 Back
BackRespiration and Metabolism: Biochemical Pathways in Human Physiology
Study Guide - Smart Notes

Respiration and Metabolism
Introduction to Metabolism
Metabolism encompasses all chemical reactions occurring within the body, divided into two main categories: anabolism and catabolism. Anabolism requires energy to synthesize large molecules, while catabolism releases energy by breaking down large molecules into smaller ones. Catabolic reactions drive the production of ATP, which is essential for anabolic processes. Many metabolic reactions involve oxidation-reduction events, and the complete catabolism of glucose requires oxygen as the final electron acceptor, a process known as aerobic cellular respiration.
Glycolysis and the Lactic Acid Pathway
Glycolysis: The First Step in Glucose Catabolism
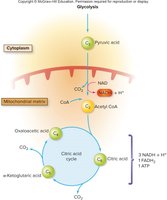
Glycolysis is the initial pathway in the breakdown of glucose, occurring in the cytoplasm and functioning anaerobically. The process begins with the phosphorylation of glucose, preventing its diffusion out of the cell. Glycolysis converts one molecule of glucose (C6H12O6) into two molecules of pyruvic acid (C3H4O3), with a net gain of 2 ATP and 2 NADH. Key enzymes involved include kinases, phosphatases, isomerases, and dehydrogenases.
Location: Cytoplasm
Products: 2 pyruvic acid, 2 NADH, 2 ATP
Net ATP: 2 ATP per glucose
Key Reaction:
Decision Point: Fate of Pyruvic Acid
After glycolysis, pyruvic acid can enter one of two pathways depending on the presence of oxygen:
Aerobic pathway: Pyruvic acid enters the mitochondria for further oxidation.
Anaerobic pathway: Pyruvic acid is converted to lactic acid.
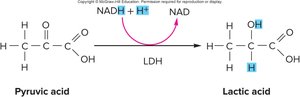
Lactic Acid Pathway (Anaerobic Metabolism)
In the absence of oxygen, NADH donates electrons to pyruvic acid, forming lactic acid and regenerating NAD+. This process, called fermentation, does not produce additional ATP beyond the initial 2 from glycolysis. Muscle cells can temporarily use this pathway, but it is not favored due to the risk of acidosis and low ATP yield. Red blood cells rely exclusively on lactic acid fermentation due to their lack of mitochondria. Excess lactic acid is transported to the liver, where it is converted back to pyruvic acid and NADH by lactic acid dehydrogenase, and can be used to regenerate glucose or glycogen.
Key Reaction:
Physiological significance: Allows ATP production in low oxygen conditions, but risks acidosis and is inefficient.
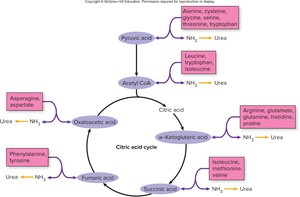
Aerobic Respiration: Citric Acid Cycle (TCA/Krebs Cycle)
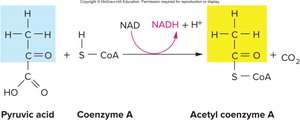
Transition Step and Entry into the Citric Acid Cycle
If oxygen is present, pyruvic acid enters the mitochondrial matrix and undergoes a transition step, where it is decarboxylated to form acetic acid, which then combines with coenzyme A to form acetyl CoA. This step is essential for linking glycolysis to the citric acid cycle.
Key Reaction:
Citric Acid Cycle (Krebs/TCA Cycle)
Acetyl CoA enters the citric acid cycle by combining with oxaloacetic acid to form citric acid. The cycle regenerates oxaloacetic acid and produces ATP, NADH, FADH2, and CO2. The cycle is aerobic and occurs in the mitochondrial matrix.
