 Back
BackPlasma Lipoproteins: Structure, Metabolism, and Clinical Implications
Study Guide - Smart Notes

Plasma Lipoproteins
Structure and Composition of Lipoproteins
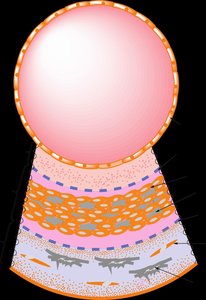
Lipoproteins are complex particles essential for the transport of lipids in the plasma. Their structure consists of an outer shell of apolipoproteins and phospholipids, surrounding a hydrophobic core containing triacylglycerols (TG) and cholesteryl esters. Most lipoproteins are spherical, except for newly secreted high-density lipoproteins (HDL), which are discoidal and become spherical through the action of lecithin-cholesterol acyltransferase (LCAT).
Apolipoproteins are amphipathic helices that stabilize lipoprotein structure and mediate interactions with enzymes and receptors.
Chylomicrons and VLDL are principal carriers of TG.
LDL and HDL are major cholesterol transporters.
Example: The amphipathic nature of apolipoproteins allows them to interact with both the hydrophobic lipid core and the aqueous plasma environment. 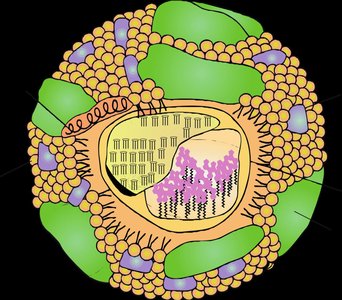
Physical Data and Composition of Lipoproteins
Lipoproteins differ in size, density, and composition, which determines their function and metabolic fate.
Chylomicrons: Largest, lowest density, highest TG content.
VLDL: High TG, lower density than LDL.
LDL: Rich in cholesterol, intermediate density.
HDL: Smallest, highest density, rich in protein.
Example: The composition of lipoproteins affects their buoyancy and their role in lipid transport. 
Lipoprotein Metabolism
Key Enzymes in Lipoprotein Metabolism
Several enzymes regulate the metabolism of lipoproteins, facilitating lipid transport and storage.
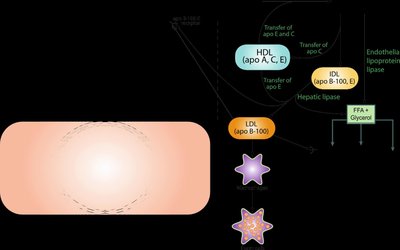
Lipoprotein lipase (LPL): A serine esterase concentrated in muscle and adipose tissue, activated by apo C-II, hydrolyzes TG in chylomicrons and VLDL.
Hepatic lipase: Located on liver cell membranes, hydrolyzes TG and phospholipids in VLDL, IDL, and HDL.
LCAT: Activated by apo A-I, converts cholesterol to cholesteryl esters in HDL.
ACAT: Intracellular enzyme that esterifies cholesterol for storage.
Example: Insulin modulates LPL activity, linking lipid metabolism to energy status.
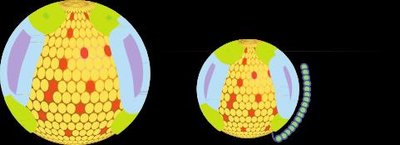
Chylomicron Metabolism (Exogenous Pathway)
Chylomicrons are assembled in the small intestine and transport dietary TG and cholesterol.
Acquire apolipoproteins (Apo B-48, A-I, A-II, A-IV) in enterocytes.
Secreted into lymph, enter blood via thoracic duct.
In circulation, acquire apo C and E from HDL, TG hydrolyzed by LPL.
Remnants rich in cholesteryl ester and apo B-48/E are removed by the liver.
Example: Chylomicron remnants are taken up by liver cells via apo E receptor-mediated endocytosis. 



Very-Low-Density Lipoprotein (VLDL) Metabolism (Endogenous Pathway)
VLDL is produced by the liver and transports endogenous TG and cholesterol.
Contains apo B-100, C-II, C-III, and E.
Acquires apo C and E from HDL in plasma.
TG hydrolyzed by LPL, apo C lost, VLDL remnants (IDL) formed.
IDL further catabolized by hepatic lipase, loses apo E, becomes LDL.
Example: LDL delivers cholesterol to peripheral tissues, maintaining balance between dietary and endogenous cholesterol. 


