Back
BackSynthesis and Function of Lung Surfactant and Membrane Lipids
Study Guide - Smart Notes

Lung Surfactant: Composition & Function
Role of Lung Surfactant in Respiration
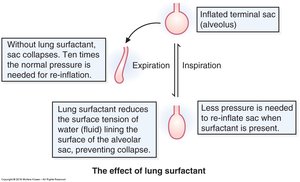
Lung surfactant is a complex mixture of lipids and proteins that lines the alveoli, playing a critical role in respiratory physiology. Its primary function is to reduce alveolar surface tension, thereby preventing alveolar collapse and improving lung compliance. This is especially vital for neonatal respiration, as premature infants often lack sufficient surfactant, leading to respiratory distress syndrome (RDS).
Reduces surface tension: Surfactant lowers the surface tension of the fluid lining the alveoli, making it easier for the lungs to expand during inspiration.
Prevents alveolar collapse: By stabilizing alveoli, surfactant ensures efficient gas exchange.
Improves lung compliance: Surfactant allows the lungs to inflate with less effort.
Clinical relevance: Deficiency in surfactant, particularly DPPC, is a major cause of neonatal RDS.
Lung Surfactant: Major Constituents
Key Lipids in Surfactant
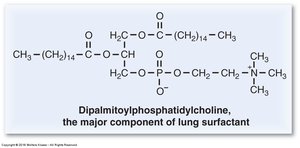
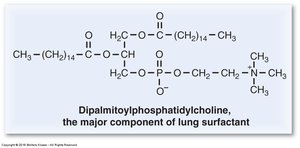
The surfactant monolayer consists mainly of phospholipids and proteins, with dipalmitoyl phosphatidylcholine (DPPC) as the predominant component. Other important constituents include phosphatidylglycerol and cholesterol. The monolayer sits at the air–liquid interface of the alveoli, facilitating its function.
DPPC: Two saturated palmitate chains (16:0) allow tight packing and maximal reduction of surface tension.
Phosphatidylglycerol: Contributes to surfactant stability and function.
Cholesterol: Modulates fluidity and stability of the surfactant layer.
Dipalmitoyl Phosphatidylcholine (DPPC)
Structure and Clinical Importance
DPPC is the most abundant phospholipid in lung surfactant. Its structure features two saturated palmitate chains, which enable tight molecular packing and maximal surface tension reduction. Premature infants often lack DPPC, resulting in respiratory distress syndrome.
Structure: Glycerol backbone, two palmitate chains, phosphate-choline head group.
Function: Maximally lowers surface tension in alveoli.
Clinical: Deficiency leads to neonatal RDS.
Phosphatidylcholine & Phosphatidylglycerol
Phospholipid Structure and Role in Fetal Lung Maturity
Phosphatidylcholine and phosphatidylglycerol are key phospholipids in surfactant. Phosphatidylcholine is crucial for fetal lung maturity, and its concentration increases late in pregnancy. Phosphatidylglycerol also contributes to surfactant function.
Phosphatidylcholine: Glycerol backbone, two fatty acids, phosphate-choline head group.
Phosphatidylglycerol: Similar backbone, glycerol head group.
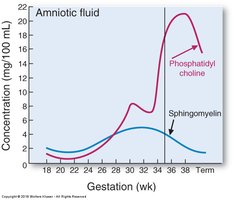
Fetal Lung Maturity: Lecithin : Sphingomyelin Ratio
Biochemical Marker for Lung Development
The lecithin (phosphatidylcholine) : sphingomyelin ratio in amniotic fluid is a historical marker for fetal lung maturity. A ratio of 2 or higher indicates mature lungs, while a ratio below 1.5 suggests risk of neonatal respiratory distress syndrome (RDS). Lecithin rises late in pregnancy, while sphingomyelin remains constant.
Measured in amniotic fluid: Used to assess fetal lung maturity.
Interpretation: Ratio ≥ 2 = mature lungs; < 1.5 = risk of RDS.
Biochemical basis: Lecithin increases late in gestation; sphingomyelin stays constant.
Formation of Phosphatidate
Phosphatidate as a Precursor of Glycerophospholipids
Phosphatidate is a key intermediate in the synthesis of membrane phospholipids. It is formed from diacylglycerol and phosphate, and serves as a precursor for various glycerophospholipids. The synthesis pathway begins with glucose, which is converted to dihydroxyacetone phosphate (DHAP), then to glycerol-3-phosphate, and finally to phosphatidate via addition of two fatty acyl-CoA molecules. This process occurs in the endoplasmic reticulum (ER) membrane.
Phosphatidate: Diacylglycerol + phosphate.
Synthesis: Glucose → DHAP → glycerol-3-P → phosphatidate.
Location: ER membrane.
CTP & Head Group Activation
Role of Cytidine Triphosphate (CTP) in Phospholipid Synthesis
CTP is essential for activating head groups during phospholipid synthesis. It forms CDP-choline and CDP-ethanolamine, which are required for the biosynthesis of phosphatidylcholine and phosphatidylethanolamine, respectively.
