 Back
BackStudy Guide: Nucleotides and Nucleic Acids (BCH20002 – Introduction to Biochemistry)
Study Guide - Smart Notes

Q1. The difference between a ribonucleotide and a deoxyribonucleotide is:
Background
Topic: Structure of Nucleotides
This question tests your understanding of the structural differences between ribonucleotides (RNA building blocks) and deoxyribonucleotides (DNA building blocks).
Key Terms and Concepts:
Ribonucleotide: Contains a ribose sugar with hydroxyl (–OH) groups at both the 2' and 3' carbons.
Deoxyribonucleotide: Contains a deoxyribose sugar, which lacks the –OH group at the 2' carbon (has –H instead).
Carbon Numbering: The carbons in the sugar ring are numbered 1' to 5'.
Step-by-Step Guidance
Recall the structure of ribose and deoxyribose sugars. Identify which carbon (2' or 3') differs between the two.
Review the functional groups attached to the 2' and 3' carbons in both sugars.
Match the correct description to the difference between ribonucleotides and deoxyribonucleotides.
Try solving on your own before revealing the answer!
Q2. Which of the base compositions for double-stranded DNA are possible?
Background
Topic: Chargaff's Rules and DNA Base Pairing
This question tests your understanding of the base composition rules for double-stranded DNA, specifically the relationships between the percentages of adenine (A), thymine (T), guanine (G), and cytosine (C).
Key Terms and Concepts:
Chargaff's Rules: In double-stranded DNA, %A = %T and %G = %C.
Base Pairing: A pairs with T, G pairs with C.
Uracil (U): Not found in DNA, only in RNA.
Step-by-Step Guidance
For each option, check if %A = %T and %G = %C.
Check if the total percentage of all bases adds up to 100%.
Eliminate any option that includes uracil (U) or violates Chargaff's rules.
Try solving on your own before revealing the answer!
Q3. Nucleotides and their derivatives can serve as:
Background
Topic: Functions of Nucleotides
This question tests your knowledge of the various biological roles of nucleotides beyond their function as building blocks of nucleic acids.
Key Terms and Concepts:
Metabolic Energy Carriers: e.g., ATP, GTP
Enzyme Cofactors: e.g., NAD+, FAD
Intracellular Signals: e.g., cAMP
Precursors for Nucleic Acid Synthesis
Step-by-Step Guidance
Recall the main functions of nucleotides in the cell.
Consider examples of nucleotides in each role listed in the options.
Determine if all the listed functions are correct or if only some are.
Try solving on your own before revealing the answer!
Q4. The phosphodiester bonds that link adjacent nucleotides in both RNA and DNA:
Background
Topic: Nucleic Acid Structure
This question tests your understanding of how nucleotides are joined together in nucleic acids and the properties of the phosphodiester bond.
Key Terms and Concepts:
Phosphodiester Bond: Covalent bond linking the 3' hydroxyl of one nucleotide to the 5' phosphate of the next.
Susceptibility to Hydrolysis: RNA is more susceptible to alkaline hydrolysis than DNA.
Charge at Neutral pH: Phosphodiester backbone is negatively charged.
Step-by-Step Guidance
Recall the structure of the nucleic acid backbone and how nucleotides are linked.
Review the properties of the phosphodiester bond, including its charge and susceptibility to hydrolysis.
Identify which statement correctly describes the phosphodiester linkage.
Try solving on your own before revealing the answer!
Q5. B-form DNA in vivo is a _____-handed helix, A-form is _____-handed and Z-form is _____-handed.
Background
Topic: DNA Helical Forms
This question tests your knowledge of the handedness (right or left) of the three main forms of DNA: B-form, A-form, and Z-form.
Key Terms and Concepts:
B-form DNA: The most common form in cells, right-handed helix.
A-form DNA: Right-handed helix, found in dehydrated samples and RNA-DNA hybrids.
Z-form DNA: Left-handed helix, occurs in certain sequences.
Step-by-Step Guidance
Recall the handedness of each DNA form (B, A, Z).
Match the correct handedness to each form in the options.
Try solving on your own before revealing the answer!
Q6. When double-stranded DNA is heated at neutral pH, which change does NOT occur?
Background
Topic: DNA Denaturation
This question tests your understanding of what happens to DNA when it is heated, specifically which bonds are broken and which are not.
