 Back
BackAerobic Respiration and ATP Synthesis: Catabolic Pathways and Energy Metabolism in Cells
Study Guide - Smart Notes

Catabolism and Energy Extraction in Cells
Overview of Metabolism
Metabolism encompasses all chemical reactions within a cell or organism, divided into two main categories: anabolism (synthesis of cellular components) and catabolism (breakdown of cellular molecules). Catabolic pathways are crucial for extracting energy from nutrients, which is then used to drive cellular processes.
Anabolism: Builds complex molecules from simpler ones, requiring energy input.
Catabolism: Breaks down complex molecules into simpler ones, releasing energy.
Energy Extraction: Catabolism is the primary means by which cells extract energy, especially through the breakdown of glucose and other macromolecules.

Catabolic Pathways: Glycolysis, Fermentation, and Aerobic Respiration
Glycolysis and Substrate-Level Phosphorylation
Glycolysis is the initial pathway of glucose catabolism, occurring in the cytosol and resulting in the formation of pyruvate. ATP is generated directly via substrate-level phosphorylation, where a high-energy phosphate group is transferred from a metabolic intermediate to ADP.
Substrate-level phosphorylation: Direct transfer of phosphate to ADP (e.g., from 1,3-bisphosphoglycerate).
Oxidative phosphorylation: Major ATP production method in animals, occurring in mitochondria.
Oxidation and Reduction in Cellular Respiration
Energy extraction involves redox reactions, where electrons are transferred from nutrients to oxygen (eventually). In biochemistry:
Oxidation: Removal of electrons (and hydrogen ions), also called dehydrogenation.
Reduction: Addition of electrons (and hydrogen ions), also called hydrogenation.
External Electron Acceptors and Coenzymes
During catabolism, electrons are transferred to coenzymes such as NAD+ and FAD, forming NADH and FADH2. These reduced coenzymes carry high-energy electrons to the electron transport chain or are recycled during fermentation.
NAD+ and FAD: Common electron acceptors in cellular respiration.
NADH and FADH2: Reduced forms that store energy in their electrons.
Fermentation: Recycles NADH to NAD+ in the absence of oxygen.
Aerobic Respiration: Steps and Mitochondrial Structure
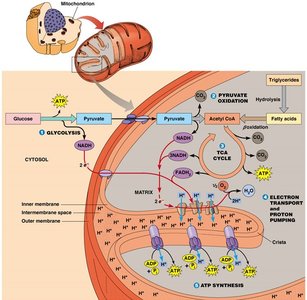
Major Steps of Aerobic Respiration
Aerobic respiration is a multi-step process that occurs mainly in the mitochondria and yields much more ATP than fermentation.
Step | Location | Main Reaction |
|---|---|---|
Glycolysis | Cytosol | Glucose → Pyruvate |
Pyruvate Oxidation | Mitochondrial Matrix | Pyruvate → Acetyl CoA |
TCA Cycle | Mitochondrial Matrix | Acetyl CoA → CO2 |
Electron Transfer | Inner Mitochondrial Membrane | e- flow from coenzymes to O2 |
ATP Synthesis | Inner Mitochondrial Membrane | Proton motive force drives ATP synthesis |

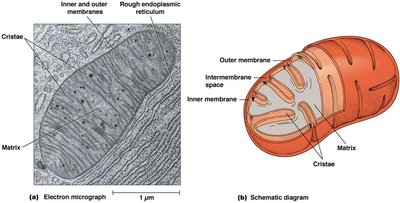
Mitochondrial Structure
The mitochondrion is the main site for aerobic respiration. Its inner membrane forms extensive infolds called cristae to increase surface area for energy production.
Pyruvate Oxidation and the TCA Cycle
Conversion of Pyruvate to Acetyl CoA
Pyruvate, a 3-carbon molecule produced by glycolysis, is converted into a 2-carbon molecule, Acetyl CoA, via oxidative decarboxylation. This process generates one NADH per pyruvate.
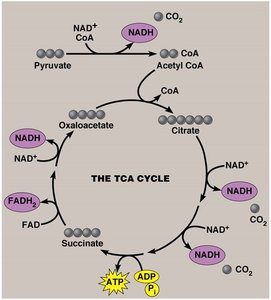
The Tricarboxylic Acid (TCA) Cycle
The TCA cycle (also called the citric acid cycle) is a series of reactions that completes the oxidation of Acetyl CoA to CO2. It generates ATP and stores most of the energy in the form of NADH and FADH2.
Continuous input of Acetyl CoA and release of CO2 maintains the cycle.
One ATP (or GTP) is produced per cycle, but most energy is stored in reduced coenzymes.
Catabolism of Other Macromolecules
Entry of Fats and Proteins into Catabolic Pathways
Other macromolecules such as fats and proteins can also be catabolized for energy. Fatty acids are converted to Acetyl CoA via β-oxidation, and certain amino acids enter the TCA cycle directly or through other pathways.
Fats: Triglycerides are hydrolyzed to glycerol and fatty acids; fatty acids undergo β-oxidation in mitochondria.
Proteins: Hydrolyzed to amino acids; some enter the TCA cycle directly (e.g., alanine, aspartate, glutamate).
Electron Transport Chain and Oxidative Phosphorylation
Electron Transport and Proton Gradient Formation
Electrons from NADH and FADH2 are transferred through a series of protein complexes (I-IV) in the inner mitochondrial membrane. This transfer is coupled to the pumping of protons (H+) across the membrane, creating a proton gradient (proton motive force, pmf).
Complexes I-IV: Sequentially transfer electrons and pump protons.
Proton Gradient: Results in a higher concentration of H+ in the intermembrane space.

