 Back
BackComprehensive Study Notes on Proteins and Amino Acids
Study Guide - Smart Notes

Proteins: Definition and Biological Importance
Overview of Proteins
Proteins are essential macromolecules found in every cell, forming the structural framework of tissues and organs. They are polymers composed of amino acids linked by peptide bonds. All proteins are constructed from a set of 20 amino acids, which act as the building blocks.
Structural proteins (e.g., collagen, elastin) provide strength and support to tissues.
Enzymes act as biocatalysts, accelerating biochemical reactions.
Defense proteins (antibodies) protect against infections.
Transport proteins (hemoglobin, albumin) carry molecules throughout the body.
Hormonal proteins (insulin, growth hormone) regulate physiological processes.
Contractile proteins (actin, myosin) enable movement.
Storage proteins (ferritin, hemosiderin) store nutrients and ions.
Proteins regulate gene expression, function as ion channels, and stabilize DNA.
Food Sources Rich in Proteins
Dietary proteins are obtained from both animal and plant sources. Examples include fish, chicken, eggs, cashews, lentils, chickpeas, mung beans, and soybeans. 
Amino Acids: Structure and Classification
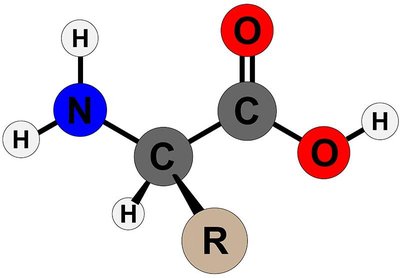
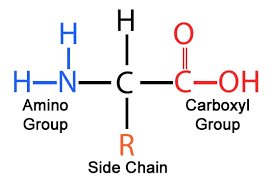
General Structure of Amino Acids
Amino acids are the monomers of proteins. Each amino acid consists of a central α-carbon bonded to an amino group (–NH₂), a carboxyl group (–COOH), a hydrogen atom, and a variable side chain (R group).
At physiological pH, the carboxyl group is deprotonated (–COO⁻) and the amino group is protonated (–NH₃⁺).
The chemical properties and biological functions of proteins are determined by the R groups.
Essential and Non-Essential Amino Acids
Amino acids are classified based on whether the body can synthesize them.
Essential amino acids: 9 amino acids that must be obtained from the diet (e.g., methionine, threonine, histidine, valine, phenylalanine, isoleucine, tryptophan, lysine, leucine).
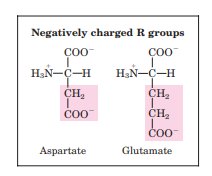
Non-essential amino acids: 11 amino acids that the body can synthesize (e.g., alanine, asparagine, glutamate, glutamine, cysteine, glycine, proline, serine, tyrosine, aspartate, arginine).


Stereochemistry of Amino Acids
Most amino acids (except glycine) are chiral, possessing an asymmetric α-carbon. They exist as two enantiomers: L- and D-forms.
L-amino acids are incorporated into proteins in living organisms.
D-amino acids are rare and found in some bacterial cell walls and peptide antibiotics.
The configuration is determined by the position of the amino group in the Fischer projection.

