 Back
BackIntroduction to Enzyme Kinetics: Principles and Mathematical Foundations
Study Guide - Smart Notes

Introduction to Enzyme Kinetics
Definition and Scope
Enzyme kinetics is the branch of biochemistry that studies the rates of enzyme-catalyzed reactions and the factors affecting these rates. It provides quantitative insights into how enzymes function, how their activity is regulated, and how they interact with substrates and other molecules.
Kinetics refers to the measurement and analysis of reaction rates in chemical and biochemical systems.
The rate of enzyme activity is defined as the change in substrate or product concentration per unit time.
Factors Affecting Enzyme Activity
The rate of enzyme-catalyzed reactions depends on several key factors:
Enzyme concentration
Ligand concentration (including substrates, inhibitors, and activators)
pH
Ionic strength
Temperature
Importance of Kinetic Studies
Studying enzyme kinetics is crucial for understanding:
Enzyme mechanisms and catalytic strategies
The physiological role of enzymes and their regulation in cells
How enzyme activity responds to changes in metabolite concentrations
Identifying amino acid residues in the active site
Screening and developing drugs as enzyme inhibitors
Comparing the efficiency and specificity of different enzymes
Basic Concepts in Reaction Kinetics
Rate of Reaction
The rate of a chemical reaction is determined by:
The concentration of reactant(s) (substrates in enzymology)
The rate constant, k
For a unimolecular (first-order) reaction, the rate equation is:
where is the initial reaction rate, is the first-order rate constant (units: s-1), and is the substrate concentration.
Units of Enzyme Activity
One enzyme unit (U) is the amount of enzyme that catalyzes the conversion of 1 micromole (µmol) of substrate per minute under standard conditions.
Alternatively, the katal is the SI unit, defined as the amount of enzyme that converts 1 mole of substrate per second.
Mathematically:
Measuring Enzyme Activity
By the rate of substrate disappearance:
By the rate of product appearance:
Relationship Between Enzyme Activity and Substrate Concentration
Hyperbolic Relationship
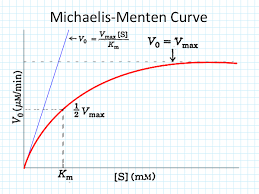
As substrate concentration increases, the rate of an enzyme-catalyzed reaction initially rises rapidly, then levels off to a maximum velocity (Vmax). This produces a hyperbolic curve, characteristic of Michaelis-Menten kinetics.
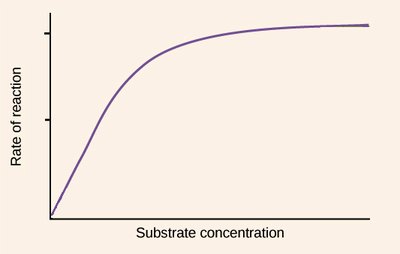
Explanation of the Hyperbolic Curve
The hyperbolic shape arises because, at low substrate concentrations, the rate is proportional to [S] (first-order kinetics). As [S] increases, the enzyme becomes saturated, and the rate approaches Vmax (zero-order kinetics with respect to [S]).
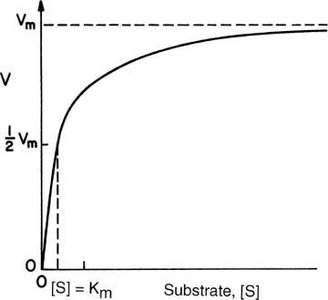
Mathematical Representation of the Hyperbolic Curve
Linear relationship:
Parabolic relationship:
Hyperbolic relationship (Michaelis-Menten):
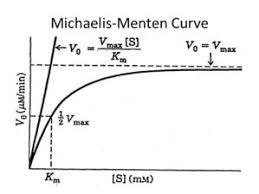
Michaelis-Menten Equation
The Michaelis-Menten equation quantitatively describes the hyperbolic relationship between reaction rate and substrate concentration:
V0: Initial velocity
Vmax: Maximum velocity (when enzyme is saturated)
Km: Michaelis constant (substrate concentration at which V0 = ½ Vmax)
Special Cases of the Michaelis-Menten Equation
When [S] << Km: (linear relationship)
When [S] >> Km: (rate independent of [S])
When [S] = Km:
Mathematical Manipulation of the Michaelis-Menten Equation
Any variable in the Michaelis-Menten equation can be solved for, given the others. This is useful for determining kinetic parameters from experimental data.
For example, to solve for Km:
Summary Table: Key Kinetic Parameters
Parameter | Definition | Units |
|---|---|---|
V0 | Initial velocity | mol L-1 s-1 |
Vmax | Maximum velocity | mol L-1 s-1 |
Km | Michaelis constant | mol L-1 |
k | First-order rate constant | s-1 |
Applications of Enzyme Kinetics
Understanding enzyme mechanisms and regulation
Drug discovery and inhibitor screening
Comparative analysis of enzyme efficiency
Biotechnological and clinical diagnostics
Additional info: The Michaelis-Menten model assumes a simple one-substrate reaction and steady-state conditions. More complex models exist for multi-substrate or allosteric enzymes.
