Back
BackChemical Kinetics: Temperature, Rate, Mechanisms, and Catalysis
Study Guide - Smart Notes

Chemical Kinetics
Temperature and Rate
Chemical kinetics studies the speed of chemical reactions and the factors that affect it. One of the most important factors is temperature, which has a significant impact on reaction rates.
Temperature Effect: As temperature increases, the rate of most chemical reactions increases, often doubling for each 10°C rise.
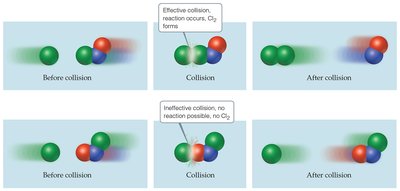
Collision Model: Molecules must collide with proper orientation and sufficient energy to react. Not all collisions result in product formation.
Concentration Effect: Higher reactant concentration leads to more frequent collisions and faster reactions.
Orientation Effect: Only collisions with the correct orientation lead to product formation; many collisions are ineffective.
Activation Energy (Ea) and Energy Diagrams
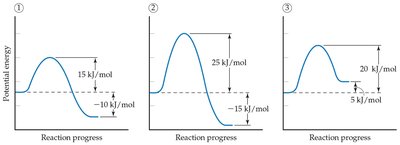
Activation energy is the minimum energy required for a reaction to occur. It acts as an energy barrier that reactants must overcome to form products.
Definition: Activation energy (Ea) is the threshold energy that molecules must possess to react.
Kinetic Energy: Only molecules with kinetic energy greater than Ea can react.
Energy Barrier: Higher activation energy generally means a slower reaction.
Energy Diagram: Shows the energy changes during a reaction, including the formation of an activated complex.
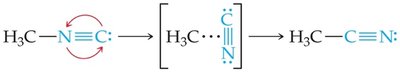
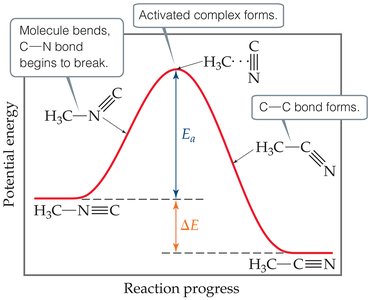
Effect of Temperature on Molecular Energy Distribution
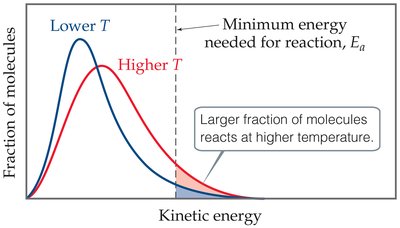
Increasing temperature raises the average kinetic energy of molecules, increasing both the frequency of collisions and the fraction of molecules with enough energy to react.
Boltzmann Distribution: Shows the spread of molecular kinetic energies at different temperatures.
Fraction of Reactive Molecules: At higher temperatures, a larger fraction of molecules have KE ≥ Ea.
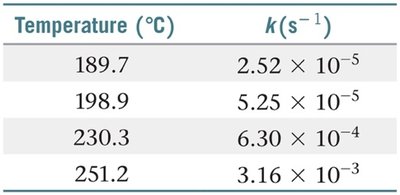
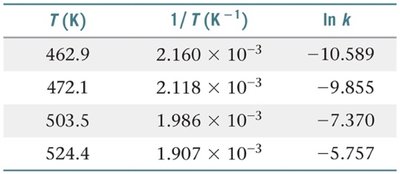
The Arrhenius Equation
The Arrhenius equation quantitatively relates the rate constant of a reaction to temperature and activation energy.
Equation:
Frequency Factor (A): Represents the frequency of collisions with correct orientation; influenced by reactant structure.
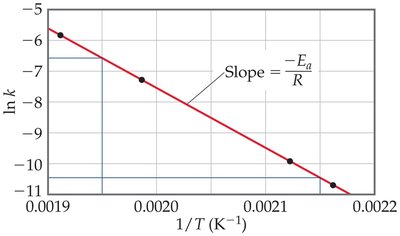
Graphical Analysis: Taking the natural logarithm yields , which is linear in vs. .
Two-Point Form:
Reaction Mechanisms
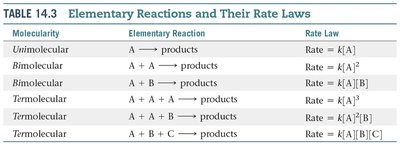
Elementary Reactions and Molecularity
A reaction mechanism describes the sequence of elementary steps by which a chemical reaction occurs. Each elementary reaction involves a specific number of molecules, called its molecularity.
Elementary Reaction: A single molecular event.
Molecularity: Number of reactant molecules involved in an elementary step (unimolecular, bimolecular, termolecular).
