 Back
BackEssential Concepts in Biological Membranes and Noncovalent Interactions
Study Guide - Smart Notes

Cell Membranes and Their Structure
Overview of the Cell Membrane
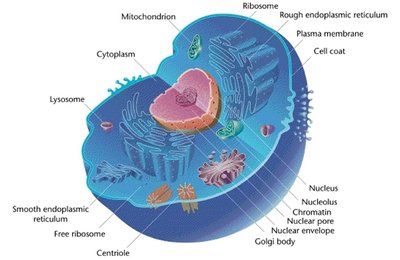
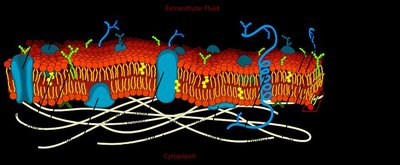
The cell membrane is a critical boundary that separates the interior of the cell from its external environment. It maintains the physical integrity of the cell, prevents leakage of cellular contents, and regulates the entry and exit of nutrients, ions, and other molecules. The membrane is primarily composed of a phospholipid bilayer interspersed with proteins, which together create a selectively permeable barrier.
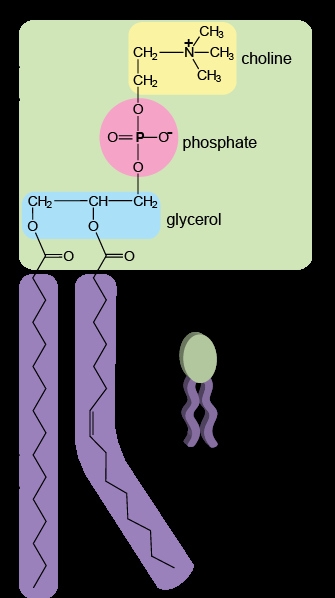
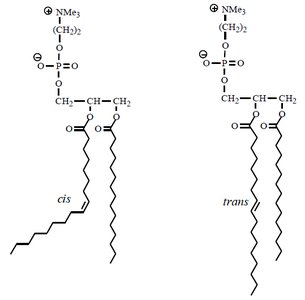
Phospholipid Bilayer: Consists of hydrophilic (polar) head groups facing the aqueous environments inside and outside the cell, and hydrophobic (nonpolar) fatty acid tails oriented inward, away from water.
Proteins: Embedded within or associated with the bilayer, these serve as channels, receptors, and enzymes.
Flexibility: The membrane's structure allows for flexibility, enabling cells to move or squeeze through tight spaces.
Noncovalent Interactions in Biomolecules
Types of Chemical Bonds
Biomolecular structure and function are governed by both covalent and noncovalent interactions. Covalent bonds involve the sharing of electron pairs between atoms, forming the backbone of molecules. Noncovalent interactions, though weaker, are essential for the dynamic assembly and function of biological macromolecules.
Covalent Bonds: Strong, intramolecular forces (e.g., C–C, C–H bonds).
Noncovalent Interactions: Weaker, intermolecular forces (e.g., hydrogen bonds, ionic interactions, van der Waals forces).
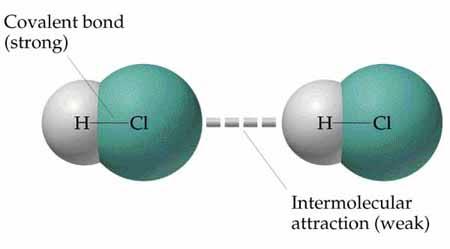
Relative Energies of Bonds
The energies of covalent and noncovalent bonds differ significantly. Covalent bonds are much stronger (150–400 kJ/mol), while noncovalent interactions range from less than 1 kJ/mol (van der Waals) to about 20 kJ/mol (hydrogen bonds).

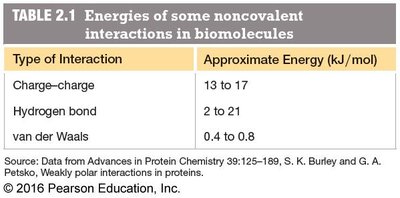
Type of Interaction | Approximate Energy (kJ/mol) |
|---|---|
Charge–charge | 13 to 17 |
Hydrogen bond | 2 to 21 |
van der Waals | 0.4 to 0.8 |
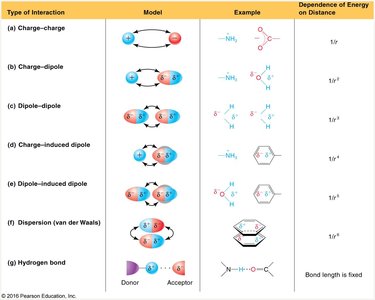
Types of Noncovalent Interactions
Noncovalent interactions are essential for the structure and function of proteins, nucleic acids, and membranes. They include:
Electrostatic (Ionic) Interactions
Dipole-Dipole Interactions
Ion-Dipole Interactions
Induced Dipole Interactions
Hydrogen Bonds
van der Waals Interactions
Electrostatic (Ionic) Interactions
Electrostatic interactions occur between charged particles (ions) and are described by Coulomb’s Law:
In biological systems, the medium's dielectric constant (ε) reduces the force:
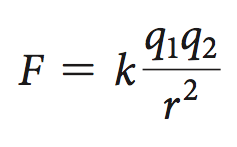
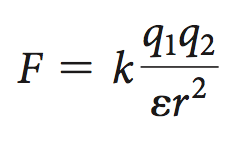
Electrostatic interactions are strongest in nonpolar environments and decrease with increasing dielectric constant (e.g., water).
These interactions are crucial for the initial binding of drugs to their targets.
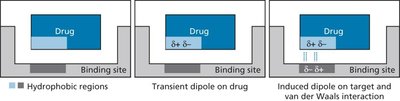
Dipole-Dipole and Related Interactions
Dipole-dipole interactions occur between molecules with permanent dipole moments. The strength of these interactions decreases rapidly with distance.
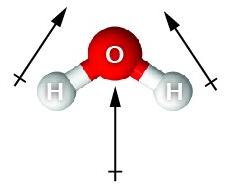
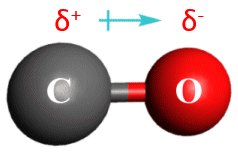
Permanent Dipoles: Molecules like HCl, CO, and H2O have permanent dipole moments due to differences in electronegativity.
Induced Dipoles: Nonpolar molecules can develop temporary dipoles when in proximity to polar molecules or ions.
Ion-Dipole Interactions: Occur when an ion interacts with a polar molecule, stronger than dipole-dipole interactions.

