Back
BackLipids and Biological Membranes: Structure, Properties, and Functions
Study Guide - Smart Notes

Lipids and Membranes
Major Functions and Properties of Lipids
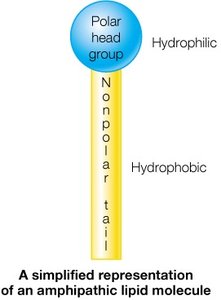
Lipids are a diverse group of biomolecules essential for energy storage, membrane structure, and cellular signaling. Unlike carbohydrates, amino acids, or nucleotides, lipids are characterized by their limited solubility in aqueous media. Most lipids are amphipathic, containing both hydrophobic (nonpolar) and hydrophilic (polar) regions, which is critical for their biological roles.
Energy Storage: Lipids, especially fats, store metabolic energy efficiently due to their highly reduced carbon atoms.
Membrane Structure: Lipids form the structural basis of biological membranes, creating barriers and compartments within cells.
Signaling: Certain lipids act as signaling molecules, mediating cellular communication and responses.
The Molecular Structure and Behavior of Lipids
Fatty Acids
Fatty acids are the fundamental building blocks of many lipids. They consist of a hydrophilic carboxylate group attached to a long hydrocarbon chain (typically 12–24 carbons). Fatty acids are classified as saturated (no double bonds) or unsaturated (one or more cis double bonds). The degree of unsaturation and chain length influence the physical properties of fatty acids, such as melting point and fluidity.
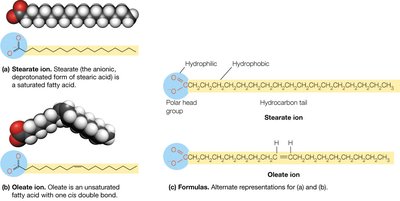
Saturated Fatty Acids: No double bonds; higher melting points; less fluid.
Unsaturated Fatty Acids: One or more cis double bonds; lower melting points; more fluid.
Fluidity: Decreases with increasing chain length and decreasing number of double bonds.
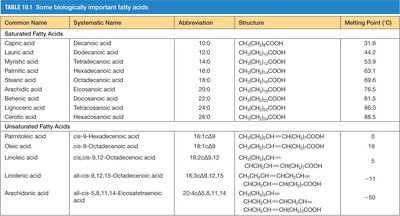
Common Name | Systematic Name | Abbreviation | Structure | Melting Point (°C) |
|---|---|---|---|---|
Capric acid | Decanoic acid | 10:0 | CH3(CH2)8COOH | 31.6 |
Palmitic acid | Hexadecanoic acid | 16:0 | CH3(CH2)14COOH | 63 |
Oleic acid | cis-9-Octadecenoic acid | 18:1(Δ9) | CH3(CH2)7CH=CH(CH2)7COOH | 16 |
Linoleic acid | cis,cis-9,12-Octadecadienoic acid | 18:2(Δ9,12) | CH3(CH2)4CH=CHCH2CH=CH(CH2)7COOH | -5 |
Fats (Triacylglycerols)
Fats, or triacylglycerols, are formed by the esterification of glycerol with three fatty acids. They serve as the primary storage form of energy in many organisms and provide thermal insulation. The high reduction state of their carbon atoms makes them efficient energy sources.
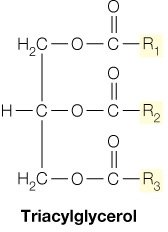
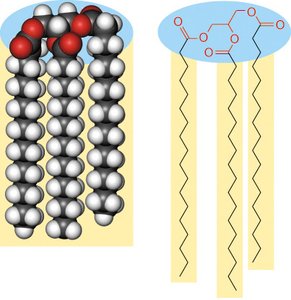
Structure: Glycerol backbone esterified with three fatty acids (R1, R2, R3).
Function: Energy storage, heat production, and insulation.
Soaps and Saponification
Soaps are produced by treating fats with strong bases (NaOH or KOH), resulting in the formation of fatty acid salts (soaps) and glycerol. This process is called saponification. Soaps form micelles around oils, aiding in emulsification. Synthetic detergents, such as sodium dodecyl sulfate (SDS), are not precipitated by calcium or magnesium ions, unlike soaps.
