 Back
BackMetabolism Basics: Chemical Logic and Bioenergetics
Study Guide - Smart Notes

Metabolism: Core Principles and Overview
Introduction to Intermediary Metabolism
Metabolism encompasses the chemical processes that occur within living organisms to maintain life. Intermediary metabolism refers to the synthesis (anabolism) and degradation (catabolism) of small molecules, known as metabolic intermediates. These reactions are organized into metabolic pathways, which are highly regulated and interconnected to efficiently manage cellular energy and biosynthetic needs.
Catabolic pathways break down energy-containing nutrients (e.g., glucose, fatty acids) to produce ATP, NADH, and FADH2, releasing energy and generating low-energy products such as CO2 and H2O.
Anabolic pathways use ATP and reducing power to synthesize complex biomolecules (proteins, lipids, nucleic acids, carbohydrates) from simpler precursors.
Amphibolic pathways can function both anabolically and catabolically, depending on cellular needs.
Metabolic complexity arises from the intelligent reuse and regulation of a limited set of core reactions and intermediates, rather than from an endless variety of unique reactions.
Central Pathways of Energy Metabolism
Central metabolic pathways are the main routes through which cells extract and store energy. These include glycolysis, the citric acid cycle (Krebs or TCA cycle), oxidative phosphorylation, fatty acid oxidation, gluconeogenesis, fatty acid synthesis, and photosynthesis. In aerobic organisms, most pathways converge at the citric acid cycle.
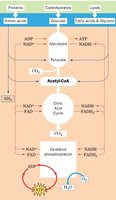
Glucose Metabolism and Glycolysis
Glucose is a primary energy source, metabolized through glycolysis to produce pyruvate in a series of ten enzymatic steps. Under anaerobic conditions, pyruvate is converted to lactate; under aerobic conditions, it is converted to acetyl-CoA, which enters the citric acid cycle for further oxidation to CO2.
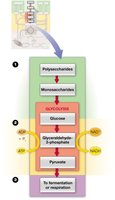
Bioenergetics of Metabolic Pathways
ATP: The Universal Energy Currency
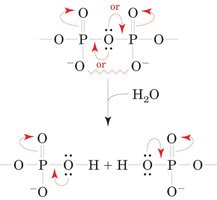
ATP (adenosine triphosphate) is the primary energy carrier in cells. It is produced mainly by the oxidation of carbon fuels and acts as a free-energy donor in most energy-requiring processes, including motion, active transport, and biosynthesis. The hydrolysis of ATP is highly exergonic, with a standard free energy change (ΔG°') of approximately −30.5 kJ·mol−1 for ATP → ADP + Pi.
High-energy phosphoanhydride bonds in ATP are responsible for its large negative ΔG of hydrolysis.
Other nucleoside triphosphates (GTP, UTP, CTP) can also drive biosynthetic reactions.
Phosphoryl-Transfer Potential
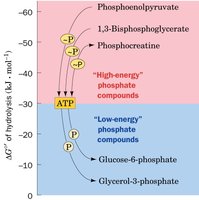
The phosphoryl-transfer potential of a molecule is a measure of its tendency to transfer a phosphoryl group to an acceptor. ATP has an intermediate phosphoryl-transfer potential, allowing it to accept and donate phosphate groups efficiently in metabolism.
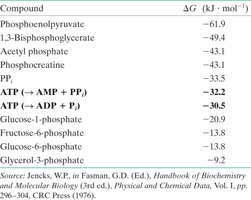
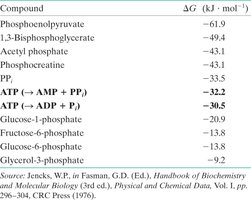
Compound | ΔG (kJ·mol−1) |
|---|---|
Phosphoenolpyruvate | −61.9 |
1,3-Bisphosphoglycerate | −49.4 |
Acetyl phosphate | −43.1 |
Phosphocreatine | −43.1 |
PPi | −33.5 |
ATP (→ AMP + PPi) | −32.2 |
ATP (→ ADP + Pi) | −30.5 |
Glucose-1-phosphate | −20.9 |
Fructose-6-phosphate | −13.8 |
Glucose-6-phosphate | −13.8 |
Glycerol-3-phosphate | −9.2 |
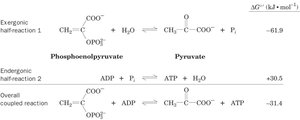
Coupling of Reactions
Thermodynamically unfavorable reactions (positive ΔG) can be driven by coupling them to favorable reactions (negative ΔG), such as ATP hydrolysis. This principle is essential for constructing metabolic pathways that proceed in the desired direction.
Structural Basis for ATP's High Phosphoryl Potential
The high phosphoryl-transfer potential of ATP is due to:
Resonance stabilization of hydrolysis products (especially inorganic phosphate, Pi).
Electrostatic repulsion among negatively charged phosphate groups in ATP.
Increase in entropy upon hydrolysis (one ATP yields two products: ADP and Pi).
Stabilization by hydration of the hydrolysis products.
Hierarchy of Phosphoryl-Transfer Compounds
ATP occupies an intermediate position among phosphorylated compounds, allowing it to serve as both a donor and acceptor of phosphate groups in cellular reactions.
Oxidation and Energy Capture
Oxidation of Carbon Fuels
Oxidation reactions involve the loss of electrons from fuel molecules, with the electrons ultimately transferred to oxygen in aerobic organisms. The more reduced a carbon atom, the more energy is released upon oxidation. Fats, being more reduced than carbohydrates, yield more energy per gram when oxidized.
Coupling Oxidation to ATP Synthesis
The energy released from the oxidation of metabolic intermediates (e.g., glyceraldehyde 3-phosphate) is often captured in the form of high-energy phosphate compounds, which are then used to synthesize ATP.

