 Back
BackThe Three-Dimensional Structure of Proteins: Levels, Elements, and Structural Principles
Study Guide - Smart Notes

The Four Levels of Protein Structure
Overview of Protein Structural Hierarchy
Proteins exhibit a hierarchical organization, with four distinct structural levels: primary, secondary, tertiary, and quaternary. Each level contributes to the overall shape and function of the protein.
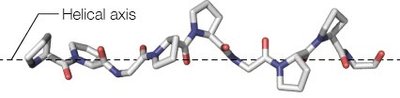
Primary Structure: The linear sequence of amino acids in a polypeptide chain, determined by covalent peptide bonds.
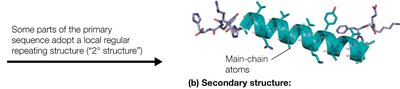
Secondary Structure: Local regions of the polypeptide chain adopt regular, repeating structures stabilized by hydrogen bonds, such as α-helices and β-sheets.
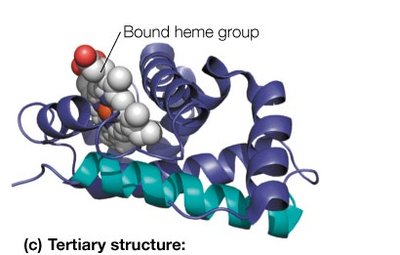
Tertiary Structure: The overall three-dimensional folding of a single polypeptide chain, resulting from interactions among secondary structure elements and side chains.
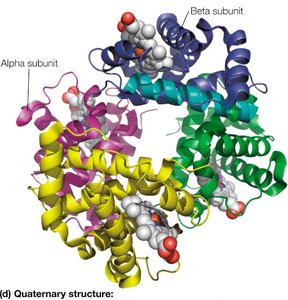
Quaternary Structure: The spatial arrangement of multiple polypeptide chains (subunits) in a multisubunit complex.

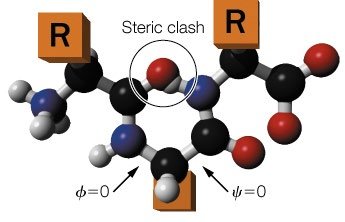
Bond Rotations and Peptide Backbone
Peptide Bond Geometry and Rotational Freedom
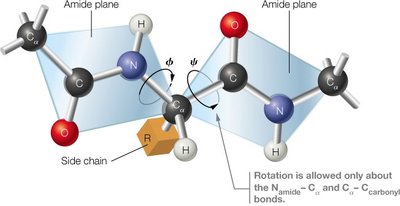
The peptide bond has partial double-bond character, making it planar and restricting rotation. Free rotation is allowed only about the bonds adjacent to the α-carbon (Cα): the N–Cα (phi, φ) and Cα–C (psi, ψ) bonds. The allowed rotations are limited by steric interactions between atoms.
Amide Plane: The peptide bond is rigid and planar.
Rotational Angles: φ (phi) and ψ (psi) describe the backbone conformation.
Steric Hindrance: Some combinations of φ and ψ are disallowed due to atomic clashes.
Secondary Structure Elements
α-Helix
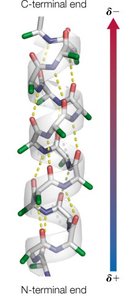
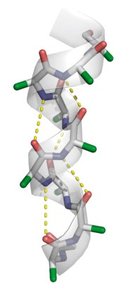
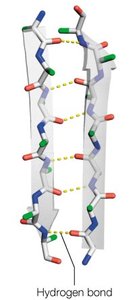
The α-helix is a right-handed helical structure stabilized by hydrogen bonds between the carbonyl oxygen of residue i and the amide hydrogen of residue i+4. It has distinct structural parameters and amphipathic properties.
Residues per turn: 3.6
Rise per residue: 1.5 Å
Pitch: 5.4 Å per turn
Hydrogen bonds: Nearly parallel to the helix axis
Amphipathic nature: Side chains radiate outward, often forming hydrophilic and hydrophobic faces
β-Sheet
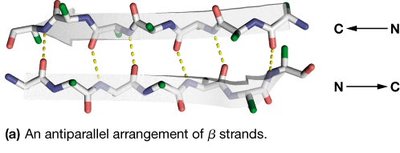
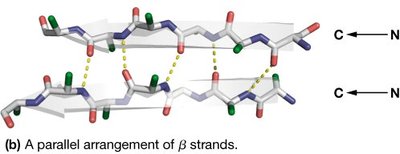
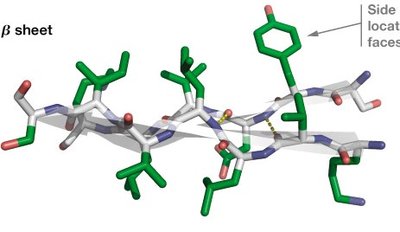
β-sheets are formed by two or more β-strands connected by hydrogen bonds. Strands can be parallel or antiparallel, with side chains alternating above and below the sheet plane.
Residues per turn: 2.0
Rise per residue: 3.2 Å (antiparallel), 3.4 Å (parallel)
Pitch: 6.4 Å (antiparallel), 6.8 Å (parallel)
Hydrogen bonds: Between adjacent strands
Amphipathic nature: Alternating side chain polarity
Other Secondary Structures
310-Helix: Right-handed helix, 3 residues per turn, less common.
Polyproline II Helix: Left-handed helix, prevalent in collagen, not stabilized by hydrogen bonds.
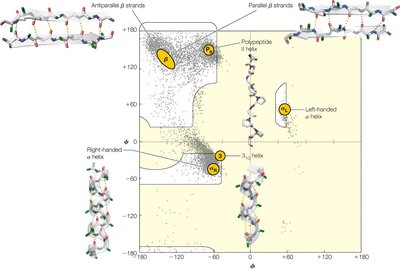
Ramachandran Plot and Steric Constraints
Ramachandran Plot Interpretation
The Ramachandran plot maps the allowed φ and ψ angles for amino acid residues, showing regions corresponding to α-helices, β-sheets, and other structures. Steric hindrance restricts the conformational space.
Allowed regions: Indicate stable secondary structures
Glycine: More allowed angles due to small side chain
Proline: Restricted angles due to cyclic structure
Fibrous and Globular Proteins
Structural and Functional Differences
Proteins are classified as fibrous or globular based on their tertiary structure and function. Fibrous proteins provide structural support, while globular proteins are involved in chemical reactions and are generally soluble.
Fibrous Proteins: Elongated, regular amino acid sequence, insoluble, structural role (e.g., keratin, collagen, fibroin)
Globular Proteins: Compact, irregular sequence, soluble, functional diversity