Saponification Value (SV): The amount of KOH (mg) needed to neutralize the fatty acids from 1 g of fat. SV is inversely proportional to fatty acid chain length.
Iodine Value (IV): The amount of iodine (g) absorbed by 100 g of fat, indicating the degree of unsaturation.
Formulas:
Saturated FA:
Unsaturated FA: (x = number of double bonds)
Waxes
Waxes are esters formed from long-chain fatty acids and long-chain alcohols. Due to their minimal polar regions, waxes are completely insoluble in water and serve as protective coatings in plants and animals.
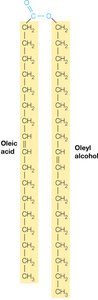
The Lipid Constituents of Biological Membranes
Lipids, Micelles, and Bilayers
Lipids are the primary constituents of biological membranes. Fatty acids tend to form spherical micelles, while lipids with two hydrophobic tails (such as phospholipids) form bilayers, the fundamental structure of biological membranes. The major classes of membrane-forming lipids include glycerophospholipids, glycoglycerolipids, sphingolipids, and glycosphingolipids.
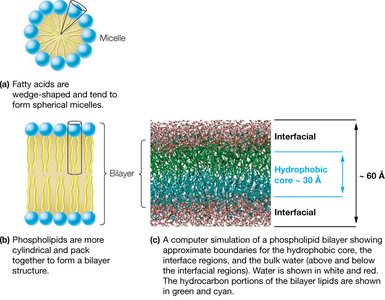
Micelles: Spherical structures formed by single-tailed lipids (e.g., fatty acids).
Bilayers: Planar structures formed by double-tailed lipids (e.g., phospholipids).
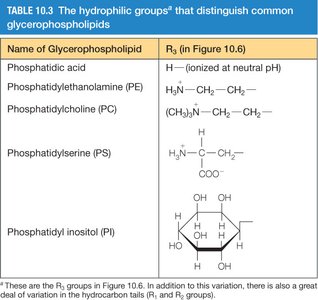
Glycerophospholipids
Glycerophospholipids are the major class of naturally occurring phospholipids, characterized by a glycerol backbone, two fatty acid tails, and a phosphate-containing head group. The nature of the head group determines the specific type of glycerophospholipid.
Name | Head Group (R3) |
|---|---|
Phosphatidic acid | H (ionized at neutral pH) |
Phosphatidylethanolamine (PE) | H2N–CH2–CH2– |
Phosphatidylcholine (PC) | (CH3)3N+–CH2–CH2– |
Phosphatidylserine (PS) | H3N+–CH–COO– |
Phosphatidylinositol (PI) | Inositol ring |
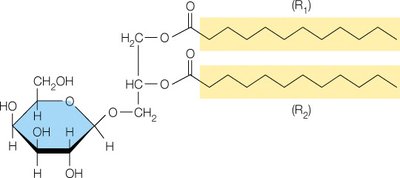
Glycoglycerolipids
Glycoglycerolipids are membrane lipids with a carbohydrate moiety linked to the head group. They are especially abundant in plant and bacterial membranes.
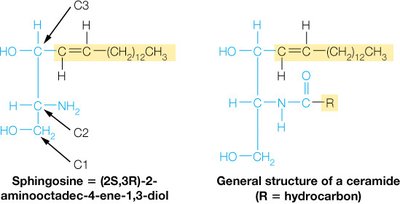
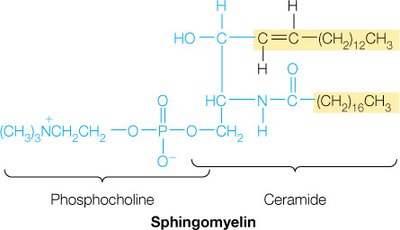
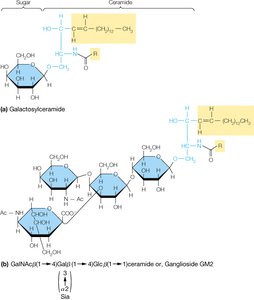
Sphingolipids
Sphingolipids are membrane lipids in which a fatty acid is linked to the amino alcohol sphingosine via an amide bond. They play key roles in membrane structure and cell recognition.
