 Back
BackEnzymes: Catalysis and Kinetics – Structure, Function, and Regulation
Study Guide - Smart Notes

Enzymes: Catalysis and Kinetics
Introduction to Enzymes
Enzymes are specialized proteins that act as biological catalysts, accelerating chemical reactions in living organisms. Nearly all biochemical reactions in cells are catalyzed by enzymes, making them essential for life. Enzymes are highly specific, both in the reactions they catalyze and the substrates they bind.
Definition: Enzymes are protein molecules that increase the rate of biochemical reactions without being consumed in the process.
Biological Importance: Thousands of reactions occur simultaneously in cells, and enzymes ensure these proceed efficiently and selectively.
Example: DNA polymerase can replicate the entire E. coli genome in about 60 minutes.
Cellular Environment and Enzyme Activity
Cells are densely packed with macromolecules, creating a crowded environment that influences enzyme activity and substrate availability. This crowding affects reaction rates and the efficiency of metabolic pathways.
Crowding: The cytoplasm contains high concentrations of proteins, nucleic acids, and other macromolecules.
Impact: Molecular crowding can enhance or inhibit enzyme activity by affecting diffusion and molecular interactions.
Necessity of Catalysis
Many biochemical reactions are thermodynamically favorable but proceed extremely slowly without catalysis. Enzymes lower the activation energy required, allowing reactions to occur at biologically relevant rates.
Example: Sucrose and glucose are stable compounds that do not spontaneously react under physiological conditions.
Equation: The oxidation of glucose is highly exergonic:


Thermodynamics vs. Kinetics
Although some reactions are thermodynamically possible, they may not occur spontaneously due to high activation energy barriers. Enzymes provide an alternative reaction pathway with a lower activation energy, enabling the reaction to proceed rapidly.
Thermodynamic Potentiality: The difference in free energy () between reactants and products determines if a reaction is possible, but not how fast it occurs.
Activation Energy (): The energy required to reach the transition state; enzymes lower this barrier.

Enzyme Mechanisms and Specificity
Enzymes use a variety of physical and chemical mechanisms to accelerate reactions, including substrate orientation, strain induction, and stabilization of transition states. They are highly specific, often catalyzing only one type of reaction or acting on a specific substrate.
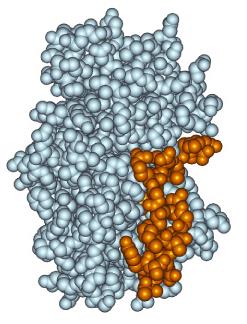
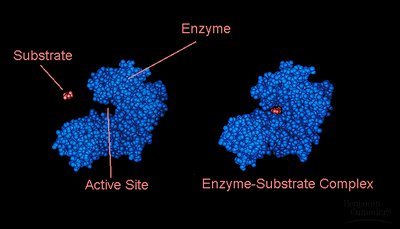
Active Site: The region of the enzyme where substrate binding and catalysis occur. The active site provides a unique microenvironment that facilitates the reaction.
Specificity: Achieved through molecular complementarity between the enzyme and its substrate.
Classification of Enzymes
Enzymes are classified by the International Commission on Enzymes into six main classes based on the type of reaction they catalyze. Each enzyme is assigned an Enzyme Commission (E.C.) number that reflects its class, subclass, and sub-subclass.
Six Main Classes:
Oxidoreductases
Transferases
Hydrolases
Lyases
Isomerases
Ligases
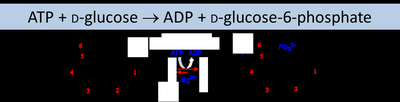
Example: Hexokinase (E.C. 2.7.1.2) catalyzes the transfer of a phosphate group from ATP to glucose.
E.C. Number | Systematic Name and Subclasses |
|---|---|
1 | Oxidoreductases (oxidation-reduction reactions) |
2 | Transferases (transfer of functional groups) |
3 | Hydrolases (hydrolysis reactions) |
4 | Lyases (addition to double bonds) |
5 | Isomerases (isomerization reactions) |
6 | Ligases (formation of bonds with ATP cleavage) |

Cofactors and Coenzymes
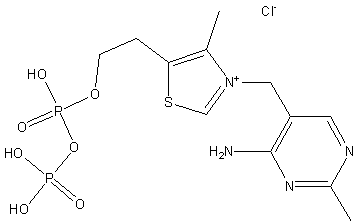
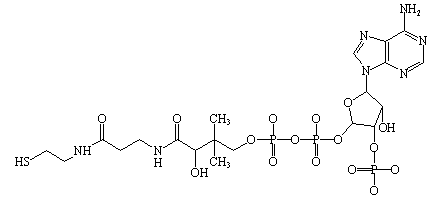
Some enzymes require additional non-protein molecules called cofactors to function. These can be metal ions or organic molecules (coenzymes) such as NAD or FAD.
Cofactors: Inorganic ions (e.g., Mg2+, Zn2+) that assist in catalysis.
Coenzymes: Organic molecules that participate in the reaction, often derived from vitamins.
Enzyme Kinetics
Enzyme kinetics is the study of the rates of enzyme-catalyzed reactions. Key parameters include the maximum reaction velocity (Vmax) and the Michaelis constant (KM), which reflects the substrate concentration at which the reaction rate is half-maximal.
Vmax: The maximum rate achieved by the system, at saturating substrate concentration.
KM: The substrate concentration at which the reaction rate is half of Vmax.
Flux: The total amount of substrate converted to product over time in a metabolic pathway.
Enzyme Regulation and Control
Cells regulate enzyme activity to control metabolic pathways and respond to changing conditions. Regulation can occur through changes in enzyme concentration, covalent modification, allosteric regulation, or compartmentalization.
Regulatory Enzymes: Specific enzymes control the rate of entire pathways (rate-determining steps).
Mechanisms: Include feedback inhibition, phosphorylation, and changes in gene expression.
Enzyme Catalysis: Mechanistic Overview
Enzyme-catalyzed reactions proceed through the formation of an enzyme-substrate complex, transition state stabilization, and product release. The overall reaction can be summarized as:
Transition State: The highest energy state during the reaction; enzymes lower the free energy of activation ().
Effect on Equilibrium: Enzymes accelerate the rate at which equilibrium is reached but do not alter the equilibrium position.
Summary of Enzyme Properties
Enzymes only catalyze thermodynamically favorable reactions.
They lower the activation energy () but do not affect the overall free energy change () of the reaction.
Enzymes are not consumed during the reaction and can be reused.
They provide cells with precise control over metabolic pathways and flux.