CTP: Cytidine triphosphate, activates head groups.
CDP-choline: Precursor for phosphatidylcholine.
CDP-ethanolamine: Precursor for phosphatidylethanolamine.
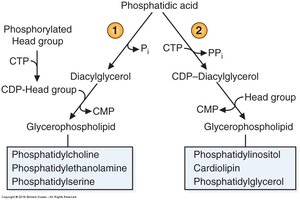
Two Strategies to Make Phospholipids
Pathways for Glycerophospholipid Synthesis
Phospholipids are synthesized via two main strategies: activation of the head group or activation of diacylglycerol. The first strategy involves forming CDP-head groups (e.g., CDP-choline, CDP-ethanolamine) for phosphatidylcholine, phosphatidylethanolamine, and phosphatidylserine. The second strategy activates diacylglycerol (CDP-DAG) for phosphatidylinositol, cardiolipin, and phosphatidylglycerol.
Strategy 1: CDP-head group activation for PC, PE, PS.
Strategy 2: CDP-diacylglycerol activation for PI, cardiolipin, PG.
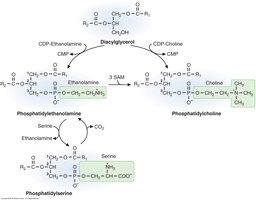
Interconversions of Phospholipids
Base-Exchange Reactions and Methylation
Phospholipids can be interconverted through base-exchange reactions and methylation. Phosphatidylethanolamine (PE) can be converted to phosphatidylserine (PS) and vice versa. PE can also be methylated to form phosphatidylcholine (PC), a process that consumes S-adenosylmethionine (SAM).
PE ↔ PS: Base-exchange reactions.
PE → PC: Three methylations using SAM as donor.
Largest SAM-consuming reaction: PE to PC methylation.
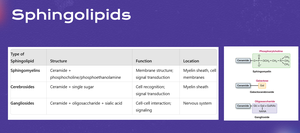
Glycosphingolipids Overview
Structure and Function of Glycosphingolipids
Glycosphingolipids are sphingolipids containing a ceramide backbone and a carbohydrate (glycan) head group. They are important for cell recognition, signaling, and membrane structure.
Ceramide backbone: Fundamental to sphingolipid structure.
Glycan head group: Provides specificity and function.
Ceramide Synthesis
Pathway and Biological Importance
Ceramide is synthesized from serine and palmitoyl-CoA, forming the backbone via PLP (vitamin B6)-dependent reaction. Reduction by NADPH produces dihydrosphingosine, which is acylated to dihydroceramide and then oxidized by FAD to ceramide. Ceramide is a precursor for sphingomyelin and glycosphingolipids.
Serine: Provides head and first carbons.
Palmitate: Provides long hydrocarbon chain.
Ceramide: Precursor for sphingomyelin and glycosphingolipids.

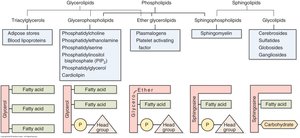
Sphingomyelin & Glycolipids
Types and Synthesis of Sphingolipids
Sphingomyelin is formed by combining ceramide with phosphocholine, while cerebrosides and gangliosides are synthesized by adding glucose or oligosaccharides to ceramide. These molecules are essential for membrane structure and cell signaling.
Sphingomyelin: Ceramide + phosphocholine.
Cerebroside: Ceramide + glucose.
Ganglioside: Cerebroside + oligosaccharide.
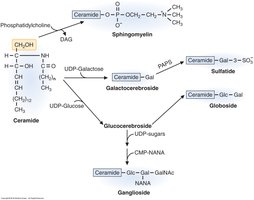
Synthesis of Cerebrosides and Gangliosides
Enzymatic Pathways and Sugar Donors
Cerebrosides are synthesized in the Golgi apparatus by adding UDP-sugar to ceramide via glycosyltransferase. The sugar is attached to the C-1 hydroxyl of ceramide, forming a glycosidic bond. UDP-glucose yields glucocerebroside, while UDP-galactose yields galactocerebroside. All glycosphingolipid sugars are derived from activated nucleotide sugars.
UDP-glucose: Donor for glucocerebroside.
UDP-galactose: Donor for galactocerebroside.
Enzyme: Glycosyltransferase.
ABO Blood Group Antigens & Transfusion Compatibility
Structure and Enzymatic Basis of Blood Group Antigens
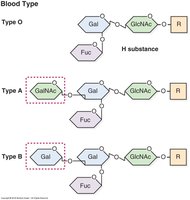
ABO blood group antigens are oligosaccharide structures present on red blood cell membrane glycoproteins and glycolipids. All types are built on the H antigen precursor. Type O has only the H antigen, type A adds N-acetylgalactosamine (GalNAc), type B adds galactose, and type AB has both sugars. The specific enzymes encoded by the A, B, and O alleles determine the antigenic structure.
Type O: H antigen only.
Type A: H antigen + GalNAc.
Type B: H antigen + galactose.
Type AB: Both A and B sugars present.
Enzymes: A allele adds GalNAc, B allele adds galactose, O allele is inactive.