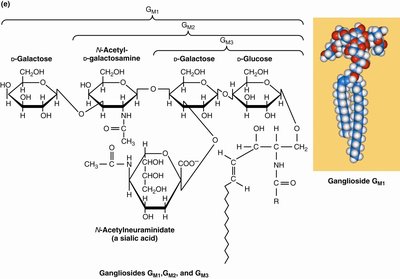
Glycosphingolipids
Glycosphingolipids are sphingolipids with carbohydrate groups attached to their head groups. They are important for cell-cell recognition and signaling.
Cholesterol
Cholesterol is a membrane lipid based on a tetracyclic hydrocarbon structure. It is only weakly amphipathic due to its single hydroxyl group. Cholesterol modulates membrane fluidity and is the precursor to all steroids.
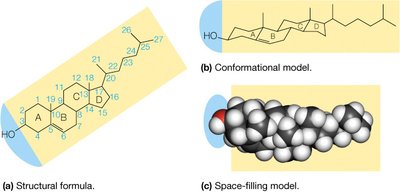
At low temperatures: Increases membrane fluidity by preventing fatty acid packing.
At high temperatures: Decreases membrane fluidity by restricting phospholipid movement.
Biological role: Maintains membrane integrity and fluidity; precursor for steroid hormones.
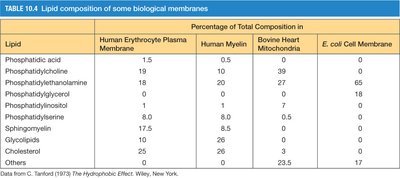
Lipid Composition of Biological Membranes
The lipid composition of biological membranes varies by cell type and organelle, reflecting functional specialization.
Lipid | Human Erythrocyte Plasma Membrane | Human Myelin | Bovine Heart Mitochondria | E. coli Cell Membrane |
|---|---|---|---|---|
Phosphatidic acid | 1.5 | 0.5 | 0 | 0 |
Phosphatidylcholine | 19 | 10 | 39 | 0 |
Phosphatidylethanolamine | 18 | 20 | 27 | 65 |
Phosphatidylglycerol | 0 | 0 | 0 | 0 |
Phosphatidylinositol | 8 | 0 | 7 | 0 |
Phosphatidylserine | 10 | 0 | 0 | 0 |
Sphingomyelin | 17.5 | 0 | 0 | 0 |
Glycolipids | 10 | 26 | 0 | 0 |
Cholesterol | 25 | 26 | 0 | 0 |
Others | 0 | 0 | 23.5 | 17 |
The Structure and Properties of Membranes and Membrane Proteins
Membrane Structure – The Fluid Mosaic Model
Biological membranes are described by the fluid mosaic model, in which a lipid bilayer forms a two-dimensional liquid matrix with embedded proteins. Lipids and proteins can diffuse laterally, but the membrane also contains specialized domains such as lipid rafts.
Lipid Rafts: Microdomains rich in cholesterol, sphingolipids, and certain proteins, often involved in signaling and protein sorting.
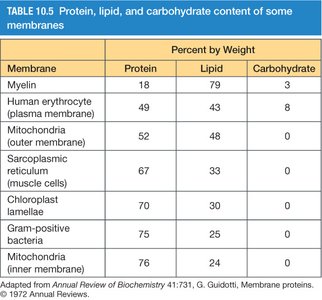
Evidence for Membrane Fluidity
Membrane fluidity is demonstrated by experiments in which fluorescently labeled membrane proteins mix after cell fusion, indicating lateral diffusion within the membrane. The rate of diffusion depends on membrane composition and temperature.
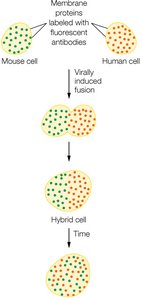
Gel State and Liquid Crystal State of Membranes
Membranes can exist in a gel state (ordered, less fluid) or a liquid crystal state (disordered, more fluid). The transition between these states occurs at the melting temperature (Tm), which is influenced by fatty acid chain length, degree of saturation, and cholesterol content.
