 Back
BackStructural Proteins: Keratin, Silk, and Collagen
Study Guide - Smart Notes

Structural Proteins
Overview
Structural proteins are essential for providing mechanical support and integrity to cells and tissues. Their function is dictated by their amino acid sequence and secondary structure. Three major fibril proteins—keratin, silk fibroin, and collagen—exemplify the relationship between protein structure and function.
Keratin
Structure and Function
Keratin is a family of fibrous proteins found in nails, hair, horns, and feathers. It exists in both α (alpha) and β (beta) forms, with over 30 variants that are tissue-specific. Keratin is classified into type I (acidic, negatively charged) and type II (basic, positively charged) proteins.
α-Keratin: Forms helical structures with a pitch of 5.1 Å (distinct from the standard α-helix pitch of 5.4 Å). This altered pitch allows for closely associated pairs of helices, each consisting of a type I and type II protein.
Coiled-Coil Structure: Keratin helices form left-handed coils (coiled-coil) with a 7-residue pseudo-repeat. The helical wheel model shows that a and d residues are nonpolar and face the same side, creating a hydrophobic strip that aligns between two helices.
Assembly: Keratin assembles hierarchically: dimer → protofilament → microfibril → macrofibril → hair.
Cysteine Content: α-Keratin is rich in cysteine, forming disulfide bonds. Hard keratin has high cysteine content, while soft keratin has lower cysteine content. Perms chemically reduce disulfide bonds (R-S---S-R) to 2R-SH, affecting hair structure.
Mechanical Properties: Keratin helices are stretchy and can elongate. β-Keratin, found in feathers and nails, is more rigid and stiff.
Disease Associations: Keratin-related diseases include epidermolysis bullosa simplex and epidermolytic hyperkeratosis, which involve loss of mechanical integrity in the skin.
Silk Fibroin
Structure and Function
Silk fibroin is a structural protein found in spider and insect webs, cocoons, nests, and egg sacks. It forms an almost fully extended β-pleated sheet, which is strong but cannot be stretched.
Composition: Silk is composed of fibroin and sericin. Sericin is an amorphous, gummy protein that can be removed by boiling water, leaving behind fibroin.
β-Sheet Structure: The primary sequence is (-Gly-Ser-Gly-Ala-Gly-Ala-)N. Alanine residues from one sheet interdigitate with alanine from another sheet, contributing to the strength of silk fibers.
Physical Properties: Silk fibers are strong when extended but cannot be stretched due to the fully extended sheet form of fibroin.
Species Variation: Silks from different species have different interdigitating groups, resulting in varying physical properties.
Example: Spiders can support their weight on web material due to the strength of silk fibroin.
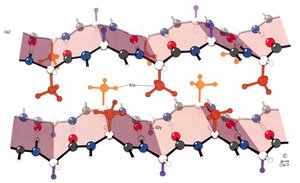
Collagen
Structure and Function
Collagen is the most abundant protein in mammals, providing structural support in bones, teeth, cartilage, tendons, ligaments, blood vessels, and skin. It is strong, flexible, and stretchy, with several types (e.g., Type I, II, III) differing in tissue distribution and function.
Triple Helical Structure: Collagen consists of three left-handed helices that combine to form a right-handed triple helix. The primary sequence is Gly-X-Y, where X is often proline and Y is often hydroxyproline.
Amino Acid Composition: Collagen is rich in glycine (1/3 of residues), 15-30% hydroxyproline, and some hydroxylysine. Hydroxyproline stabilizes the triple helix via hydrogen bonding.
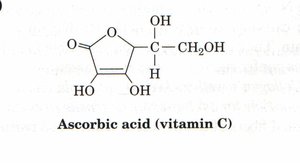
Vitamin C Requirement: Vitamin C (ascorbic acid) is required for the formation of hydroxyproline. Without prolyl hydroxylase (which requires vitamin C), collagen denatures at lower temperatures, leading to diseases such as scurvy.
Crosslinking: Collagen is covalently crosslinked, a process requiring lysine oxidase. Inhibition of lysine oxidase (e.g., by β-aminopropionitrile from sweet pea) leads to diseases such as lathyrism, osteogenesis imperfecta, Ehlers-Danlos syndrome, and osteoarthritis.
Organization: Collagen helices are organized into fibrils, with hydrophobic repulsion and possible Van der Waals attraction driving fibril formation. Collagen contains 0.4–12% carbohydrate (linked sugars).
Example: Scurvy results from vitamin C deficiency, causing skin lesions, broken blood vessels, poor wound healing, and loss of teeth.
Summary Table: Structural Proteins Comparison
Protein | Structure | Main Amino Acids | Function | Associated Diseases |
|---|---|---|---|---|
Keratin | α-helix (coiled-coil), β-sheet | Cysteine, hydrophobic residues | Hair, nails, feathers, skin | Epidermolysis bullosa simplex, hyperkeratosis |
Silk Fibroin | β-pleated sheet | Glycine, alanine, serine | Webs, cocoons, fibers | None specific |
Collagen | Triple helix | Glycine, proline, hydroxyproline | Bones, skin, cartilage, vessels | Scurvy, osteogenesis imperfecta, Ehlers-Danlos syndrome |
Key Equations
Collagen Triple Helix Pitch:
Keratin Helical Pitch:
Disulfide Bond Reduction:
Additional info:
Keratin, silk fibroin, and collagen are classic examples of proteins whose function is determined by their secondary structure.
Collagen's stability is highly dependent on post-translational modifications, especially hydroxylation of proline and lysine residues.
Vitamin C deficiency impairs collagen synthesis, leading to connective tissue disorders.
