 Back
BackIntroduction to Enzyme Kinetics: Principles and Mathematical Foundations
Study Guide - Smart Notes

Introduction to Enzyme Kinetics
Definition and Scope
Enzyme kinetics is the study of the rates at which enzyme-catalyzed reactions proceed and the factors that affect these rates. It provides quantitative insights into how enzymes function and how their activity can be modulated by various factors.
Kinetics in biochemistry refers to measuring and analyzing the rates of chemical or biochemical reactions.
The rate of enzyme activity is defined as the change in concentration of substrate or product per unit time.
Factors Affecting Enzyme Activity
Enzyme kinetics investigates how different variables influence the rate of enzyme-catalyzed reactions:
Enzyme concentration
Ligand concentration (including substrates, inhibitors, and activators)
pH
Ionic strength
Temperature
Importance of Kinetic Studies
Kinetic analysis is crucial for understanding enzyme function and regulation:
Provides information on enzyme mechanisms.
Reveals the role of enzymes under physiological conditions and their response to metabolite concentrations.
Offers clues about enzyme regulation in vivo.
Helps identify amino acid residues in the active site.
Useful for drug screening and comparison of enzyme efficiencies.
Basic Concepts in Reaction Kinetics
Rate of Reaction
The rate of a chemical reaction is determined by:
The concentration of reactant(s) (substrate in enzymology)
The rate constant, k
For a unimolecular reaction (first-order reaction):
where is the initial reaction rate, is the first-order rate constant (units: s-1), and is the substrate concentration.
Units of Enzyme Activity
Enzyme activity is quantified as follows:
One unit of enzyme activity is the amount of enzyme that catalyzes the formation of one micromole of product per minute under standard conditions.
Expressed as:
Measuring Enzyme Activity
Enzyme activity () can be determined by:
The rate of substrate disappearance:
The rate of product appearance:
Relationship Between Enzyme Activity and Substrate Concentration
Hyperbolic Relationship
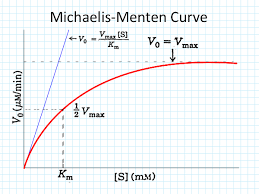
As substrate concentration increases, the rate of reaction initially rises sharply and then levels off, forming a hyperbolic curve. This reflects the saturation of enzyme active sites at high substrate concentrations.
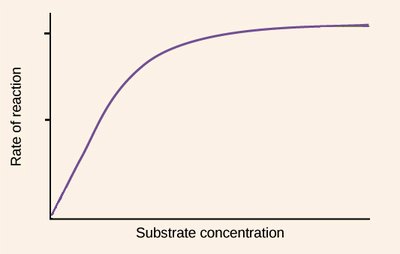
Explanation of the Hyperbolic Curve
The hyperbolic shape arises because, at low substrate concentrations, the rate is proportional to [S] (first-order kinetics). As [S] increases, the enzyme becomes saturated, and the rate approaches a maximum value (zero-order kinetics with respect to [S]).
Mathematical Representation of Enzyme Kinetics
Linear and Parabolic Equations
Different mathematical models describe the relationship between variables:
Linear:
Parabolic:
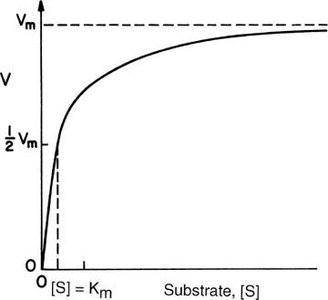
Michaelis-Menten Equation
The Michaelis-Menten equation quantitatively describes the hyperbolic relationship between reaction rate and substrate concentration:
: Initial reaction velocity
: Maximum reaction velocity
: Michaelis constant (substrate concentration at which )
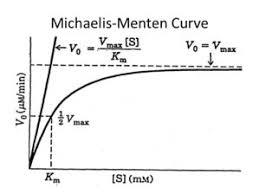
Special Cases of the Michaelis-Menten Equation
When [S] << Km: (linear relationship)
When [S] >> Km: (rate independent of [S])
When [S] = Km:
Mathematical Manipulation
Any term in the Michaelis-Menten equation can be isolated to solve for unknowns, such as , , or , depending on the experimental data available.
Summary Table: Key Parameters in Enzyme Kinetics
Parameter | Definition | Units |
|---|---|---|
V0 | Initial reaction velocity | mol/L/s or µmol/min |
Vmax | Maximum reaction velocity | mol/L/s or µmol/min |
Km | Michaelis constant | mol/L or mM |
k | First-order rate constant | s-1 |
Additional info: The Michaelis-Menten model assumes a simple enzyme-substrate interaction and does not account for allosteric effects or multi-substrate reactions, which require more complex models.