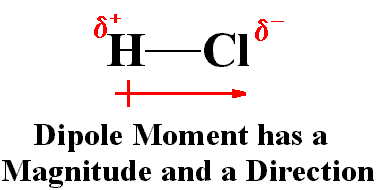
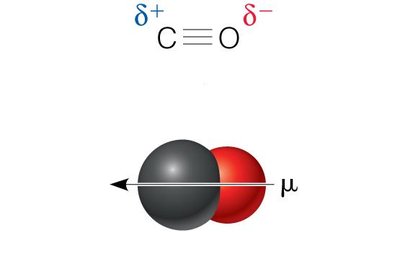
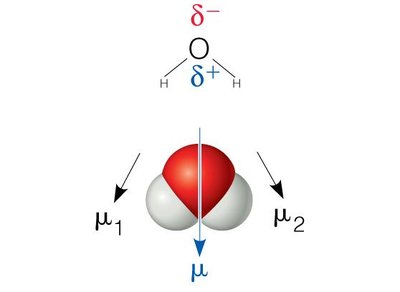
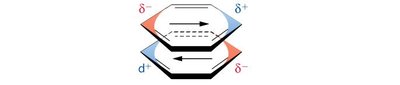
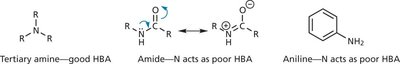
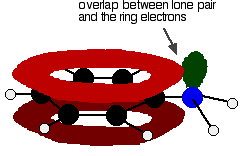
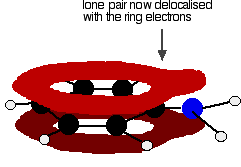
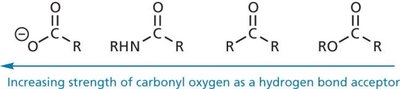
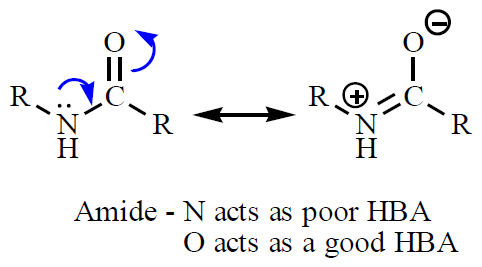
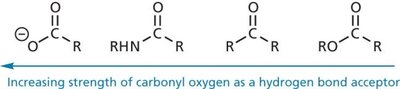
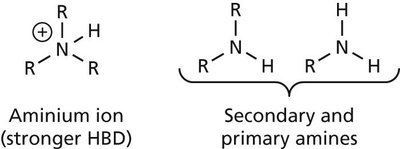
Hydrogen Bonds
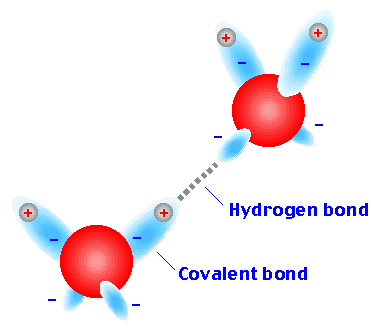
Hydrogen bonds are a special type of dipole-dipole interaction involving a hydrogen atom covalently bonded to an electronegative atom (usually O or N), which interacts with another electronegative atom. Hydrogen bonds are crucial for the structure of proteins and nucleic acids.
Donor: The group supplying the hydrogen (e.g., –OH, –NH).
Acceptor: The electronegative atom with a lone pair (e.g., O, N).
Optimal hydrogen bonds are linear (180° angle).
van der Waals Interactions
van der Waals interactions are weak, transient attractions between all atoms and molecules, arising from temporary dipoles due to fluctuations in electron distribution. Although individually weak, their cumulative effect can be significant in stabilizing macromolecular structures.
Important for the close packing of molecules in membranes and protein folding.
Strength decreases rapidly with distance.
Desolvation and Hydrophobic Interactions
Desolvation Penalties
Before a drug or ligand binds to its target, polar regions are typically solvated by water molecules. The removal of these water molecules (desolvation) requires energy, which must be compensated by the stabilization energy gained from binding.
Desolvation is necessary for effective binding in aqueous environments.
The net free energy change () for binding is given by:
Increased entropy () from releasing ordered water molecules around hydrophobic regions favors binding.
Hydrophobic Interactions
Hydrophobic interactions occur when nonpolar regions of molecules associate to minimize their exposure to water, thereby increasing the entropy of the system. These interactions are crucial for membrane formation, protein folding, and molecular recognition.
Hydrophobic regions are not solvated, and water forms an ordered layer around them.
Binding of hydrophobic regions releases ordered water, increasing entropy and favoring binding.