Key Terms and Concepts:
Denaturation: Separation of DNA strands due to breaking of hydrogen bonds.
N-glycosidic Bond: Covalent bond between base and sugar, not broken during denaturation.
Hydrogen Bonds: Broken between base pairs (A-T, G-C).
Step-by-Step Guidance
List the types of bonds present in DNA (hydrogen bonds, phosphodiester bonds, N-glycosidic bonds).
Recall which bonds are disrupted during heating (denaturation) and which remain intact.
Identify the change that does NOT occur during DNA heating.
Try solving on your own before revealing the answer!
Q7. In nucleotides and nucleic acids, syn and anti conformations relate to:
Background
Topic: Nucleotide Structure and Conformation
This question tests your understanding of the conformational flexibility of nucleotides, specifically the orientation of the base relative to the sugar.
Key Terms and Concepts:
Syn and Anti Conformations: Refer to the rotation of the base around the glycosidic bond (sugar-base bond).
Phosphodiester Bond: Links nucleotides, not involved in syn/anti conformations.
Step-by-Step Guidance
Recall what the syn and anti conformations describe in nucleotides.
Identify which bond allows for this rotation (sugar-base bond).
Match the correct answer to the description of syn and anti conformations.
Try solving on your own before revealing the answer!
Q8. In the Watson-Crick model of DNA structure (now called B-form DNA):
Background
Topic: DNA Double Helix Structure
This question tests your knowledge of the key features of the Watson-Crick (B-form) DNA model, including base pairing and helix structure.
Key Terms and Concepts:
Base Pairing: A-T pairs (2 hydrogen bonds), G-C pairs (3 hydrogen bonds).
Helix Structure: Bases are on the inside, sugar-phosphate backbone on the outside.
Strand Orientation: Strands are antiparallel.
Step-by-Step Guidance
Recall the number of hydrogen bonds between A-T and G-C pairs.
Review the arrangement of bases and backbone in the helix.
Identify which statement accurately describes the Watson-Crick model.
Try solving on your own before revealing the answer!
Q9. What are the components of a nucleoside?
Background
Topic: Nucleoside Structure
This question tests your understanding of the basic components that make up a nucleoside.
Key Terms and Concepts:
Nucleoside: Consists of a nitrogenous base linked to a pentose sugar (ribose or deoxyribose).
No Phosphate Group: Nucleosides do not contain phosphate groups (those are in nucleotides).
Step-by-Step Guidance
Identify the two main components of a nucleoside.
Recall the difference between a nucleoside and a nucleotide.
Try solving on your own before revealing the answer!
Q10. Number the carbon atoms in the furanose ring according to the correct convention.
Background
Topic: Sugar Structure in Nucleotides
This question tests your ability to correctly number the carbons in the pentose (furanose) ring of nucleotides.
Key Terms and Concepts:
Furanose Ring: Five-membered ring structure of ribose or deoxyribose.
Numbering Convention: Carbons are numbered 1' to 5', starting at the anomeric carbon (attached to the base).
Step-by-Step Guidance
Identify the anomeric carbon (1') attached to the nitrogenous base.
Number the carbons sequentially around the ring to the 5' carbon (attached to the CH2OH group).
Try solving on your own before revealing the answer!
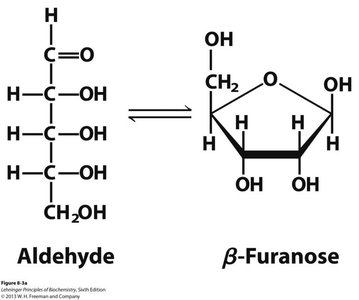
Q11. Which of these sugars is a component of DNA?
Background
Topic: DNA Sugar Component
This question tests your ability to distinguish between ribose and deoxyribose sugars and identify which is found in DNA.
Key Terms and Concepts:
Deoxyribose: The sugar in DNA, lacks an –OH group at the 2' carbon.
Ribose: The sugar in RNA, has –OH groups at both 2' and 3' carbons.
Step-by-Step Guidance
Examine the structures and identify which sugar lacks the 2' –OH group.
Recall that deoxyribose is the sugar in DNA.
Try solving on your own before revealing the answer!
Q12. Classify each of the nitrogenous bases below as either purines or pyrimidines.
