 Back
BackNucleotides and Nucleic Acids: Structure, Function, and Biological Roles
Study Guide - Smart Notes

Nucleotides and Nucleic Acids
Introduction
Nucleotides and nucleic acids are fundamental biomolecules essential for the storage, transmission, and expression of genetic information. This section explores their structures, functions, and the chemical principles underlying their roles in biochemistry.
Functions of Nucleotides and Nucleic Acids
Biological Roles
Genetic Information Storage: Deoxyribonucleic acid (DNA) stores hereditary information in all living organisms.
Transmission of Genetic Information: Messenger RNA (mRNA) carries genetic instructions from DNA to ribosomes for protein synthesis.
Processing of Genetic Information: Ribozymes (catalytic RNA molecules) participate in RNA processing and catalysis.
Protein Synthesis: Transfer RNA (tRNA) and ribosomal RNA (rRNA) are essential for translating genetic code into proteins.
Energy Currency: Adenosine triphosphate (ATP) provides energy for cellular metabolism.
Enzyme Cofactors: Nicotinamide adenine dinucleotide (NAD+) and others are involved in redox reactions.
Signal Transduction: Cyclic adenosine monophosphate (cAMP) acts as a second messenger in cellular signaling pathways.
Structure of Nucleotides and Nucleosides
Basic Components
Nucleotide: Composed of a nitrogenous base, a pentose sugar, and one or more phosphate groups.
Nucleoside: Consists of a nitrogenous base and a pentose sugar (no phosphate group).
Nitrogenous Bases
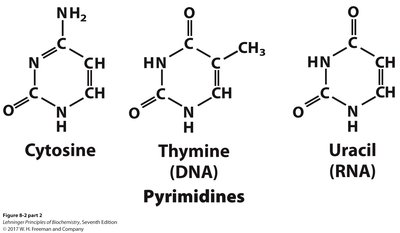
Pyrimidines: Cytosine, Thymine (DNA), Uracil (RNA)
Purines: Adenine, Guanine
All bases are planar, aromatic, and absorb UV light (250–270 nm).
Pentose Sugars
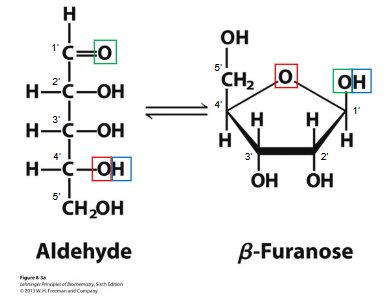
Ribose: Found in RNA; contains a 2'-hydroxyl group.
Deoxyribose: Found in DNA; lacks a 2'-hydroxyl group (has H instead).
Sugars exist in β-furanose (ring) form in nucleic acids.
Phosphate Group
Typically attached to the 5' carbon of the sugar.
Can be present as mono-, di-, or triphosphate (e.g., AMP, ADP, ATP).
Phosphates are negatively charged at physiological pH.
Linkages and Directionality
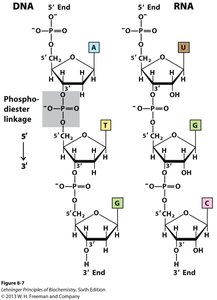
Phosphodiester Bonds
Nucleotides are linked via phosphodiester bonds between the 3'-OH of one sugar and the 5'-phosphate of the next.
This forms the sugar-phosphate backbone of DNA and RNA, imparting directionality (5' → 3').
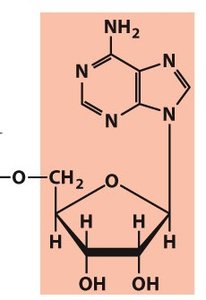
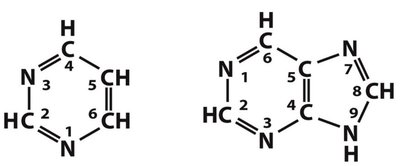
β-N-Glycosidic Bond
Connects the base to the sugar at the 1' carbon (N1 of pyrimidines, N9 of purines).
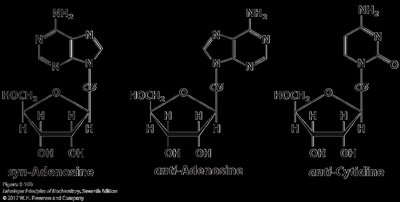
Allows for syn and anti conformations; anti is most common in nucleic acids.
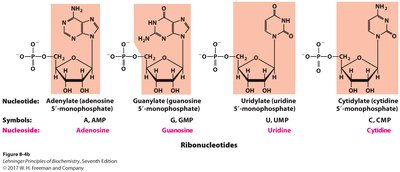
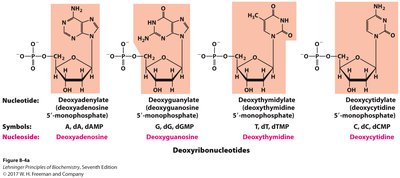
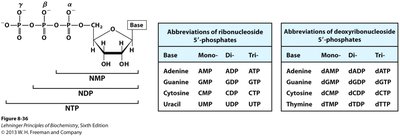
Nomenclature of Nucleotides
Ribonucleotides and Deoxyribonucleotides
Systematic names are based on the base, sugar, and number of phosphates.
