 Back
BackEnzyme Structure, Function, and Kinetics: A Comprehensive Study Guide
Study Guide - Smart Notes

Enzymes: Biological Catalysts
Introduction to Enzymes
Enzymes are highly specific biological catalysts that accelerate chemical reactions in living organisms. They exhibit remarkable specificity for their substrates and are regulated to meet cellular needs. Enzyme activity is sensitive to environmental factors such as temperature and pH, and their three-dimensional structure is essential for catalytic function.
Stereospecificity: Enzymes catalyze reactions with high stereoselectivity, often acting on only one stereoisomer of a substrate.
Specificity: Refers to the enzyme's selectivity for its substrate, ensuring precise biochemical transformations.
Catalytic Power: Defined as the ratio of the rate of the enzyme-catalyzed reaction to the uncatalyzed rate.
Regulation: Enzyme activity is tightly regulated to maintain metabolic balance.
Sensitivity: Enzymes function optimally within specific temperature and pH ranges.
Enzyme Classification and Nomenclature
Enzymes are systematically classified based on the reactions they catalyze. Each enzyme is assigned an Enzyme Commission (E.C.) number, which provides detailed information about its function. Major enzyme databases include BRENDA, ExPASy, and the IUBMB Enzyme Explorer.
Six Major Classes: Oxidoreductases, Transferases, Hydrolases, Lyases, Isomerases, Ligases.
E.C. Number: A numerical classification scheme for enzymes, based on the chemical reactions they catalyze.
Examples of Major Enzyme Classes
The following table summarizes the six major enzyme classes, with examples and the reactions they catalyze:
Class | Example (Reaction Type) | Reaction Catalyzed |
|---|---|---|
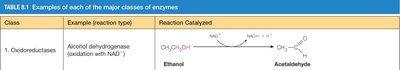
1. Oxidoreductases | Alcohol dehydrogenase (oxidation with NAD+) | Ethanol → Acetaldehyde |
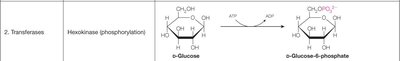
2. Transferases | Hexokinase (phosphorylation) | β-D-Glucose → β-D-Glucose-6-phosphate |
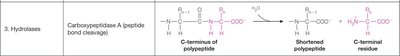
3. Hydrolases | Carboxypeptidase A (peptide bond cleavage) | Polypeptide (C-terminus) → Shortened polypeptide + C-terminal residue |
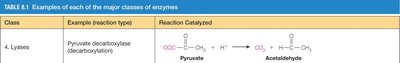
4. Lyases | Pyruvate decarboxylase (decarboxylation) | Pyruvate → Acetaldehyde + CO2 |
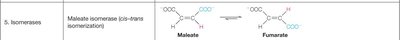
5. Isomerases | Malate isomerase (cis-trans isomerization) | Malate → Fumarate |
6. Ligases | Pyruvate carboxylase (carboxylation) | Pyruvate + CO2 → Oxaloacetate |

Enzyme Kinetics
Reaction Rates and Order
Enzyme kinetics studies the rates of enzyme-catalyzed reactions. The rate of a reaction depends on substrate concentration, enzyme concentration, and environmental conditions. The rate constant (k) determines how quickly a reaction proceeds.
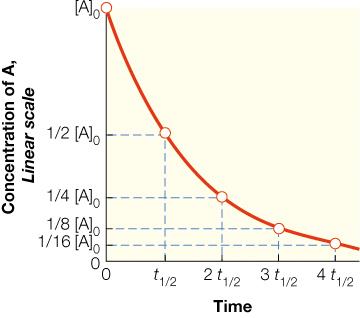
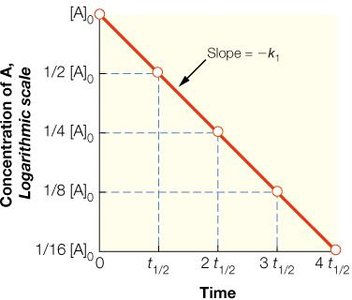
First-Order Reactions: The rate depends linearly on the concentration of one reactant.
Rate Law:
Integrated Rate Law:
Half-Life (t1/2): The time required for the concentration of a reactant to decrease by half.
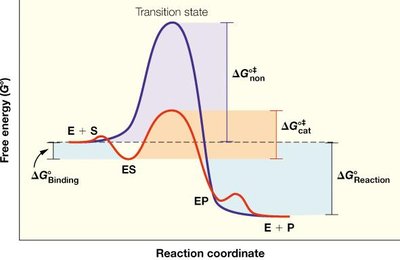
Transition State Theory and Activation Energy
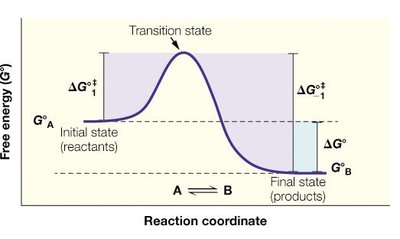
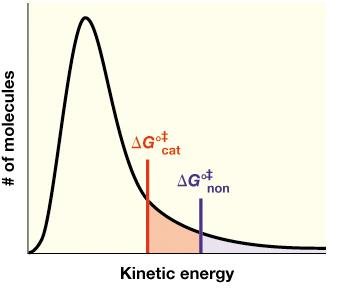
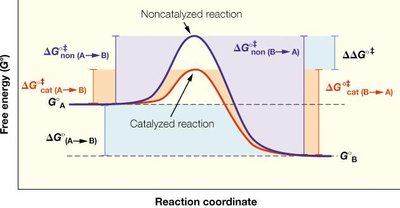
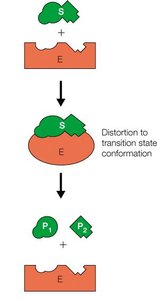
Enzymes accelerate reactions by lowering the activation energy (ΔG‡) required to reach the transition state. The transition state is a high-energy, unstable intermediate between reactants and products.
