 Back
BackDNA and the Gene: Synthesis and Repair – Study Notes
Study Guide - Smart Notes

DNA and the Gene: Synthesis and Repair
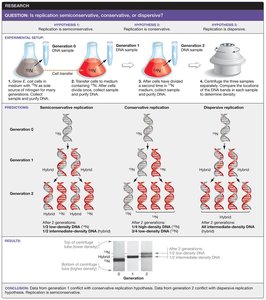
Testing Early Hypotheses about DNA Replication
Understanding how DNA replicates was a foundational question in molecular biology. Three main hypotheses were proposed:
Semiconservative replication: Each daughter DNA molecule consists of one parental (old) strand and one newly synthesized strand.
Conservative replication: The parental DNA molecule remains intact, and an entirely new molecule is synthesized.
Dispersive replication: Parental DNA is fragmented, and each daughter molecule contains interspersed segments of old and new DNA.
The Meselson–Stahl experiment provided evidence supporting the semiconservative model by using isotopic labeling and density gradient centrifugation to distinguish old and new DNA strands.
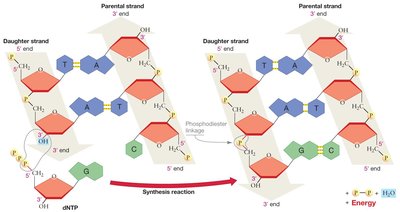
A Model for DNA Synthesis
DNA synthesis is catalyzed by the enzyme DNA polymerase, which adds nucleotides to the 3′ end of a growing DNA strand. DNA polymerases can only synthesize DNA in the 5′ → 3′ direction. The building blocks for DNA synthesis are deoxyribonucleoside triphosphates (dNTPs), which have high potential energy due to their three phosphate groups. This energy drives the formation of phosphodiester bonds between nucleotides.
Directionality: DNA polymerase adds nucleotides only to the 3′ end, so synthesis always proceeds 5′ → 3′.
Energy: Hydrolysis of dNTPs releases energy, making the reaction exergonic.
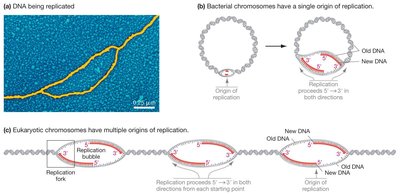
Origin and Direction of DNA Replication
DNA replication begins at specific locations called origins of replication. In bacteria, there is a single origin per chromosome, while eukaryotic chromosomes have multiple origins. Replication proceeds bidirectionally, forming replication bubbles with two replication forks moving in opposite directions.
Bacterial chromosomes: One origin, one replication bubble.
Eukaryotic chromosomes: Multiple origins, multiple replication bubbles.
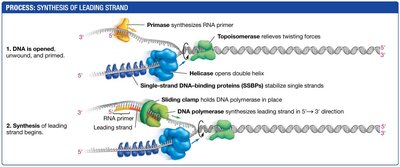
Opening and Stabilizing the DNA Helix
Several proteins are required to open and stabilize the DNA double helix during replication:
DNA helicase: Unwinds the double helix by breaking hydrogen bonds between strands.
Single-strand DNA-binding proteins (SSBPs): Bind to separated strands to prevent them from re-annealing.
Topoisomerase: Relieves tension ahead of the replication fork by cutting and rejoining DNA strands.
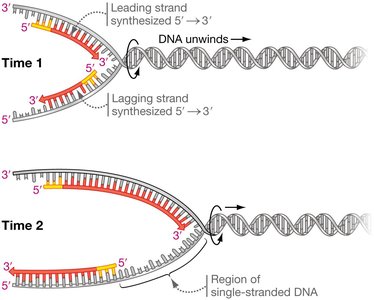
Synthesis of the Leading Strand
The leading strand is synthesized continuously toward the replication fork. DNA polymerase cannot initiate synthesis de novo; it requires a short RNA primer synthesized by primase (an RNA polymerase). DNA polymerase then extends the primer by adding dNTPs to the 3′ end.
Primer: Short RNA sequence base-paired to the template strand.
Continuous synthesis: Leading strand is synthesized in one continuous piece.
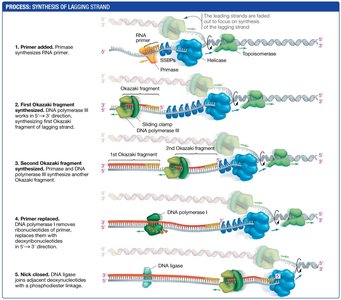
Synthesis of the Lagging Strand
The lagging strand is synthesized discontinuously, away from the replication fork, as short fragments called Okazaki fragments. Each fragment begins with an RNA primer. DNA polymerase synthesizes DNA until it reaches the next primer, and the fragments are later joined by DNA ligase.
Discontinuous replication: Multiple primers are required; synthesis occurs in short segments.
Okazaki fragments: Short DNA fragments synthesized on the lagging strand.
DNA ligase: Joins Okazaki fragments into a continuous strand.