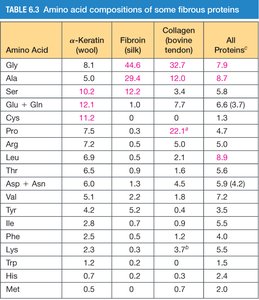
Amino Acid Composition of Fibrous Proteins
Different fibrous proteins have characteristic amino acid compositions, which influence their mechanical properties and structure.
Amino Acid | α-Keratin (wool) | Fibroin (silk) | Collagen (bovine tendon) | All Proteins |
|---|---|---|---|---|
Gly | 8.1 | 44.6 | 32.7 | 7.9 |
Ala | 5.0 | 29.4 | 12.0 | 8.7 |
Ser | 10.2 | 12.2 | 3.4 | 6.8 |
Glu + Gln | 12.1 | 3.0 | 7.7 | 6.3 |
Cys | 11.2 | 0.0 | 0.2 | 2.3 |
Pro | 7.5 | 0.0 | 22.7 | 4.7 |
Arg | 7.2 | 0.0 | 5.0 | 5.0 |
Leu | 6.6 | 0.9 | 1.6 | 8.6 |
Thr | 6.6 | 0.9 | 1.6 | 5.2 |
Asp + Asn | 5.4 | 0.2 | 0.7 | 5.8 |
Val | 5.1 | 2.2 | 1.7 | 7.5 |
Tyr | 4.2 | 5.2 | 0.5 | 3.5 |
Phe | 2.5 | 3.0 | 1.2 | 4.0 |
Lys | 1.7 | 0.2 | 0.2 | 5.9 |
Trp | 1.2 | 0.0 | 0.0 | 1.1 |
Met | 0.5 | 0.0 | 0.7 | 2.0 |
Fibrous Proteins: α-Keratin and Modifications
α-Keratin Structure and Function
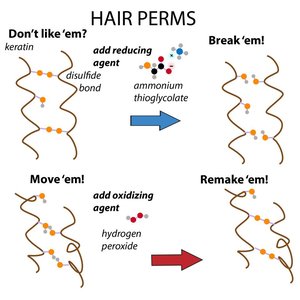
α-Keratin consists of two α-helices coiled around each other, forming a left-handed coiled-coil. Hydrophobic residues repeat every 3–4 positions, maximizing hydrophobic contact. Disulfide bonds between cysteine residues harden the structure, with more bonds in nails than in hair.
Coiled-coil: Left-handed, hydrophobic interface
Disulfide bonds: Increase hardness and stability
Hair perms: Chemical agents break and reform disulfide bonds to reshape hair


Summary Table: Parameters of Secondary Structures
Comparison of Structural Parameters
The following table summarizes the key parameters that define common secondary structures in proteins.
Structure Type | Residues per turn | Rise per residue (Å) | Pitch (Å/turn) |
|---|---|---|---|
β-strand (antiparallel) | 2.0 | 3.2 | 6.4 |
β-strand (parallel) | 2.0 | 3.4 | 6.8 |
α-helix | 3.6 | 1.5 | 5.4 |
310 helix | 3.0 | 2.0 | 6.0 |
Polypeptide II helix | 3.0 | 3.1 | 9.3 |
*Additional info: Tables and diagrams are recreated and expanded for clarity and completeness based on textbook conventions.*
