 Back
BackFatty Acids, Eicosanoids, and Lipid Metabolism
Study Guide - Smart Notes

Lipids: Structure, Classification, and Functions
Definition and General Properties
Lipids are a diverse group of organic compounds characterized by their solubility in non-polar solvents and poor solubility in water. They can be classified as either polar or non-polar, with each class serving distinct biological functions.
Polar lipids: Fatty acids, cholesterol, glycerophosphatides, glycosphingolipids
Non-polar lipids: Triacylglycerols, cholesteryl esters
Functions of Lipids:
Thermal insulation
Energy storage (as triacylglycerols, TG)
Metabolic fuels
Membrane components (phospholipids & cholesterol)
Hormones (steroids & vitamin D)
Precursors of prostanoids & leukotrienes
Vitamins A, D, E, & K
Emulsifying agents in digestion & absorption (bile acids)
Surfactants in alveolar membranes (phosphatidylcholine)
Fatty Acids: Types and Properties
Saturated Fatty Acids
Saturated fatty acids (SFAs) contain no double bonds between carbon atoms. They are commonly found in animal fats and have higher melting points compared to unsaturated fatty acids.
Common Name | Systemic Name | Molecular Formula | Structural Formula | Melting Point (°C) |
|---|---|---|---|---|
Lauric | n-Dodecanoic | C12H24O2 | CH3[CH2]10COOH | 44 |
Myristic | n-Tetradecanoic | C14H28O2 | CH3[CH2]12COOH | 58 |
Palmitic* | n-Hexadecanoic | C16H32O2 | CH3[CH2]14COOH | 63 |
Stearic* | n-Octadecanoic | C18H36O2 | CH3[CH2]16COOH | 70 |
Arachidic | n-Eicosanoic | C20H40O2 | CH3[CH2]18COOH | 76 |
*Palmitic and stearic acids are the most abundant fatty acids in animal lipids.
Unsaturated Fatty Acids (USFAs)
Unsaturated fatty acids contain one or more double bonds, usually in the cis configuration. They are important for membrane fluidity and serve as precursors for bioactive molecules.
Monounsaturated FAs: Palmitoleic acid (16:1Δ9), Oleic acid (18:1Δ9)
Polyunsaturated FAs: Linoleic acid, α-Linolenic acid, Arachidonic acid
Double bonds are introduced by fatty acid oxygenases in the endoplasmic reticulum, requiring O2 and NADPH.
Functions of Unsaturated Fatty Acids
Maintain fluidity of triglyceride reserves and phospholipid membranes
Serve as precursors for eicosanoids (prostaglandins, prostacyclins, thromboxanes, leukotrienes)
Fatty Acid Oxidation
Overview and Cellular Compartmentalization
Fatty acid oxidation is an aerobic process that occurs primarily in the mitochondria. It is not a simple reversal of fatty acid biosynthesis and involves distinct enzymes and cellular compartments.
Increased during starvation and diabetes mellitus, leading to ketone body production in the liver
Requires molecular oxygen (O2)
Ketone Body Metabolism
Ketogenesis and Ketone Body Utilization
Ketone bodies are produced in the liver from acetyl-CoA during periods of increased fatty acid oxidation (e.g., fasting, diabetes). They serve as alternative energy sources for peripheral tissues, especially the brain during prolonged fasting.
Major ketone bodies: Acetoacetate, β-hydroxybutyrate, acetone
Formed via condensation of acetyl-CoA, catalyzed by HMG-CoA synthase and lyase
Oxidized in extrahepatic tissues to generate acetyl-CoA for the TCA cycle
Key reactions:
Acetyl-CoA → Acetoacetyl-CoA → HMG-CoA → Acetoacetate → β-hydroxybutyrate/acetone
Fatty Acid Synthesis
Pathway and Regulation
Fatty acid synthesis occurs mainly in the liver cytosol and involves the polymerization of two-carbon units from acetyl-CoA to form palmitic acid (16:0). The process requires NADPH and ATP and is catalyzed by a multi-enzyme complex called fatty acid synthase.