Prefixes: 'deoxy' for DNA nucleotides; abbreviations use 'd' (e.g., dATP).
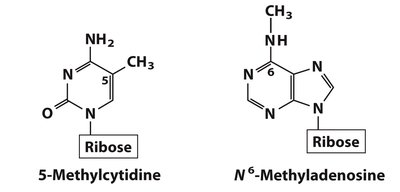
Minor and Modified Nucleosides
DNA Modifications
5-Methylcytidine: Common in eukaryotes; involved in epigenetic regulation.
N6-Methyladenosine: Found in bacteria; marks DNA for repair and regulation.
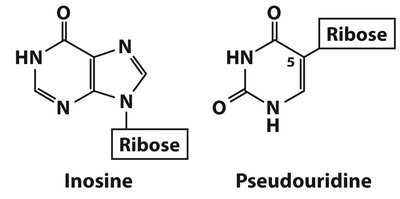
RNA Modifications
Inosine: Found in tRNA; enhances codon recognition.
Pseudouridine (Ψ): Found in tRNA and rRNA; stabilizes RNA structure.
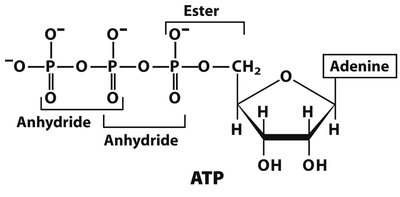
High-Energy Phosphoanhydride Bonds
ATP and Energy Transfer
ATP contains two high-energy phosphoanhydride bonds between its three phosphate groups.
Hydrolysis of these bonds releases energy for cellular processes.
DNA Structure and Base Pairing
Primary and Secondary Structure
Primary Structure: Linear sequence of nucleotides.
Secondary Structure: Double helix formed by complementary base pairing (Watson-Crick pairs).
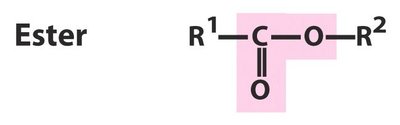
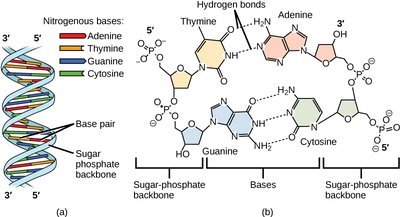
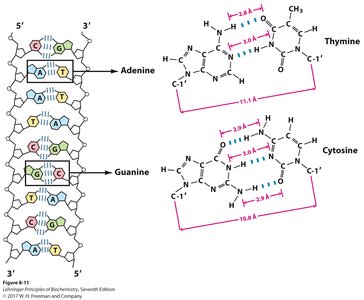
Base Pairing Rules
Adenine (A) pairs with Thymine (T) via 2 hydrogen bonds.
Guanine (G) pairs with Cytosine (C) via 3 hydrogen bonds.
Base stacking interactions further stabilize the helix.
DNA Denaturation and Renaturation
Denaturation
Loss of secondary structure (double helix) due to heat or pH changes; primary structure remains intact.
Measured by melting temperature (Tm), which increases with GC content and DNA length.
Renaturation (Annealing)
Reformation of the double helix when conditions return to normal.
RNA Structure and Types
Types of RNA
mRNA (Messenger RNA): Carries genetic code from DNA to ribosomes.
tRNA (Transfer RNA): Brings amino acids to ribosomes during translation.
rRNA (Ribosomal RNA): Structural and catalytic component of ribosomes.
miRNA (Micro-RNA): Regulates gene expression post-transcriptionally.
RNA Secondary Structures
Hairpins, bulges, internal loops, pseudoknots, and multi-branched loops are common secondary structures formed by intramolecular base pairing.
These structures are critical for RNA function, stability, and interactions.
Summary Table: Key Bonds in DNA and RNA
Bond Type | DNA | RNA | Function |
|---|---|---|---|
Phosphodiester Bonds (Covalent) | Present | Present | Forms the sugar-phosphate backbone |
β-N-Glycosidic Bond (Covalent) | Present | Present | Links base to sugar |
Hydrogen Bonds (Non-Covalent) | Present (between strands) | Present (in secondary structures) | Stabilizes base pairing |
Base Stacking (Van der Waals & Hydrophobic) | Present | Present (weaker) | Additional structural stability |
Ionic Interactions (Electrostatic) | Present | Present | Neutralizes negative charge, aids folding |
Conclusion
Nucleotides and nucleic acids are central to the molecular basis of life. Their structures, chemical properties, and modifications enable the storage, transmission, and regulation of genetic information, as well as the catalysis and regulation of essential cellular processes.