Activation Energy (ΔG‡): The energy barrier that must be overcome for a reaction to proceed.
Transition State: The highest energy point along the reaction coordinate.
Rate Constant Expression:
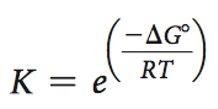
Enzyme Mechanisms
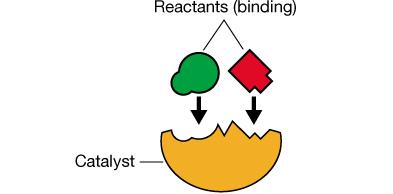
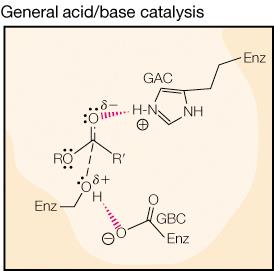
Enzymes employ various mechanisms to stabilize the transition state and enhance reaction rates, including proximity/orientation effects, acid-base catalysis, and electrostatic stabilization.
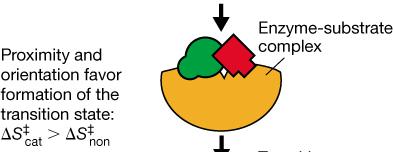
Proximity and Orientation: Enzymes bring substrates together in the correct orientation to favor transition state formation.
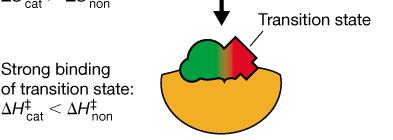
Transition State Stabilization: Enzymes bind the transition state more tightly than the substrate or product, lowering ΔG‡.
General Acid/Base Catalysis: Enzyme side chains donate or accept protons to facilitate the reaction.
Electrostatic Stabilization: Charged groups or metal ions stabilize charged intermediates.
Enzyme Kinetics: Michaelis-Menten Model
Basic Model and Equations
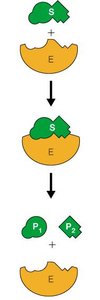
The Michaelis-Menten model describes the kinetics of many enzyme-catalyzed reactions. It assumes the formation of an enzyme-substrate (ES) complex as an intermediate.
Reaction Scheme:
Initial Velocity:
Total Enzyme:
Michaelis-Menten Equation:
Lineweaver-Burk Plot:
Enzyme Inhibition
Enzyme inhibitors decrease or abolish enzyme activity. Inhibition can be competitive, noncompetitive, or uncompetitive, each affecting kinetic parameters differently.
Competitive Inhibition: Inhibitor binds to the active site, increasing apparent KM but not affecting Vmax.
Noncompetitive Inhibition: Inhibitor binds to an allosteric site, decreasing Vmax without changing KM.
Uncompetitive Inhibition: Inhibitor binds only to the ES complex, decreasing both Vmax and KM.
Example: Captopril is a competitive inhibitor of angiotensin-converting enzyme (ACE), used to treat hypertension.
Coenzymes and Vitamins
Role of Coenzymes
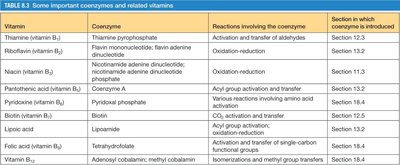
Many enzymes require non-protein cofactors called coenzymes, often derived from vitamins, to carry out catalysis. Coenzymes participate in group transfer reactions, oxidation-reduction, and other processes.
Vitamin | Coenzyme | Reactions Involving the Coenzyme |
|---|---|---|
Thiamine (B1) | Thiamine pyrophosphate | Activation and transfer of aldehydes |
Riboflavin (B2) | FMN, FAD | Oxidation-reduction |
Niacin (B3) | NAD+, NADP+ | Oxidation-reduction |
Pantothenic acid (B5) | Coenzyme A | Acyl group activation and transfer |
Pyridoxine (B6) | Pyridoxal phosphate | Various reactions involving amino acids |
Biotin | Biotin | CO2 activation and transfer |
Folic acid | Tetrahydrofolate | Activation and transfer of single-carbon functional groups |
Vitamin B12 | Adenosyl cobalamin, methyl cobalamin | Isomerizations and methyl group transfers |
Summary
Enzymes are essential biological catalysts with remarkable specificity and catalytic power. Their activity is regulated and dependent on their structure and environmental conditions. Understanding enzyme classification, kinetics, mechanisms, inhibition, and the role of coenzymes is fundamental to biochemistry.
