 Back
BackEnzyme Kinetics and Enzyme Inhibition: Study Notes
Study Guide - Smart Notes

Enzyme Kinetics and Enzyme Inhibition
Overview of Enzyme Kinetics and Inhibition
Enzyme kinetics is the study of the rates at which enzyme-catalyzed reactions proceed and how these rates are affected by various factors, including inhibitors. Enzyme inhibition is a central concept in biochemistry and pharmaceutical development, as many drugs function by inhibiting specific enzymes. Understanding the types and mechanisms of inhibition is crucial for designing effective therapeutics.
Enzyme Kinetics: Focuses on the chemical reactions catalyzed by enzymes and their reaction rates.
Enzyme Inhibition: Refers to the decrease in enzyme activity due to the presence of inhibitors, which can alter reaction rates.
Pharmaceutical Relevance: Many drugs are designed to inhibit enzymes, affecting processes such as pain, cholesterol synthesis, cancer cell division, and blood flow.
Examples: Aspirin (cyclooxygenase inhibitor), Statins (HMG-CoA reductase inhibitor), Taxol (microtubulin inhibitor), Sildenafil (phosphodiesterase inhibitor).
Factors Determining Reaction Rate
The rate of an enzymatic reaction depends on the energy barrier between substrate and product, known as the standard free energy of activation (). Enzymes lower this energy barrier, increasing the fraction of molecules that can reach the transition state and thus speeding up the reaction.
Forward and Reverse Reactions: Enzymatic reactions can proceed in both directions (substrate to product and vice versa).
Activation Energy (): High activation energy results in slow reactions; enzymes lower this barrier.
Michaelis-Menten Equation
Fundamentals of Michaelis-Menten Kinetics
The Michaelis-Menten equation describes the relationship between the rate of an enzyme-catalyzed reaction and substrate concentration. It is foundational for understanding enzyme kinetics.
Reaction Scheme:
Initial Velocity ():
Michaelis-Menten Equation:
Definitions:
: Maximum velocity of the reaction (units: concentration/time).
: Michaelis constant; substrate concentration at which is half of (units: concentration).
Affinity: Lower indicates higher enzyme affinity for substrate.
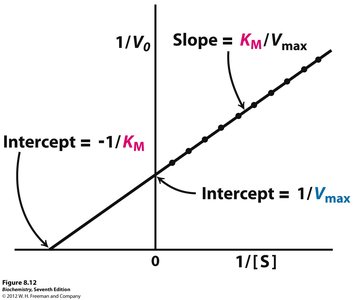
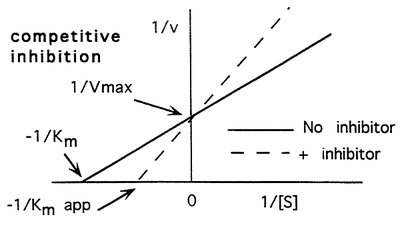
Lineweaver-Burk Plot
The Lineweaver-Burk plot is a double reciprocal transformation of the Michaelis-Menten equation, used to determine and more accurately.
Equation:
Axes: (y-axis) vs (x-axis)
Intercepts: (y-intercept), (x-intercept)
Slope:
Types of Enzyme Inhibition
Overview of Inhibition Types
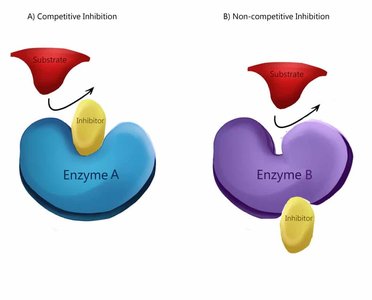
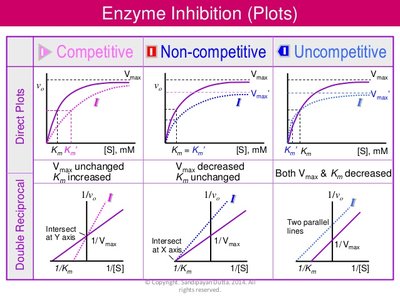
Enzyme inhibitors are classified based on their interaction with the enzyme and substrate. The three main types are competitive, uncompetitive, and noncompetitive inhibition, each affecting enzyme kinetics differently.
Competitive Inhibition: Inhibitor competes with substrate for the active site.
Uncompetitive Inhibition: Inhibitor binds only to the enzyme-substrate complex.
Noncompetitive Inhibition: Inhibitor binds to an allosteric site, not the active site.
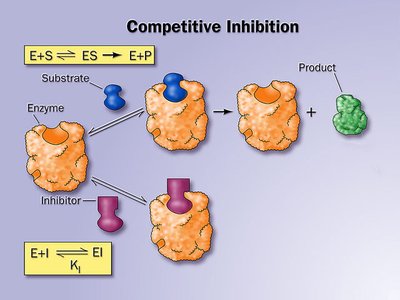
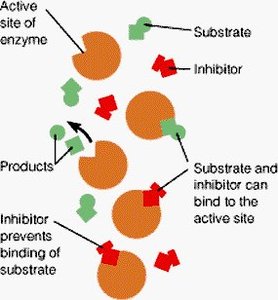
Competitive Inhibition
Competitive inhibitors resemble the substrate and bind to the enzyme's active site, preventing substrate binding. This inhibition can be overcome by increasing substrate concentration.
Effect on Kinetics: Increases (apparent affinity decreases), remains unchanged.
Mechanism: Substrate and inhibitor compete for the same site.
Example: Many drugs act as competitive inhibitors, such as statins.
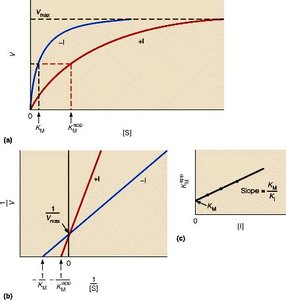
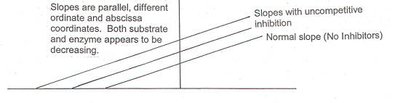
Uncompetitive Inhibition
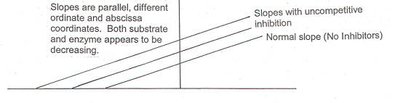
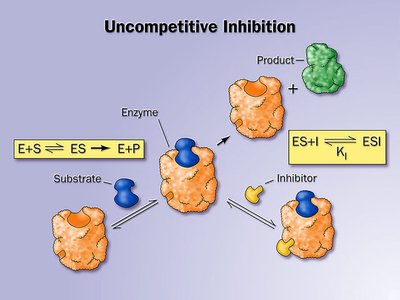
Uncompetitive inhibitors bind only to the enzyme-substrate complex, preventing product formation. This type of inhibition is rare and typically occurs in multi-substrate reactions.
Effect on Kinetics: Both and decrease.
Mechanism: Inhibitor binds after substrate is already bound.
Example: Some drugs targeting multi-substrate enzymes act via uncompetitive inhibition.