Chemiosmotic Coupling Model
The chemiosmotic hypothesis (proposed by Peter Mitchell) explains that the energy from electron transport is used to generate a proton gradient, which then drives ATP synthesis as protons flow back into the matrix through ATP synthase.
Proton Motive Force (pmf): Electrochemical potential across the inner mitochondrial membrane.
ATP Synthase: Enzyme complex that synthesizes ATP using the energy from proton flow.
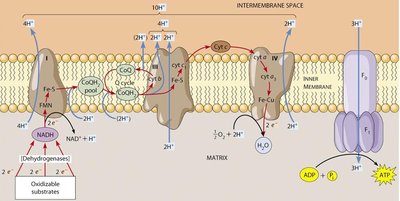
ATP Synthase Structure and Mechanism
Structure of ATP Synthase (F0F1 Complex)
ATP synthase consists of two main components: F0 (membrane-embedded proton channel) and F1 (catalytic ATP synthesis unit). Proton flow through F0 causes rotation, which induces conformational changes in F1 to synthesize ATP.
F0: Static (a, b subunits) and mobile (c subunits) components form the proton channel.
F1: Mobile (γ, ε subunits) and static (3α, 3β, δ subunits) components catalyze ATP synthesis.
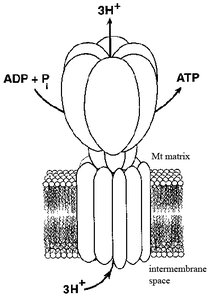
Binding-Change Model for ATP Synthesis
The binding-change model describes how the rotation of the γ subunit within F1 causes the three β subunits to cycle through three conformations: open (O), loose (L), and tight (T). This cycling allows ADP and Pi to bind, ATP to be synthesized, and ATP to be released.
Open (O): ADP and Pi bind or ATP is released.
Loose (L): ADP and Pi are held in place.
Tight (T): ATP is synthesized from ADP and Pi.
ATP Yield and Efficiency of Aerobic Respiration
Counting ATP Production
The complete oxidation of one glucose molecule through glycolysis, the TCA cycle, and oxidative phosphorylation yields a maximum of 30–38 ATP molecules, depending on the cell type and shuttle systems used.
Glycolysis: 2 ATP (substrate-level), 2 NADH
Pyruvate Oxidation: 2 NADH
TCA Cycle: 2 ATP (GTP), 6 NADH, 2 FADH2
Electron Transport/Oxidative Phosphorylation: Each NADH yields ~2.5–3 ATP, each FADH2 yields ~1.5–2 ATP
Step | ATP Produced |
|---|---|
Glycolysis | 2 |
TCA Cycle (direct) | 2 |
Oxidative Phosphorylation | 28–34 |
Total | 30–38 |
Efficiency of Energy Conversion
The efficiency of aerobic respiration is about 50%, with the rest of the energy lost as heat. This is significantly higher than most man-made machines.
ΔG for glucose oxidation: −686 kcal/mol
ATP hydrolysis: −10 to −14 kcal/mol
Energy conserved: ~300–380 kcal/mol (for 30–38 ATP)
Uncoupling of Electron Transport and ATP Synthesis
Uncoupling Agents and Brown Fat
Certain agents (e.g., 2,4-dinitrophenol, DNP) and proteins (e.g., thermogenin in brown fat) can uncouple electron transport from ATP synthesis, causing energy to be released as heat instead of being stored as ATP. This is especially important in newborn mammals and hibernating animals for thermogenesis.
Thermogenin: Uncoupling protein in brown fat mitochondria, allows protons to re-enter the matrix without ATP synthesis, generating heat.
Physiological significance: Helps maintain body temperature in infants and hibernating animals who cannot shiver or move to generate heat.
Summary Table: Key Steps and Energy Yield in Aerobic Respiration
Pathway | Main Product(s) | ATP Yield |
|---|---|---|
Glycolysis | 2 Pyruvate, 2 NADH | 2 |
Pyruvate Oxidation | 2 Acetyl CoA, 2 NADH | 0 |
TCA Cycle | 4 CO2, 6 NADH, 2 FADH2, 2 ATP | 2 |
Oxidative Phosphorylation | H2O, ATP | 28–34 |
Additional info: The actual ATP yield varies depending on the shuttle systems used to transport electrons from cytosolic NADH into mitochondria and the efficiency of the electron transport chain in different tissues.