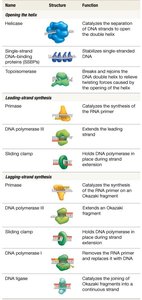
Proteins Required for DNA Synthesis in Bacteria
DNA replication involves a coordinated set of proteins, each with a specific function:
Protein | Structure | Function |
|---|---|---|
Helicase | Enzyme | Separates DNA strands |
SSBPs | Protein | Stabilizes single-stranded DNA |
Topoisomerase | Enzyme | Relieves twisting forces |
Primase | Enzyme | Synthesizes RNA primer |
DNA polymerase III | Enzyme | Extends leading/lagging strand |
Sliding clamp | Protein | Holds DNA polymerase in place |
DNA polymerase I | Enzyme | Removes RNA primer, replaces with DNA |
DNA ligase | Enzyme | Joins Okazaki fragments |
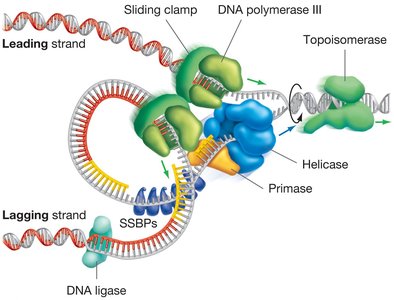
The Replisome: DNA-Synthesizing Machine
The replisome is a large, dynamic complex of enzymes and proteins responsible for DNA synthesis at the replication fork. It coordinates the activities of all proteins involved in replication, ensuring efficient and accurate DNA synthesis.
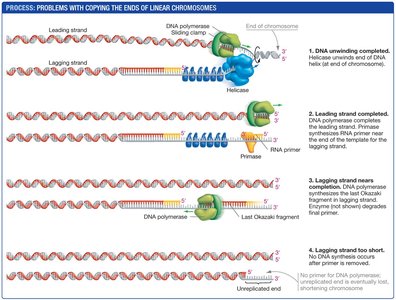
Replicating the Ends of Linear Chromosomes
Replication of linear eukaryotic chromosomes presents a unique challenge at the ends, known as telomeres. The lagging strand cannot be fully replicated, leading to progressive shortening of chromosomes with each cell division. Telomeres consist of repetitive, non-coding DNA sequences.
End replication problem: After removal of the final RNA primer, DNA polymerase cannot fill the gap, resulting in chromosome shortening.
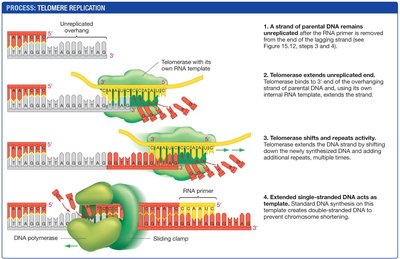
Telomerase and Telomere Replication
Telomerase is an enzyme that extends telomeres by adding repetitive DNA sequences, using an RNA template carried within the enzyme. This prevents chromosome shortening in cells where telomerase is active (e.g., gametes, stem cells).
Mechanism: Telomerase binds to the overhang, extends it, and allows normal DNA synthesis to complete the lagging strand.
Biological significance: Most somatic cells lack telomerase, leading to limited cell divisions. Cancer cells often reactivate telomerase, enabling unlimited division.
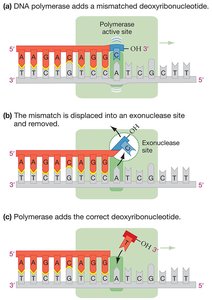
Correcting Mistakes in DNA Synthesis
DNA polymerase has high fidelity but occasionally incorporates incorrect bases. Several mechanisms ensure accuracy:
Proofreading: DNA polymerase detects and removes mismatched nucleotides using its exonuclease activity.
Mismatch repair: Specialized enzymes recognize and repair mismatches missed during replication.
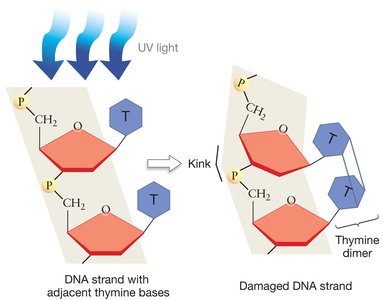
Repairing Damaged DNA
DNA is constantly exposed to damaging agents such as UV light, X-rays, and chemicals. One common form of damage is the formation of thymine dimers due to UV exposure, which creates kinks in the DNA and blocks replication.
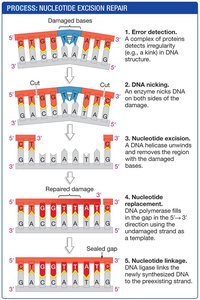
Nucleotide Excision Repair
The nucleotide excision repair system recognizes and removes damaged DNA segments. The process involves:
Detection of DNA damage (e.g., thymine dimers).
Removal of the damaged DNA segment.
Filling in the gap using the undamaged strand as a template.
Sealing the new DNA with DNA ligase.
Summary Table: Key Enzymes and Functions in DNA Replication and Repair
Enzyme/Protein | Function |
|---|---|
DNA polymerase | Synthesizes new DNA, proofreads |
Primase | Synthesizes RNA primers |
Helicase | Unwinds DNA helix |
SSBPs | Stabilize single-stranded DNA |
Topoisomerase | Relieves supercoiling |
DNA ligase | Joins DNA fragments |
Telomerase | Extends telomeres |
Exonuclease | Removes mismatched bases |
Nucleotide excision repair complex | Removes and replaces damaged DNA |
Key Equations:
Phosphodiester bond formation (generalized):