Products per glucose: 2 ATP, 6 NADH, 2 FADH2, 4 CO2
Coenzymes: NAD+ and FAD
Function of coenzymes: Electron carriers for oxidative phosphorylation
Aerobic Respiration: Electron Transport Chain (ETC) and Oxidative Phosphorylation
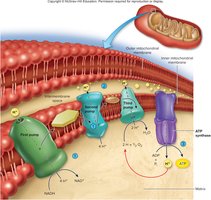
Electron Transport Chain (ETC)
The ETC is located on the inner mitochondrial membrane and is responsible for converting the energy stored in NADH and FADH2 into ATP. Electrons are passed along a series of transporters, pumping H+ ions into the intermembrane space, creating a gradient. ATP synthase uses this gradient to synthesize ATP from ADP and Pi. Oxygen serves as the final electron acceptor, forming water as a byproduct.
Key Reaction:
ATP yield: Each NADH yields ~2.5 ATP, each FADH2 yields ~1.5 ATP
Total ATP per glucose: 30-32 (actual), 36-38 (theoretical)
Interconversion of Glucose, Lactic Acid, and Glycogen
Storing and Mobilizing Glucose
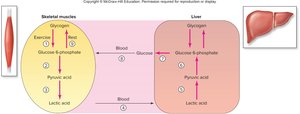
Cells cannot store glucose directly due to osmotic effects; instead, glucose is converted to glucose-6-phosphate and then stored as glycogen via glycogenesis. Glycogen is found in the liver, skeletal muscle, and cardiac muscle. When glucose is needed, glycogen is broken down via glycogenolysis to glucose-1-phosphate, then glucose-6-phosphate. Only the liver can release free glucose into the bloodstream due to the presence of glucose-6-phosphatase.
Glycogenesis: Formation of glycogen from glucose
Glycogenolysis: Breakdown of glycogen to glucose-1-phosphate
Locations: Liver, skeletal muscle, cardiac muscle
Cori Cycle and Gluconeogenesis
The Cori cycle describes the transport of lactic acid from muscle to liver, where it is converted back to pyruvic acid and then to glucose via gluconeogenesis. This glucose can be returned to muscle cells or stored as glycogen.
Gluconeogenesis: Synthesis of glucose from non-carbohydrate precursors
Cori Cycle: Occurs between muscle and liver, recycling lactic acid
Metabolism of Lipids and Proteins
Lipid Metabolism: Lipogenesis and Lipolysis
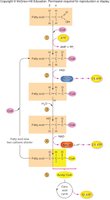
Excess glucose is converted to fatty acids and triglycerides via lipogenesis. When energy is needed, triglycerides are broken down into fatty acids and glycerol via lipolysis. Fatty acids undergo β-oxidation to form acetyl CoA, which enters the citric acid cycle. Glycerol can be converted to glucose in the liver.
Lipogenesis: Formation of lipids from acetyl CoA
Lipolysis: Breakdown of triglycerides to fatty acids and glycerol
β-oxidation: Fatty acids → acetyl CoA (1 acetyl CoA per 2 carbons)
ATP yield: 16-carbon fatty acid → 108 ATP
Ketogenesis
When fatty acid breakdown exceeds utilization, the liver converts acetyl CoA into ketone bodies via ketogenesis. Ketone bodies are water-soluble and can be used as energy sources by various tissues. Excess ketone bodies can lead to ketosis.
Amino Acid Metabolism
Amino acids are primarily used for protein synthesis but can be converted to glucose or fat if in excess. The conversion of amino acids to glucose is another form of gluconeogenesis. Amino acids can also be synthesized from citric acid cycle intermediates by transamination.
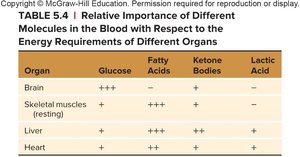
Summary: Energy Source Preferences by Organ
Different organs preferentially use different energy sources. The brain relies heavily on glucose, while muscles and the liver can utilize fatty acids, ketone bodies, and lactic acid.
Organ | Glucose | Fatty Acids | Ketone Bodies | Lactic Acid |
|---|---|---|---|---|
Brain | +++ | – | + | – |
Skeletal muscles (resting) | + | +++ | + | – |
Liver | ++ | ++ | + | + |
Heart | + | ++ | ++ | + |
Additional info: These notes expand on the original content to provide a comprehensive, self-contained study guide suitable for biochemistry students, including definitions, equations, and organ-specific energy preferences.