Regulation of Metabolic Pathways
Control Mechanisms
Metabolic pathways are regulated at key steps to ensure efficient energy use and biosynthesis. Regulation can occur through:
Allosteric control (e.g., feedback inhibition by ATP in glycolysis).
Covalent modification (e.g., phosphorylation by protein kinases).
Compartmentalization of opposing pathways in different cellular locations.
Regulation of enzyme synthesis and degradation.
Energy Charge
The energy charge of a cell, defined by the relative concentrations of ATP, ADP, and AMP, regulates the rates of catabolic and anabolic pathways. High energy charge inhibits ATP-generating pathways and stimulates ATP-utilizing pathways.
Photosynthesis and Energy Conversion
Photosynthetic ATP Synthesis
In chloroplasts, high-energy electrons are transported through two photosystems, generating reducing power and synthesizing ATP in a process analogous to mitochondrial oxidative phosphorylation. However, in photosynthesis, electrons are energized by light rather than by the oxidation of carbon fuels.
Summary Table: Key Phosphorylated Intermediates and Their ΔG of Hydrolysis
Compound | ΔG (kJ·mol−1) |
|---|---|
Phosphoenolpyruvate | −61.9 |
1,3-Bisphosphoglycerate | −49.4 |
Acetyl phosphate | −43.1 |
Phosphocreatine | −43.1 |
PPi | −33.5 |
ATP (→ AMP + PPi) | −32.2 |
ATP (→ ADP + Pi) | −30.5 |
Glucose-1-phosphate | −20.9 |
Fructose-6-phosphate | −13.8 |
Glucose-6-phosphate | −13.8 |
Glycerol-3-phosphate | −9.2 |
Key Equations
ATP hydrolysis:
Standard free energy change:
Coupled reaction example (phosphoenolpyruvate and ADP):

Additional info:
ATP may also function as a hydrotrope, helping to keep proteins soluble in the crowded cellular environment.
Metabolic flux refers to the flow of material through a metabolic pathway.
Control points in pathways are typically irreversible steps, often catalyzed by enzymes with high flux control coefficients.
