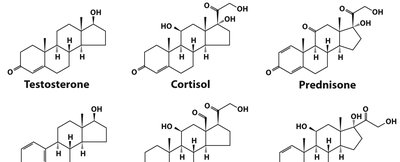
Biologically Active Lipids
Signaling Molecules and Vitamins
Biologically active lipids, present in small amounts, play crucial roles as signaling molecules (e.g., eicosanoids, prostaglandins) and as lipid-soluble vitamins (A, D, E, K). Vitamin D is synthesized from cholesterol and regulates calcium metabolism, while vitamin A (retinol) is derived from β-carotene and is essential for vision and hormone signaling.
Summary Table: Selected Fatty Acids
Common Name | Systematic Name | Structure | Carbon Skeleton | Melting Point (°C) |
|---|---|---|---|---|
Lauric acid | n-Dodecanoic acid | CH3(CH2)10COOH | 12:0 | 44.2 |
Palmitic acid | n-Hexadecanoic acid | CH3(CH2)14COOH | 16:0 | 63.1 |
Stearic acid | n-Octadecanoic acid | CH3(CH2)16COOH | 18:0 | 69.6 |
Oleic acid | cis-9-Octadecenoic acid | CH3(CH2)7CH=CH(CH2)7COOH | 18:1(Δ9) | 13.4 |
Linoleic acid | cis,cis-9,12-Octadecadienoic acid | CH3(CH2)4CH=CHCH2CH=CH(CH2)7COOH | 18:2(Δ9,12) | -5 |
Additional info: This summary table is based on the provided data and standard biochemistry references.
