 Back
Back7.2 Separable Differential Equations
Study Guide - Smart Notes

7.2 Separable Differential Equations
Introduction to Separable Differential Equations
Separable differential equations are a class of first-order differential equations that can be written in the form \( \frac{dy}{dx} = g(x)h(y) \). These equations can be solved by separating the variables and integrating both sides.
Definition: A differential equation is separable if it can be expressed as \( \frac{dy}{dx} = g(x)h(y) \).
Solution Method: Rearrange to \( \frac{1}{h(y)}dy = g(x)dx \) and integrate both sides.
Example 1: Solving a Separable Differential Equation
Consider the equation \( \frac{dy}{dx} = ky \) with initial condition \( y(0) = 100 \).
Separate variables: \( \frac{1}{y}dy = kdx \).
Integrate both sides: \( \int \frac{1}{y}dy = \int kdx \).
Solution: \( \ln|y| = kx + C \) → \( y = 100e^{kx} \) (using the initial condition).
Applications of Separable Differential Equations
Separable differential equations are widely used in modeling real-world phenomena such as population growth, radioactive decay, and heat transfer.
Unlimited Population Growth: See example 3 (not shown here).
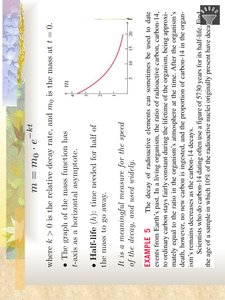
Radioactive Decay: Let \( m \) be the mass of radioactive material at time \( t \). The decay is modeled by \( \frac{dm}{dt} = -km \).
Radioactive Decay and Half-Life
The solution to the radioactive decay equation is \( m = m_0 e^{-kt} \), where \( m_0 \) is the initial mass and \( k \) is the decay constant.
Half-life (\( t_{1/2} \)): The time required for half of the substance to decay. It is given by \( t_{1/2} = \frac{\ln 2}{k} \).
The graph of the function \( m(t) \) is a decreasing exponential curve approaching zero as \( t \) increases.
Example: Calculating Decay Constant and Time
Given a half-life of 5730 years, find \( k \) and solve for the time when \( m = 0.9m_0 \).
\( k = \frac{\ln 2}{5730} \)
\( 0.9m_0 = m_0 e^{-kt} \Rightarrow t = -\frac{1}{k}\ln(0.9) \approx 871 \) years

Heat Transfer: Newton's Law of Cooling
Newton's Law of Cooling describes the rate at which an object changes temperature through heat exchange with its environment. If \( H \) is the temperature at time \( t \) and \( H_s \) is the constant surrounding temperature, then:
\( \frac{dH}{dt} = -k(H - H_s) \)
Let \( y = H - H_s \), then \( \frac{dy}{dt} = -ky \), which is a separable equation.
Solution: \( H = H_s + (H_0 - H_s)e^{-kt} \), where \( H_0 \) is the initial temperature.

Example: Cooling of a Hot Object
A household egg at 90°C is put in a bath of 10°C water. After 5 minutes, the temperature is measured. The time to reach 20°C is found using the solution above.
Apply the formula \( H = H_s + (H_0 - H_s)e^{-kt} \) with the given values.

Practice Problems
Solve each differential equation:
\( \frac{dy}{dx} = (3y^2 + 1) \)
\( \frac{dy}{dx} = xy \)
\( \frac{dy}{dx} = x^2 - 2y \)
