 Back
BackAn Introduction to Metabolism (Bio 101, Chapter 8) – Study Notes
Study Guide - Smart Notes

An Introduction to Metabolism
Overview of Metabolism
Metabolism refers to the sum of all chemical reactions that occur within a living organism. These reactions are essential for maintaining life, enabling cells to grow, reproduce, maintain their structures, and respond to environmental changes. The cell is often described as a chemical factory due to its ability to carry out a vast array of chemical transformations in a highly regulated manner.
Metabolism: The totality of an organism’s chemical reactions.
Metabolic Pathway: A series of chemical reactions that either build a complex molecule (anabolic pathway) or break down a complex molecule into simpler compounds (catabolic pathway).
Regulation: Metabolic pathways are tightly regulated in both eukaryotic and prokaryotic cells to ensure efficiency and adaptability.
Anabolism and Catabolism
Metabolic pathways are classified as either anabolic or catabolic based on their function:
Anabolic Pathways: Consume energy to build complex molecules from simpler ones (e.g., synthesis of proteins from amino acids).
Catabolic Pathways: Release energy by breaking down complex molecules into simpler compounds (e.g., cellular respiration).
Energy and Thermodynamics in Biology
Forms of Energy
Energy is the capacity to cause change or do work. In biological systems, energy exists in various forms:
Kinetic Energy: Energy of motion (e.g., movement of molecules).
Potential Energy: Stored energy due to position or structure (e.g., chemical bonds in glucose).

Thermodynamic Laws
Biological systems obey the laws of thermodynamics, which govern energy transformations:
First Law of Thermodynamics (Conservation of Energy): Energy cannot be created or destroyed, only transferred or transformed.
Second Law of Thermodynamics: Every energy transfer or transformation increases the entropy (disorder) of the universe.

Entropy and Biological Order
Although living organisms maintain order and structure, they increase the overall entropy of their surroundings through metabolic processes. For example, when organisms convert chemical energy from food into kinetic energy for movement, some energy is lost as heat, increasing entropy.
Free Energy and Biological Reactions
Free Energy (G)
Free energy is the portion of a system’s energy that can perform work when temperature and pressure are uniform. The change in free energy () determines whether a process is spontaneous.
Equation:
Spontaneous Reactions: Occur without input of energy ().
Nonspontaneous Reactions: Require energy input ().
Exergonic Reactions: Release free energy (catabolic).
Endergonic Reactions: Absorb free energy (anabolic).
ATP: The Energy Currency of the Cell
Structure and Function of ATP
Adenosine triphosphate (ATP) is the primary energy carrier in cells. It powers cellular work by transferring a phosphate group to other molecules (phosphorylation), making them more reactive.
ATP Hydrolysis: The breakdown of ATP into ADP and inorganic phosphate releases energy.
ATP Regeneration: ATP is regenerated from ADP and phosphate through cellular respiration.
Enzymes and Metabolic Regulation
Nature and Function of Enzymes
Enzymes are biological catalysts that speed up chemical reactions by lowering the activation energy required. They are highly specific for their substrates and are not consumed in the reaction.
Active Site: The region on the enzyme where the substrate binds and the reaction occurs.
Enzyme-Substrate Complex: Temporary association between enzyme and substrate during the reaction.
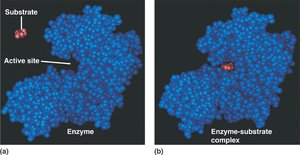
Mechanism of Enzyme Action
Enzymes facilitate reactions through the following steps:
Substrate binds to the enzyme’s active site, forming the enzyme-substrate complex.
Chemical reaction occurs, converting substrate into product(s).
Product(s) are released, and the enzyme is free to catalyze another reaction.
Factors Affecting Enzyme Activity
Enzyme activity is influenced by several environmental factors:
Temperature: Each enzyme has an optimal temperature for activity.
pH: Each enzyme has an optimal pH range.
Cofactors: Non-protein helpers required for enzyme function. These can be inorganic (e.g., iron, copper, zinc) or organic (coenzymes such as NAD+, FAD).
Enzyme Inhibition
Enzyme activity can be inhibited by molecules that reduce their activity. Inhibitors can be competitive (binding to the active site) or noncompetitive (binding elsewhere and changing enzyme shape).
Summary Table: Key Concepts in Metabolism
Concept | Definition | Example |
|---|---|---|
Metabolism | Sum of all chemical reactions in an organism | Cellular respiration, photosynthesis |
Anabolic Pathway | Builds complex molecules, consumes energy | Protein synthesis |
Catabolic Pathway | Breaks down molecules, releases energy | Glycolysis |
ATP | Main energy currency of the cell | Muscle contraction |
Enzyme | Biological catalyst | Sucrase, pepsin |
