 Back
BackProtein Purification and Analytical Techniques in Biochemistry
Study Guide - Smart Notes

Protein Purification
Importance of Protein Purification
Protein purification is a fundamental skill in biochemistry, enabling the study of protein function, structure, and application. Pure proteins are required for biochemical assays, structural determination, medical and industrial uses, and proteomics.
Understanding Protein Function: Purified proteins allow for accurate study of enzymatic activity, substrate specificity, and inhibition.
Structural Studies: Techniques like X-ray crystallography and NMR require highly purified proteins.
Medical & Industrial Applications: Many drugs and biotechnological products are proteins, necessitating rigorous purification.
Proteomics & Systems Biology: Purification is essential for identifying and characterizing proteins in complex mixtures.
Training in Laboratory Skills: Purification involves chromatography, electrophoresis, centrifugation, and quantification.
Protein Isolation Strategies
Isolation begins with selecting a rich source of protein and lysing cells to release proteins. Molecular cloning can be used to over-express proteins in host systems.
Cell Lysis Methods: Hypotonic shock, enzymatic digestion (e.g., lysozyme), mechanical disruption (blenders, French press, sonication).
Differential Centrifugation: Separates cellular components by size and density, yielding fractions like mitochondria, ribosomes, and soluble proteins.
Protein Stability and Denaturation
Proteins are sensitive to environmental conditions. Denaturation disrupts their native conformation, affecting function.
Factors Affecting Stability: Temperature, pH, proteases, oxidation, heavy metals, and bacterial contamination.
Denaturation: Loss of native structure due to disruption of hydrogen bonds, ionic bonds, Van der Waals, and hydrophobic interactions.
Activity Measurements
Enzyme activity is monitored to assess purity during purification. Spectrophotometric analysis is commonly used, applying Beer’s Law:
Beer’s Law:
Specific Activity: Activity per mg of protein, increases as purity increases.
Coupled Reactions: Used when direct assay is difficult; product of one reaction is used in another measurable reaction.
Radioactivity: Used for tracking molecules in reactions (e.g., ATP hydrolysis).
Fractionation and Purification Strategies
Physical Characteristics and Methods
Proteins can be separated based on charge, polarity, size, specificity, and solubility.
Charge: Ion exchange chromatography, electrophoresis, isoelectric focusing.
Polarity: Adsorption chromatography, paper chromatography, reverse phase chromatography, hydrophobic interaction.
Size: Dialysis, ultrafiltration, gel electrophoresis, gel filtration, ultracentrifugation.
Specificity: Affinity chromatography, immunopurification.
Solubility: Salt precipitation, detergent solubilization.
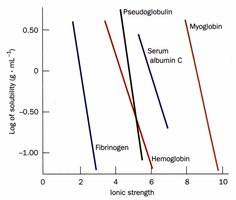
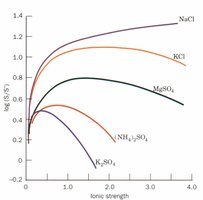
Ionic Strength and Salting Out
Ionic strength affects protein solubility and is used in purification steps like salting out. The ionic strength () is calculated as:
Formula:
Salting Out: (NH4)2SO4 is commonly used to precipitate proteins without harming them.
Chromatography Techniques
Principles of Chromatography
Chromatography separates molecules using a mobile phase and a stationary phase. The method depends on the chemistry of the molecules and the phases.
Types: Gas-liquid, liquid-liquid, ion exchange, adsorption, gel filtration, affinity chromatography.
Ion Exchange Chromatography
Ion exchange chromatography separates proteins based on charge. Anion and cation exchange resins are used depending on the protein’s net charge at a given pH.
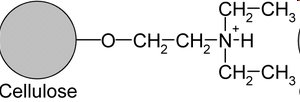
Anion Exchange: Uses positively charged resins (e.g., DEAE-cellulose) to bind negatively charged proteins.
Cation Exchange: Uses negatively charged resins (e.g., carboxymethyl cellulose) to bind positively charged proteins.
Elution: Proteins are eluted by increasing ionic strength or changing pH.
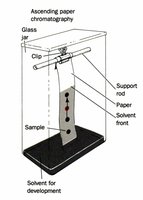
Paper Chromatography
Paper chromatography separates molecules based on partitioning between a stationary phase (paper) and a mobile phase (solvent). The partition coefficient determines migration rate.
