 Back
BackActive Transport and Energetics of Membrane Transport in Cells
Study Guide - Smart Notes

Active Transport: Protein-Mediated Movement Up the Gradient
Overview of Active Transport
Active transport is a fundamental cellular process that enables the movement of solutes against their concentration gradients, away from equilibrium. Unlike facilitated diffusion, which moves molecules down a gradient, active transport requires energy and exhibits intrinsic directionality.
Facilitated diffusion: Moves molecules down a concentration gradient, toward equilibrium.
Active transport: Moves solutes up a concentration gradient, away from equilibrium, requiring energy input.
Diffusion: Nondirectional; active transport: Directional.
Coupling of Active Transport to Energy Sources
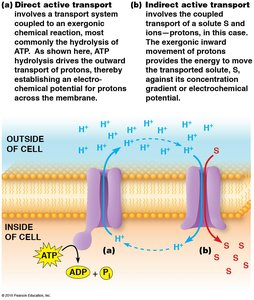
Active transport can be directly or indirectly coupled to an energy source. Direct active transport uses ATP hydrolysis, while indirect active transport relies on ion gradients established by primary pumps.
Direct active transport: ATP hydrolysis directly powers solute movement.
Indirect active transport: Utilizes ion gradients (e.g., Na+ or H+) established by ATP-driven pumps to drive the transport of other solutes.
Types of Transport ATPases
Classification of Transport ATPases
Transport ATPases are specialized proteins that facilitate direct active transport. Four main types have been identified, each with distinct structure, mechanism, location, and roles:
P-type ATPases: e.g., Na+/K+ pump
V-type ATPases: Proton pumps in organelles (vacuoles, lysosomes, Golgi)
F-type ATPases: Proton pumps, includes ATP synthases (reversible)
ABC-type ATPases: Diverse roles, including transport of various molecules
V-Type ATPases
V-type ATPases pump protons into organelles, creating an acidic environment essential for organelle function. They consist of an integral membrane component and a peripheral component.
Found in vacuoles, vesicles, lysosomes, endosomes, and Golgi complex.
Establishes acidic conditions inside organelles.
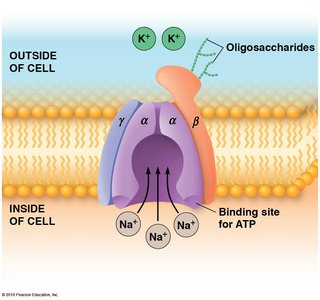
Direct Active Transport: The Na+/K+ Pump
Maintaining Electrochemical Ion Gradients
The Na+/K+ ATPase is crucial for maintaining ion gradients across the plasma membrane of animal cells. In neurons, the [K+] inside/[K+] outside ratio is about 30:1, while [Na+] inside/[Na+] outside is around 0.08:1. These gradients are essential for nerve impulse transmission and coupled transport.
Na+/K+ pump: Moves three Na+ out for every two K+ in.
Requires energy from ATP hydrolysis.
Responsible for asymmetric ion distribution.
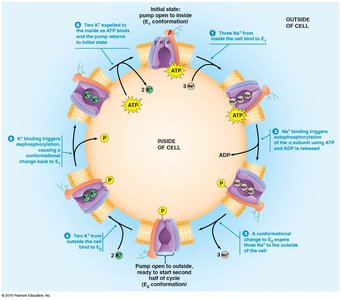
Allosteric Mechanism of the Na+/K+ Pump
The Na+/K+ pump operates via two conformational states:
E1 conformation: Open to the inside, high affinity for Na+.
E2 conformation: Open to the outside, high affinity for K+.
Indirect Active Transport (Secondary Active Transport)
Mechanism and Examples
Indirect active transport is powered by ion gradients, not ATP hydrolysis. The inward transport of molecules up their gradients is coupled to the inward movement of Na+ (animals) or H+ (plants, fungi, bacteria) down their gradients.
Most cells pump Na+ or H+ out, creating a gradient.
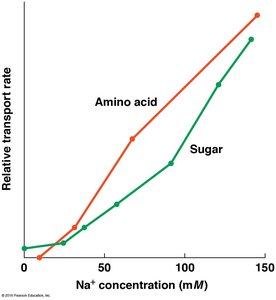
High extracellular Na+ drives uptake of sugars and amino acids.
ATP is indirectly involved via the primary pump.
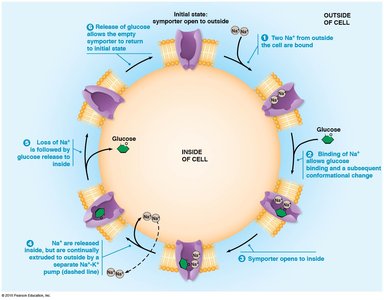
Sodium Symport and Glucose Uptake
Some cells use a Na+/glucose symporter to import glucose against its concentration gradient, especially in intestinal cells.
Glucose and amino acids are taken up even when their concentrations are lower outside the cell.
Symporters couple glucose uptake to Na+ influx.
Energetics of Transport
Transport events are energy transactions. For uncharged solutes, only the concentration gradient matters; for charged solutes, both concentration and electrical potential are relevant.
Uncharged solutes: ΔG depends on concentration gradient.
Charged solutes: ΔG depends on concentration gradient and membrane potential (Vm).
Energetics of Transport: Calculating ΔG
Uncharged Solutes
For solutes with no net charge, the free energy change (ΔG) for inward transport is calculated as:
If [S] inside < [S] outside, ΔG is negative (exergonic).
If [S] inside > [S] outside, ΔG is positive (endergonic).
Formula:
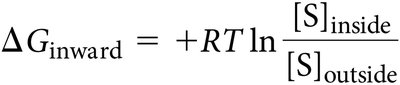
Example: Lactose Uptake
Calculating the energy requirement for inward transport of lactose:

Charged Solutes
For charged solutes, the membrane potential (Vm) must be considered. The formula is:
z = charge of solute
F = Faraday constant
Vm = membrane potential
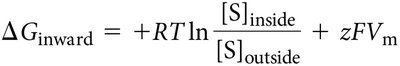
Example: Chloride Ion Uptake
For a nerve cell with [Cl–] inside = 50 mM, [Cl–] outside = 100 mM, and Vm = –60 mV:

Summary Table: Types of Active Transport
Type | Energy Source | Example | Mechanism |
|---|---|---|---|
Direct Active Transport | ATP hydrolysis | Na+/K+ pump | ATPase directly moves ions |
Indirect Active Transport | Ion gradient (Na+, H+) | Na+/glucose symporter | Coupled transport using ion gradient |
Additional info: The notes above expand on the original content by providing definitions, formulas, and examples for clarity and completeness, suitable for cell-biology college students.