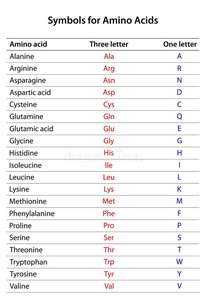
Naming and Abbreviations of Amino Acids
Each amino acid has a three-letter abbreviation and a one-letter symbol.
Amino Acid | Three Letter | One Letter |
|---|---|---|
Alanine | Ala | A |
Arginine | Arg | R |
Asparagine | Asn | N |
Aspartic acid | Asp | D |
Cysteine | Cys | C |
Glutamine | Gln | Q |
Glutamic acid | Glu | E |
Glycine | Gly | G |
Histidine | His | H |
Isoleucine | Ile | I |
Leucine | Leu | L |
Lysine | Lys | K |
Methionine | Met | M |
Phenylalanine | Phe | F |
Proline | Pro | P |
Serine | Ser | S |
Threonine | Thr | T |
Tryptophan | Trp | W |
Tyrosine | Tyr | Y |
Valine | Val | V |
Classification Based on R Groups
Amino acids are classified into five main classes based on the properties of their R groups, particularly their polarity.
Nonpolar, aliphatic: Glycine, alanine, proline, valine, leucine, isoleucine, methionine
Aromatic: Phenylalanine, tyrosine, tryptophan
Polar, uncharged: Serine, threonine, cysteine, asparagine, glutamine
Positively charged (basic): Lysine, arginine, histidine
Negatively charged (acidic): Aspartate, glutamate
Acid-Base Properties of Amino Acids
Zwitterion Concept
Amino acids can exist as zwitterions, carrying both positive (amino) and negative (carboxyl) charges in solution.
At neutral pH, amino acids are dipolar ions (zwitterions).
They can act as acids (proton donors) or bases (proton acceptors), making them amphoteric.
Isoelectric Point (pI)
The isoelectric point is the pH at which an amino acid exists as a zwitterion and has a net charge of zero.
The pI can be calculated by averaging the pKa values of the ionizable groups.
At pI, amino acids are least soluble in water and do not migrate in an electric field.
Amino Acids as Precursors of Bioactive Compounds
Neurotransmitters
Certain amino acids serve as precursors for neurotransmitters, which regulate mood, sleep, and other functions.
Tryptophan → Serotonin
Tyrosine → Dopamine, Norepinephrine, Epinephrine
Glutamate → GABA (Gamma-Aminobutyric Acid)
Hormones
Some amino acids are precursors for important hormones.
Tyrosine → Thyroid hormones (T3 & T4)
Tryptophan → Melatonin
Other Bioactive Molecules
Heme synthesis: Glycine + Succinyl-CoA → δ-Aminolevulinic acid → Heme
Creatine: Arginine + Glycine + Methionine → Creatine (energy storage in muscles)
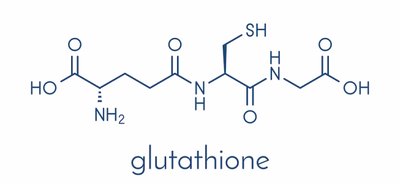
Glutathione: Glutamate + Cysteine + Glycine → Glutathione (antioxidant)
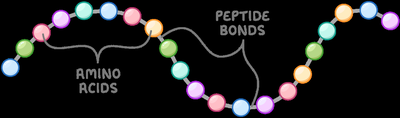
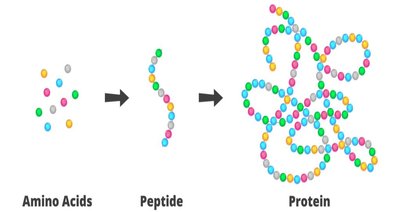
Peptides and Peptide Bond Formation
Definition and Types of Peptides
Peptides are molecules formed by linking two or more amino acids via peptide bonds. Types include dipeptides, tripeptides, oligopeptides, and polypeptides. 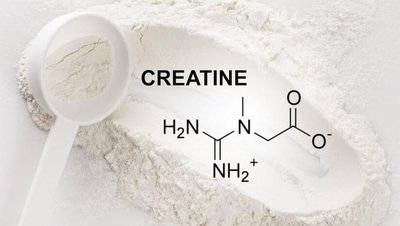
Peptide Bond Formation
Peptide bonds form between the α-carboxyl group of one amino acid and the α-amino group of another, releasing a molecule of water. This is a condensation (dehydration) reaction.
Peptide bond (amide linkage) has partial double-bond character due to resonance.
Bond length: ~1.32 Å (between single and double bond lengths).
Peptide bonds are planar and restrict rotation.
Cis and Trans Conformations
Peptide bonds can exist in cis or trans forms.
Trans form: α-carbons on opposite sides, more stable, found in most proteins.
Cis form: α-carbons on the same side, rare.
Hierarchy of Protein Structure
Primary Structure
The primary structure is the linear sequence of amino acids in a polypeptide chain, determined by DNA.
Directionality: N-terminal (–NH₃⁺) to C-terminal (–COO⁻).
Each protein has a unique primary structure.
Single amino acid substitutions can cause diseases (e.g., sickle cell anemia).
Secondary Structure
Secondary structure refers to local folding patterns stabilized mainly by hydrogen bonds.
α-Helix: Right-handed helix, stabilized by hydrogen bonds between C=O and N–H groups.
β-Pleated Sheet: Extended chain, stabilized by hydrogen bonds between adjacent strands. Can be parallel, antiparallel, or mixed.
Loops and Turns: Connect α-helices and β-sheets, often involved in functional regions.
Tertiary Structure
Tertiary structure is the three-dimensional folding of a single polypeptide chain, resulting in a compact, globular protein.
Stabilized by hydrophobic interactions, hydrogen bonds, ionic interactions, van der Waals forces, and disulfide bonds.
Domains are independent functional units within large polypeptides.
Motifs (supersecondary structures) include β–α–β, β-hairpin, and Greek key motifs.
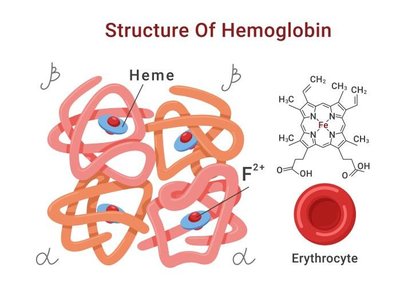
Quaternary Structure
Quaternary structure is present in proteins with more than one polypeptide chain (subunit).
Subunits may be identical or different.
Held together by non-covalent interactions and sometimes disulfide bridges.
Examples: Hemoglobin (4 subunits), Collagen (triple helix).
Protein Folding and Denaturation
Principles of Protein Folding
Protein folding is the process by which a polypeptide chain attains its native conformation.
Occurs in a stepwise manner, retaining partly correct intermediates.
Molten globule model: domains collapse into compact structures stabilized by hydrophobic interactions.
Enzymes (protein disulfide isomerase, cis–trans proline isomerase) and chaperone proteins (HSPs, chaperonins) assist folding.
Denaturation
Denaturation is the loss of native structure due to disruption of secondary, tertiary, and quaternary structures, while the primary structure remains intact.
Caused by physical (heat, UV, X-rays) and chemical agents (acids, heavy metals, urea).
Results in loss of biological activity, change in solubility, and coagulation.
Denaturation is usually irreversible, but renaturation is possible in some cases.
Methods to Prevent Denaturation
In vitro: Formaldehyde fixation, lyophilisation (freeze-drying).
In vivo: Optimum temperature and pH, buffer systems, chaperone proteins, compartmentalization, protective solutes.
Protein Extraction and Separation Techniques
Overview of Methods
Ion-Exchange Chromatography: Separates proteins based on charge.
Size Exclusion Chromatography: Separates proteins based on size.
Affinity Chromatography: Separates proteins based on specific binding interactions.
SDS-PAGE: Separates proteins based on molecular weight using polyacrylamide gel electrophoresis.
References
Berg, J.M., Tymoczko, J.L., Gatto Jr., G.J. & Stryer, L. (2015) Biochemistry. 8th ed. New York: W.H. Freeman.
Nelson, D., and Cox, M. (2005) Lehninger principles of biochemistry. 4th ed.
Rafi (2020) Biochemistry. 4th ed.