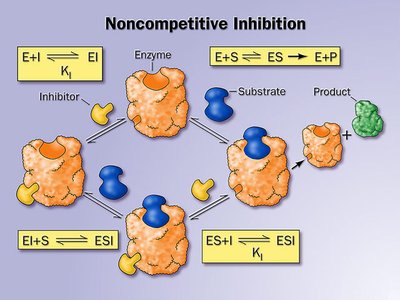
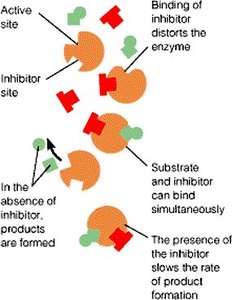
Noncompetitive Inhibition
Noncompetitive inhibitors bind to an allosteric site, causing a conformational change in the enzyme. The substrate can still bind, but enzyme activity is reduced. Increasing substrate concentration does not reverse the inhibition.
Effect on Kinetics: decreases, remains unchanged.
Mechanism: Inhibitor binds to a site other than the active site.
Example: Some kinase inhibitors act as noncompetitive inhibitors.

Comparative Summary Table: Types of Enzyme Inhibition
Type | Binding Site | Effect on | Effect on | Reversibility by Substrate |
|---|---|---|---|---|
Competitive | Active site | Increases | Unchanged | Yes |
Uncompetitive | ES complex | Decreases | Decreases | No |
Noncompetitive | Allosteric site | Unchanged | Decreases | No |
Designing Enzyme Inhibitors as Drugs
Drug Development and Enzyme Inhibition
Drug design often focuses on developing inhibitors with high binding affinity for target enzymes. Competitive inhibitors are typically more specific, while uncompetitive inhibitors provide stronger inhibition. The binding affinity, determined by , is crucial for drug efficacy and safety.
High Affinity: Lower for inhibitor means better binding and lower required dosage.
Specificity: Competitive inhibitors are often more specific to their target enzyme.
Determining Inhibition Type: Plotting vs with and without inhibitor reveals the inhibition type.
Summary of Key Concepts
Enzyme kinetics and inhibition are fundamental to biochemistry and drug development.
Michaelis-Menten and Lineweaver-Burk plots are essential tools for analyzing enzyme activity.
Competitive, uncompetitive, and noncompetitive inhibition each affect enzyme kinetics differently.
Understanding these mechanisms is critical for designing effective enzyme-targeted drugs.
