 Back
BackOxidation State Determination for Carbon Atoms in Biochemical Compounds
Study Guide - Smart Notes

Q1. Determine the oxidation state for each carbon atom in the following compounds:
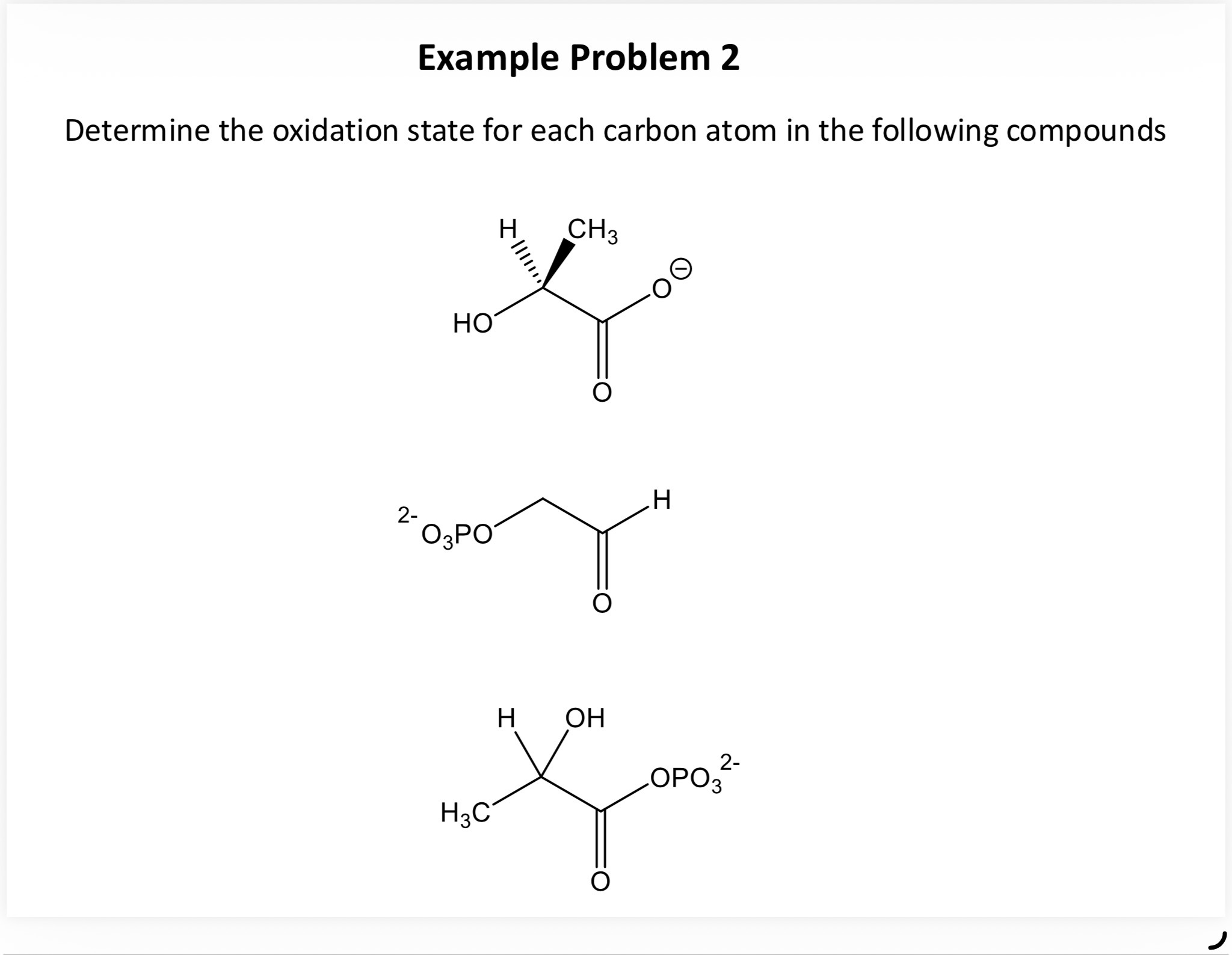
Background
Topic: Oxidation States in Organic/Biochemical Molecules
This question tests your ability to assign oxidation states to carbon atoms in complex organic molecules, a fundamental skill in biochemistry for understanding metabolic pathways and redox reactions.
Key Terms and Formulas
Oxidation State: The hypothetical charge an atom would have if all bonds to atoms of different elements were 100% ionic.
Rules for Assigning Oxidation States:
Each bond to a more electronegative atom (O, N, P, etc.) counts as +1 for carbon.
Each bond to a less electronegative atom (H) counts as -1 for carbon.
Bonds to other carbons do not affect the oxidation state.
Formula:
Step-by-Step Guidance
Identify each carbon atom in the molecule and label them (e.g., C1, C2, C3).
For each carbon, count the number of bonds to more electronegative atoms (O, N, P, etc.) and less electronegative atoms (H).
Apply the formula: for each carbon atom.
Remember to consider double bonds as two single bonds, and triple bonds as three.
Write out the oxidation state for each carbon atom, but stop before calculating the final numeric values for each.
Try solving on your own before revealing the answer!
Final Answer:
Each carbon atom's oxidation state is determined by counting its bonds to O, H, and other atoms as described. For example, a methyl carbon (CH3) attached to a carbonyl will have an oxidation state of -3, while a carbonyl carbon will be +3 or higher depending on its environment.
Carefully apply the rules to each carbon in the three compounds shown above to get the final values.