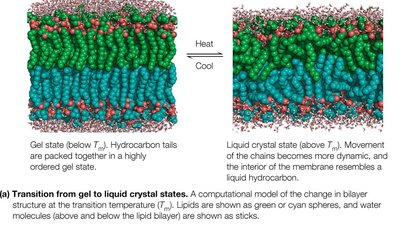
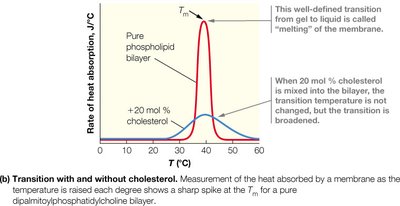
Longer chains and more saturation: Higher Tm.
Cholesterol: Broadens the transition, stabilizing membrane fluidity.
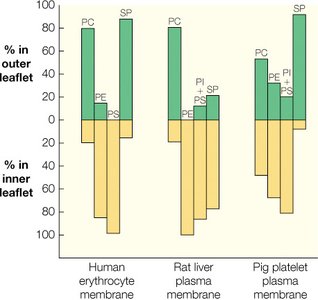
Asymmetry of Membrane Lipid Structure
Membrane lipid composition is asymmetric, with different phospholipids distributed unequally between the inner and outer leaflets of the bilayer. This asymmetry is crucial for membrane function and cell signaling.
Membrane Proteins
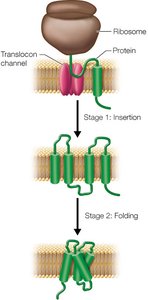
Membrane proteins can span the bilayer as α-helices or β-barrels, with hydrophobic regions interacting with the lipid tails. Their insertion into the membrane is a cotranslational process involving the ribosome and the translocon complex.
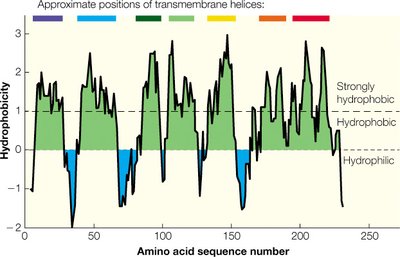
Hydrophobicity plots: Used to predict membrane-spanning regions in proteins.
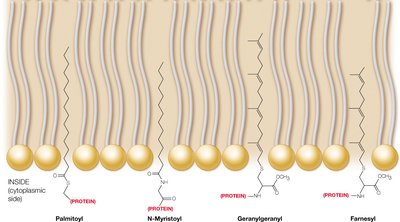
Protein Lipidation and Post-Translational Modifications
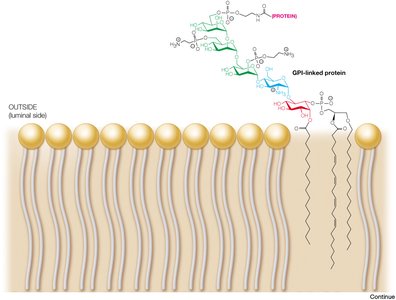
Many proteins are covalently modified with lipids, anchoring them to membranes. Glycosylphosphatidylinositol (GPI) anchors are found on the extracellular side, while other lipid modifications target proteins to specific membrane locations.
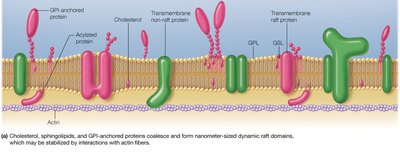
Membrane Rafts
Membrane rafts are dynamic, cholesterol- and sphingolipid-rich microdomains that organize the lateral distribution of proteins and lipids. They play roles in signaling and protein sorting, and their formation may be stabilized by actin fibers.
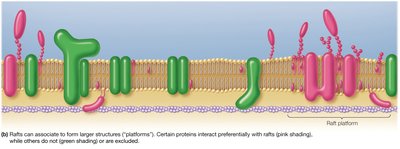
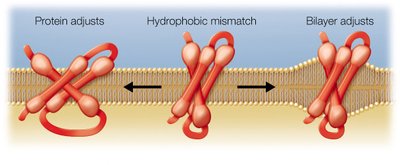
Adaptation to Hydrophobic Mismatch
Hydrophobic mismatch occurs when the hydrophobic regions of membrane proteins and the lipid bilayer do not align. Both proteins and the bilayer can adjust to minimize this mismatch, ensuring proper protein function and membrane integrity.