Back
BackCoupling Reactions and ATP Hydrolysis in Biochemistry
Study Guide - Smart Notes

Coupling Reactions in Biochemistry
Energetics of Coupled Reactions
Biochemical reactions often require coupling to proceed efficiently. Many cellular processes are energetically unfavorable and must be paired with favorable reactions, such as ATP hydrolysis, to drive them forward. The phosphorylation of glucose is a classic example of this principle.
Unfavorable Reaction: Glucose + Pi → Glucose-6-phosphate (ΔG ≈ +18.0 kJ/mol)
Favorable Reaction: ATP + H2O → ADP + Pi (ΔG ≈ -30.5 kJ/mol)
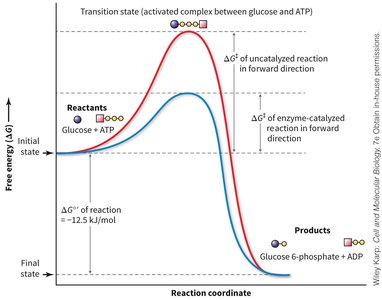
Overall Coupled Reaction: Glucose + ATP → Glucose-6-phosphate + ADP (ΔG ≈ -12.5 kJ/mol)
Key Point: The overall reaction becomes energetically favorable due to the coupling.

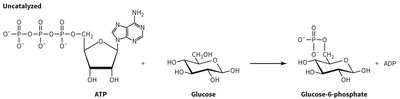
ATP Hydrolysis and Phosphorylation of Glucose
ATP hydrolysis is a central reaction in biochemistry, providing energy for many cellular processes. The transfer of a phosphate group from ATP to glucose forms glucose-6-phosphate, a key intermediate in glycolysis.
ATP Structure: Contains three phosphate groups, ribose, and adenine.
Glucose Structure: A six-carbon sugar, substrate for phosphorylation.
Product: Glucose-6-phosphate and ADP.
Importance: This reaction is the first step in glycolysis and is catalyzed by hexokinase.
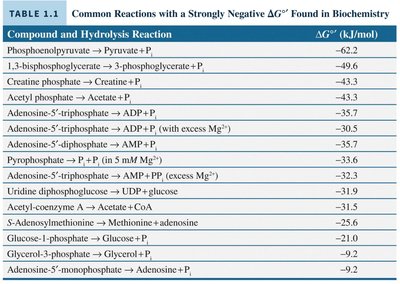
Common Reactions with Strongly Negative ΔG°'
Many biochemical reactions are characterized by strongly negative standard free energy changes (ΔG°'). These reactions are often used to drive other processes through coupling.
Compound and Hydrolysis Reaction | ΔG°' (kJ/mol) |
|---|---|
Phosphoenolpyruvate → Pyruvate + Pi | -62.2 |
1,3-bisphosphoglycerate → 3-phosphoglycerate + Pi | -49.3 |
Creatine phosphate → Creatine + Pi | -43.3 |
Acetyl phosphate → Acetate + Pi | -43.1 |
Adenosine-5'-triphosphate → ADP + Pi | -35.7 |
Adenosine-5'-triphosphate → ADP + Pi (with excess Mg2+) | -35.0 |
Adenosine-5'-triphosphate → AMP + PPi | -32.3 |
Pyrophosphate → 2 Pi (in small Mg2+) | -33.5 |
Adenosine-5'-triphosphate → AMP + PPi (excess Mg2+) | -32.3 |
Uridine diphosphoglucose → UDP + glucose | -31.5 |
Acetyl-coenzyme A → Acetate + CoA | -31.5 |
S-adenosylmethionine → Methionine + adenosine | -26.5 |
Glucose-1-phosphate → Glucose + Pi | -21.0 |
Glycerol-3-phosphate → Glycerol + Pi | -19.0 |
Adenosine-5'-monophosphate → Adenosine + Pi | -9.2 |
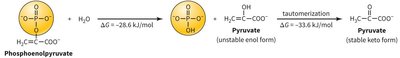
Phosphoenolpyruvate Hydrolysis
Phosphoenolpyruvate (PEP) hydrolysis is a highly exergonic reaction, contributing to the overall energetics of glycolysis. The conversion of PEP to pyruvate involves two steps: hydrolysis and tautomerization.
Hydrolysis: PEP + H2O → Pyruvate (enol form) + Pi (ΔG ≈ -26.6 kJ/mol)
Tautomerization: Pyruvate (enol form) → Pyruvate (keto form) (ΔG ≈ -33.4 kJ/mol)
Total ΔG: -60.0 kJ/mol (very favorable)
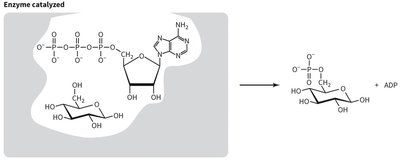
Enzyme Catalysis of Phosphorylation
Enzymes such as hexokinase catalyze the phosphorylation of glucose, greatly increasing the reaction rate and lowering the activation energy. The enzyme provides a specific environment for the reactants, facilitating the transfer of the phosphate group.
Enzyme-catalyzed reaction: ATP + Glucose → Glucose-6-phosphate + ADP
Active site: Enzyme binds both ATP and glucose, positioning them for efficient phosphate transfer.
Result: Rapid formation of glucose-6-phosphate, essential for cellular metabolism.
Reaction Coordinate and Activation Energy
The reaction coordinate diagram illustrates the effect of enzyme catalysis on the free energy profile of the phosphorylation reaction. Enzymes lower the activation energy, making the reaction proceed faster, but do not alter the overall free energy change (ΔG).
Transition State: The highest energy point along the reaction pathway.
Uncatalyzed Reaction: High activation energy, slow rate.
Enzyme-catalyzed Reaction: Lower activation energy, fast rate.
ΔG: The difference in free energy between reactants and products remains unchanged.