Low-Density Lipoprotein (LDL) and Cholesterol Distribution
LDL is the primary carrier of cholesterol to peripheral tissues.
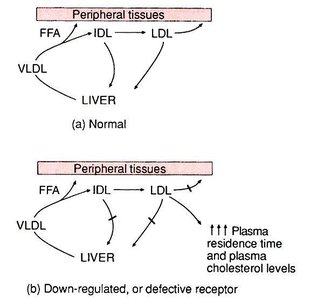
LDL receptor is a glycoprotein with five domains, binds apo B and E.
Number of LDL receptors is regulated by intracellular cholesterol levels.
Free cholesterol released into cells is used for membrane synthesis, inhibits new LDL receptor and cholesterol synthesis, stimulates ACAT and bile acid synthesis.
About 75% of LDL uptake occurs in the liver.
Example: LDL receptor-mediated endocytosis is crucial for cholesterol homeostasis. 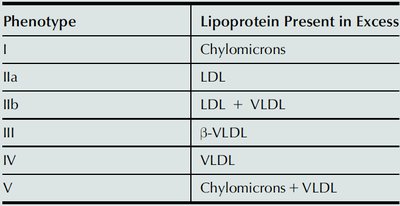
Clinical Implications: Hyperlipoproteinemia and Cardiovascular Disease
Hyperlipoproteinemia: Classification and Genetic Defects
Hyperlipoproteinemias are classified based on the excess lipoprotein present and are often caused by inherited defects.
Phenotype | Lipoprotein Present in Excess |
|---|---|
I | Chylomicrons |
IIa | LDL |
IIb | LDL + VLDL |
III | β-VLDL |
IV | VLDL |
V | Chylomicrons + VLDL |
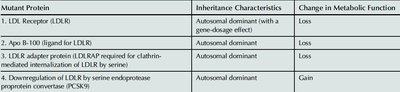
Mutant Protein | Inheritance Characteristics | Change in Metabolic Function |
|---|---|---|
LDL Receptor (LDLR) | Autosomal dominant (with a gene-dosage effect) | Loss |
Apo B-100 ligand for LDLR | Autosomal dominant | Loss |
LDLR adapter protein (LDLRAP) | Autosomal dominant | Loss |
Downregulation of LDLR by PCSK9 | Autosomal dominant | Gain |
Hypercholesterolemia and Atherosclerosis
Elevated plasma cholesterol, especially LDL cholesterol, is a major risk factor for coronary heart disease (CHD).
Reduction in plasma cholesterol lowers CHD risk.
Statins reduce both fatal and nonfatal heart attacks.
Premature CHD is strongly associated with high LDL and low HDL cholesterol.
HDL cholesterol has antiatherogenic effects, including inhibition of LDL oxidation and prevention of monocyte adhesion to endothelium.
Example: HDL prolongs the half-life of prostacyclin, promoting vasodilation and protecting against atherosclerosis. 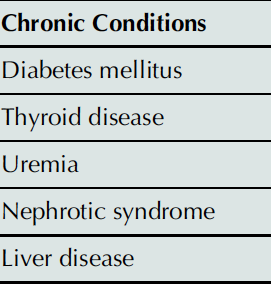
Evaluation and Measurement of Plasma Cholesterol
Plasma cholesterol levels are assessed to evaluate CHD risk.
LDL cholesterol is calculated as:
Alternatively:
HDL cholesterol is measured after precipitation of non-HDL lipoproteins.
Direct measurement uses immunoseparation with antibodies against apo A-I and E.
Example: Measurement of apo A-I, apo B, and apo E is useful for CHD risk assessment. 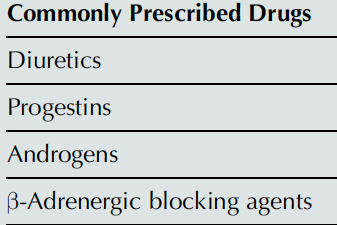
Secondary Causes of Altered Plasma Lipoprotein Levels
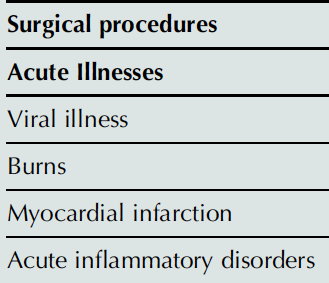
Plasma lipoprotein levels can be affected by various secondary causes, including acute illnesses, chronic conditions, and commonly prescribed drugs.
Surgical procedures |
|---|
Acute Illnesses |
Viral illness |
Burns |
Myocardial infarction |
Acute inflammatory disorders |
Chronic Conditions |
|---|
Diabetes mellitus |
Thyroid disease |
Uremia |
Nephrotic syndrome |
Liver disease |
Commonly Prescribed Drugs |
|---|
Diuretics |
Progestins |
Androgens |
β-Adrenergic blocking agents |
Lipid-Lowering Methods
Lipid-lowering strategies are essential for the prevention and treatment of cardiovascular diseases.
Dietary modification: Reduce cholesterol and saturated fat intake.
Drug therapy: Includes cholestyramine, colestipol, nicotinic acid, clofibrate, gemfibrozil, probucol, and statins.
Statins inhibit cholesterol biosynthesis and upregulate LDL receptor activity, lowering serum cholesterol and LDL.
Example: Statins are the most effective drugs for lowering LDL cholesterol and reducing CHD risk.
Summary
Plasma lipoproteins play a central role in lipid transport, metabolism, and cardiovascular health. Understanding their structure, function, and clinical implications is essential for biochemistry students and healthcare professionals. Additional info: The notes include expanded explanations and context for clarity and completeness.