Key regulatory enzymes: Acetyl-CoA carboxylase (rate-limiting), fatty acid synthase
Acetyl-CoA is carboxylated to malonyl-CoA, which donates two-carbon units for chain elongation
Elongation beyond 16 carbons occurs in the endoplasmic reticulum or mitochondria
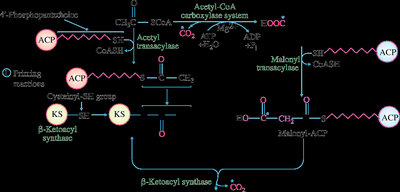
Fatty Acid Elongation and Synthesis of Long-Chain FAs
Palmitate (16:0) is the primary product of cytoplasmic fatty acid synthesis. Longer-chain fatty acids (18, 20, 22, 24 carbons) are synthesized by elongation of palmitate in the endoplasmic reticulum or mitochondria, mainly in the liver.
Metabolism of Eicosanoids
Precursors and Biosynthesis
Eicosanoids are signaling molecules derived from 20-carbon polyunsaturated fatty acids, primarily arachidonic acid. They include prostaglandins, thromboxanes, prostacyclins, and leukotrienes.
Arachidonic acid is released from membrane phospholipids by phospholipase A2
Phospholipase A2 is activated by increased intracellular Ca2+ and calmodulin
Glucocorticoids (e.g., cortisol) inhibit phospholipase A2 activity, reducing eicosanoid synthesis
Therapeutic Inhibition
Inhibitors of key enzymes in eicosanoid synthesis (e.g., NSAIDs inhibiting cyclooxygenase) are used as therapeutic agents to control inflammation and pain.
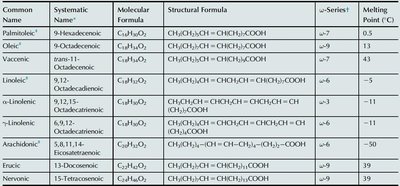
Summary Table: Common Unsaturated Fatty Acids
Common Name | Systematic Name | Molecular Formula | Structural Formula | ω-Series | Melting Point (°C) |
|---|---|---|---|---|---|
Palmitoleic | 9-Hexadecenoic | C16H30O2 | CH3(CH2)5CH=CH(CH2)7COOH | ω-7 | 0.5 |
Oleic | 9-Octadecenoic | C18H34O2 | CH3(CH2)7CH=CH(CH2)7COOH | ω-9 | 13 |
Vaccenic | trans-11-Octadecenoic | C18H34O2 | CH3(CH2)5CH=CH(CH2)9COOH | ω-7 | 44 |
Linoleic | 9,12-Octadecadienoic | C18H32O2 | CH3(CH2)4CH=CHCH2CH=CH(CH2)7COOH | ω-6 | -5 |
α-Linolenic | 9,12,15-Octadecatrienoic | C18H30O2 | CH3(CH2)3CH=CHCH2CH=CHCH2CH=CH(CH2)7COOH | ω-3 | -11 |
γ-Linolenic | 6,9,12-Octadecatrienoic | C18H30O2 | CH3(CH2)4CH=CHCH2CH=CHCH2CH=CHCOOH | ω-6 | -11 |
Arachidonic | 5,8,11,14-Eicosatetraenoic | C20H32O2 | CH3(CH2)4CH=CHCH2CH=CHCH2CH=CHCH2CH=CHCOOH | ω-6 | -50 |
Erucic | 13-Docosenoic | C22H42O2 | CH3(CH2)7CH=CH(CH2)11COOH | ω-9 | 33 |
Nervonic | 15-Tetracosenoic | C24H46O2 | CH3(CH2)7CH=CH(CH2)13COOH | ω-9 | 39 |