Molecularity | Elementary Reaction | Rate Law |
|---|---|---|
Unimolecular | A → products | Rate = k[A] |
Bimolecular | A + A → products | Rate = k[A]2 |
Bimolecular | A + B → products | Rate = k[A][B] |
Termolecular | A + A + A → products | Rate = k[A]3 |
Termolecular | A + A + B → products | Rate = k[A]2[B] |
Termolecular | A + B + C → products | Rate = k[A][B][C] |
Multistep Mechanisms and Intermediates
Most reactions occur via a series of elementary steps. Intermediates are species produced in one step and consumed in another, not appearing in the overall reaction.
Intermediates: Formed and consumed during the mechanism; do not appear in the overall equation.
Catalysts: Added to the reaction, used up in one step and regenerated in another; speed up reactions without being consumed.
Rate Law for Mechanisms: For elementary reactions, the rate law can be written directly from the balanced equation.
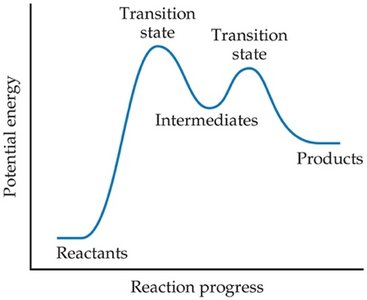
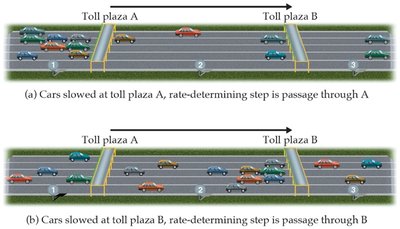
Rate-Determining Step
The slowest step in a reaction mechanism is called the rate-determining step. It controls the overall reaction rate.
Rate-Determining Step: The slowest elementary step; limits the overall rate.
Overall Rate Law: Usually equals the rate law for the slowest step.
Experimental Rate Laws and Mechanism Consistency
Rate laws must be written in terms of reactants, products, and catalysts, but not intermediates. If a proposed mechanism does not match the experimental rate law, it must be rejected.
Substitution for Intermediates: Use equilibrium relationships to express intermediate concentrations in terms of measurable species.
Mechanism Validation: Only mechanisms consistent with experimental rate laws are accepted.
Catalysis
Role and Types of Catalysts
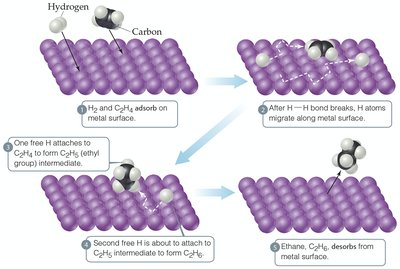
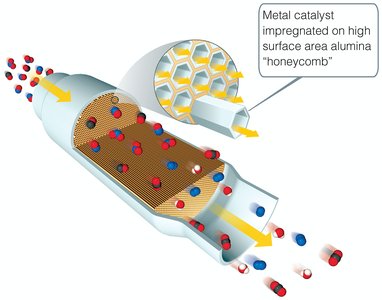
Catalysts increase reaction rates by providing alternative pathways with lower activation energy or by improving reactant orientation. They are not consumed in the overall reaction.
Homogeneous Catalysts: Same phase as reactants.
Heterogeneous Catalysts: Different phase from reactants.
Effect on Activation Energy: Catalysts lower Ea, increasing the rate constant k.
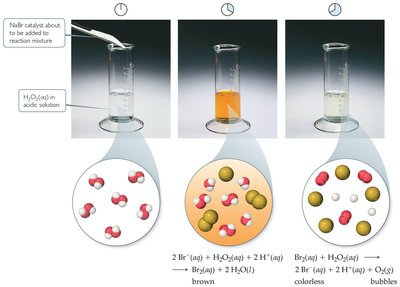
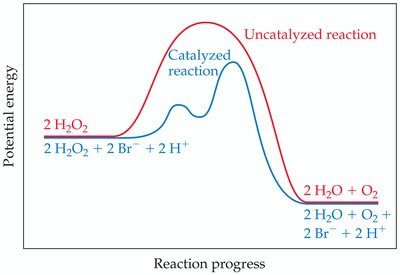
Enzymes: Biological Catalysts
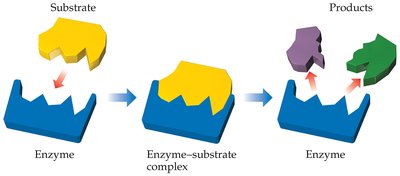
Enzymes are highly efficient biological catalysts, usually proteins, that accelerate biochemical reactions with high specificity.
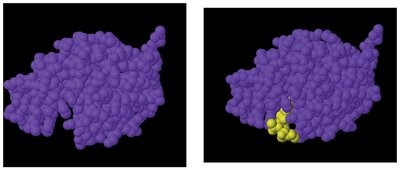
Substrate Specificity: The reactant (substrate) binds to the enzyme's active site, which matches its shape.
Mechanism: Enzyme-substrate complex forms, reaction occurs, and products are released.
Efficiency: Enzymes are much more efficient than non-biological catalysts.