Background
Topic: Nitrogenous Bases
This question tests your ability to distinguish between purine and pyrimidine bases based on their structures.
Key Terms and Concepts:
Purines: Double-ring structures (adenine, guanine).
Pyrimidines: Single-ring structures (cytosine, thymine, uracil).
Step-by-Step Guidance
Identify the number of rings in each base structure.
Classify each base as a purine (double ring) or pyrimidine (single ring).
Try solving on your own before revealing the answer!
Q13. Which of the bases below is only found in RNA?
Background
Topic: RNA vs. DNA Bases
This question tests your knowledge of which nitrogenous base is unique to RNA and not found in DNA.
Key Terms and Concepts:
Uracil (U): Found only in RNA, replaces thymine (T) in DNA.
Thymine (T): Found only in DNA.
Step-by-Step Guidance
Identify the structures of the bases provided.
Recall which base is unique to RNA.
Try solving on your own before revealing the answer!
Q14. Provide the full name of the nucleotide below. Start by listing the components:
Background
Topic: Nucleotide Nomenclature
This question tests your ability to identify and name a nucleotide based on its components: base, sugar, and phosphate group(s).
Key Terms and Concepts:
Nucleotide: Composed of a nitrogenous base, a pentose sugar, and one or more phosphate groups.
Naming: The name is based on the base, the sugar (ribose or deoxyribose), and the number of phosphates (mono-, di-, tri-).
Step-by-Step Guidance
Identify the nitrogenous base in the structure.
Determine if the sugar is ribose or deoxyribose.
Count the number of phosphate groups attached.
Combine these features to write the full name of the nucleotide.
Try solving on your own before revealing the answer!
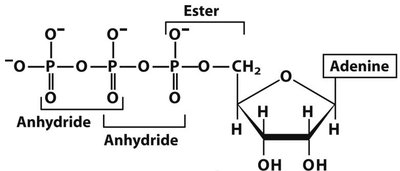
Q15. Provide the full name of the nucleotide below. Start by listing the components:
Background
Topic: Nucleotide Nomenclature
This question is similar to the previous one, but with a different base or number of phosphates.
Key Terms and Concepts:
Nucleotide: Composed of a nitrogenous base, a pentose sugar, and one or more phosphate groups.
Naming: The name is based on the base, the sugar (ribose or deoxyribose), and the number of phosphates (mono-, di-, tri-).
Step-by-Step Guidance
Identify the nitrogenous base in the structure.
Determine if the sugar is ribose or deoxyribose.
Count the number of phosphate groups attached.
Combine these features to write the full name of the nucleotide.
Try solving on your own before revealing the answer!
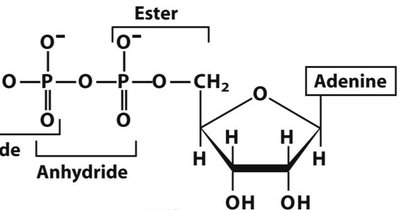
Q16. Write down the primary structure (in short-hand form) of the polynucleotide to the right.
Background
Topic: Polynucleotide Structure
This question tests your ability to interpret a polynucleotide diagram and write its sequence in short-hand notation (e.g., 5'-AUGC-3').
Key Terms and Concepts:
Primary Structure: The linear sequence of nucleotides in a nucleic acid.
Short-hand Notation: Uses single-letter abbreviations for bases (A, U, G, C for RNA; A, T, G, C for DNA).
Step-by-Step Guidance
Identify the sequence of bases from the diagram, starting from the 5' end.
Write the sequence using the correct abbreviations and orientation (5' to 3').
Try solving on your own before revealing the answer!
Q17. What is the primary reason for the relative instability of RNA, in comparison to DNA?
Background
Topic: Chemical Stability of Nucleic Acids
This question tests your understanding of why RNA is more chemically unstable than DNA.
Key Terms and Concepts:
2' Hydroxyl Group: RNA has a –OH at the 2' position, making it more susceptible to hydrolysis.
Deoxyribose: DNA lacks the 2' –OH, making it more stable.
Step-by-Step Guidance
Recall the structural difference between ribose (RNA) and deoxyribose (DNA).
Explain how the presence of the 2' –OH group in RNA leads to increased susceptibility to hydrolysis.
Try solving on your own before revealing the answer!