Visualization: Radioactivity, fluorescence, UV absorbency, and dyes (e.g., ninhydrin).
Gel Filtration (Size Exclusion) Chromatography
Gel filtration separates proteins based on size. The matrix contains pores; small proteins enter the pores and elute later, while large proteins are excluded and elute earlier.
Common Matrices: Dextran, agarose, polyacrylamide.
Applications: Determining molecular mass, desalting proteins.
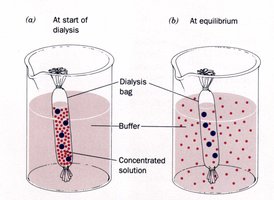
Dialysis
Dialysis uses semipermeable membranes to separate molecules by size. Small molecules diffuse out, while large proteins remain inside the dialysis bag.
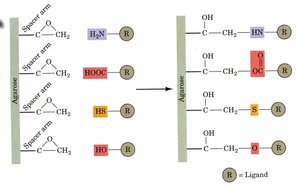
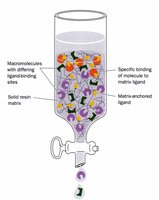
Affinity Chromatography
Affinity chromatography exploits specific binding between a protein and a ligand attached to a matrix. Only proteins with affinity for the ligand bind, allowing for highly selective purification.
Example: Citrate dehydrogenase binds to citrate-linked matrix; eluted with excess citrate.
Purification Tables
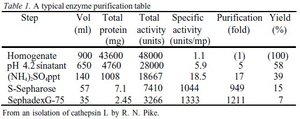
Interpreting Purification Tables
Purification tables summarize the progress of protein purification, showing volume, total protein, total activity, specific activity, purification fold, and yield.
Step | Vol (ml) | Total protein (mg) | Total activity (units) | Specific activity (units/mg) | Purification (fold) | Yield (%) |
|---|---|---|---|---|---|---|
Homogenate | 900 | 43600 | 48000 | 1.1 | 1 | 100 |
pH 4.2 sinatant | 650 | 4760 | 28000 | 5.9 | 5 | 58 |
(NH4)2SO4 ppt | 140 | 1088 | 18667 | 18.5 | 17 | 39 |
S-Sepharose | 57 | 7.1 | 7410 | 1044 | 949 | 15 |
Sephadex G-75 | 35 | 2.45 | 3266 | 1333 | 1211 | 7 |
Electrophoresis
Principles of Electrophoresis
Electrophoresis separates molecules based on their migration in an electric field, which depends on charge and size. The frictional force opposes migration.
Formula: and
Types: Paper electrophoresis, gel electrophoresis (polyacrylamide, agarose).
Visualization: Dyes (Coomassie blue, silver stain), fluorescence, radioactivity.
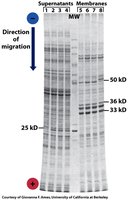
SDS-PAGE
SDS-PAGE uses sodium dodecyl sulfate to denature proteins and impart a uniform negative charge, allowing separation by molecular mass.
Isoelectric Focusing
Isoelectric focusing separates proteins based on their isoelectric point (pI) in a pH gradient. Proteins migrate until they reach the pH where their net charge is zero.
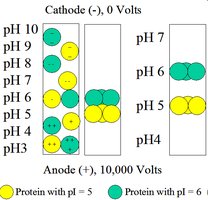
Two-Dimensional Gel Electrophoresis
Combines isoelectric focusing and SDS-PAGE for maximum separation, useful for proteomics and detecting post-translational modifications.
Summary Table: Protein Purification Methods
Characteristic | Method |
|---|---|
Charge | Ion exchange chromatography, electrophoresis, isoelectric focusing |
Polarity | Adsorption chromatography, paper chromatography, reverse phase chromatography, hydrophobic interaction |
Size | Dialysis, ultrafiltration, gel electrophoresis, gel filtration, ultracentrifugation |
Specificity | Affinity chromatography, immunopurification |
Solubility | Salt precipitation, detergent solubilization |
Additional info: Academic context was added to clarify the principles, applications, and formulas for each technique, and to ensure completeness for exam preparation.
