 Back
BackBioenergetics and Glycolysis: Cellular Energy Flow and Metabolism
Study Guide - Smart Notes

Bioenergetics: The Flow of Energy in the Cell
Introduction to Cellular Energy
Energy is fundamental for all cellular processes, enabling biosynthesis, movement, transport, temperature regulation, and bioluminescence. Cells acquire energy from their environment and convert it into usable forms, primarily ATP.
Biosynthesis: Formation of new chemical bonds (anabolic pathways).
Cellular and Subcellular Movement: Mechanical work, such as muscle contraction or vesicle transport.
Transport of Biomolecules: Movement against concentration gradients.
Transport of Ions: Movement against electrochemical gradients.
Regulation of Body Temperature: Heat generation in homeothermic organisms.
Bioluminescence: Production of light by organisms (e.g., luciferase in fireflies).

Phototrophs versus Chemotrophs
Energy Acquisition Strategies
Cells are classified based on their primary energy source: phototrophs use light, while chemotrophs use chemical compounds.
Phototrophs: Capture light energy from the sun and convert it into chemical energy, stored as ATP. Example: plants, algae, and some bacteria.
Chemotrophs: Obtain energy by oxidizing chemical bonds in organic or inorganic molecules to produce ATP. Example: animals, fungi, and many bacteria.
Photosynthesis (Phototrophs):
Cellular Respiration (Chemotrophs):


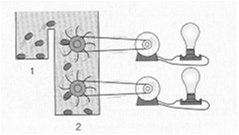
Thermodynamics in Biological Systems
The Laws of Thermodynamics
Bioenergetics applies thermodynamic principles to biological systems, describing how energy is transferred and transformed in cells.
First Law (Conservation of Energy): Energy can be converted from one form to another but cannot be created or destroyed.
Second Law (Thermodynamic Spontaneity): In every physical or chemical change, the universe tends toward greater disorder (entropy).
Order: High free energy; Chaos: Low free energy.
Free Energy Change (ΔG)
ΔG measures the spontaneity of a chemical reaction:
Spontaneous Reaction: ΔG < 0 (exergonic, energy-releasing)
Non-spontaneous Reaction: ΔG > 0 (endergonic, energy-requiring)
At Equilibrium: ΔG = 0
Equation:
Equilibrium Constant (Keq)
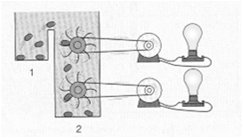
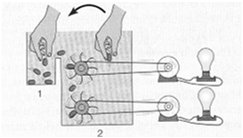
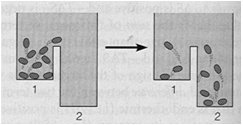
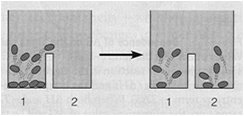
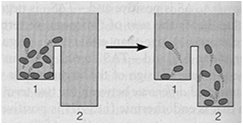
The equilibrium constant () is the ratio of product concentrations to reactant concentrations at equilibrium. At equilibrium, there is no net change in concentrations, and ΔG = 0. Cells, however, maintain a steady state, not equilibrium, to remain alive.
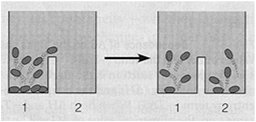
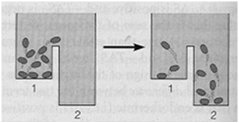
Steady State versus Equilibrium
Cells operate at a steady state, where reactions move toward equilibrium but never reach it due to continuous energy input. A cell at equilibrium is dead.
Summary Table: ΔG and Reaction Types
Reaction Type | ΔG Value |
|---|---|
Endergonic | Positive |
Exergonic | Negative |
Spontaneous | Negative |
At Equilibrium | Zero |
At Steady State | Negative |
Enzymes and Activation Energy
Role of Enzymes in Biochemical Reactions
Enzymes are biological catalysts that accelerate reactions by lowering the activation energy, without altering the ΔG of the reaction.
Decrease Activation Energy: Enzymes provide an alternative pathway with lower activation energy.
Do Not Change ΔG: The overall free energy change remains the same.
ATP: The Energy Currency of the Cell
ATP Synthesis and Hydrolysis
ATP (adenosine triphosphate) is the primary energy carrier in cells. Its synthesis stores energy, while hydrolysis releases energy for cellular work.
ATP Synthesis: Energy is stored by forming high-energy phosphoanhydride bonds.
ATP Hydrolysis: Energy is released by breaking these bonds, powering cellular processes.


ATP/ADP System
The ATP/ADP system allows cells to conserve and release energy efficiently. Chemotrophs store energy from nutrient catabolism in ATP's high-energy bonds.
Glycolysis: The Central Pathway of Glucose Catabolism
Overview of Glycolysis
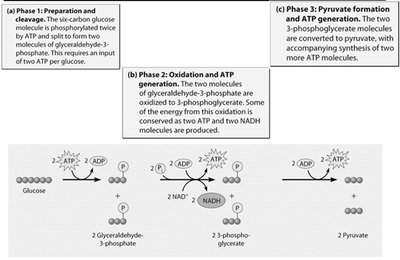
Glycolysis is a universal metabolic pathway that splits glucose into two molecules of pyruvate, generating ATP and NADH. It consists of an energy investment phase and an energy payoff phase.
Energy Investment Phase: 2 ATP are consumed.
Energy Payoff Phase: 4 ATP and 2 NADH are produced.
Net Gain: 2 ATP and 2 NADH per glucose molecule.
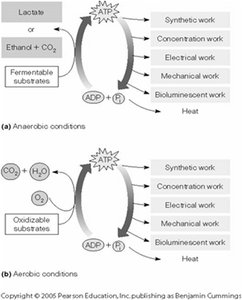
Fate of Pyruvate: Aerobic vs. Anaerobic Conditions
The fate of pyruvate depends on oxygen availability:
Aerobic Respiration: Pyruvate is fully oxidized to CO2 and H2O, generating more ATP (e.g., in humans).
Anaerobic Fermentation: Pyruvate is converted to lactate (in animals) or ethanol and CO2 (in yeast), regenerating NAD+ for glycolysis.


Carbohydrate Entry into Glycolysis
Alternative Substrates and Storage Carbohydrates
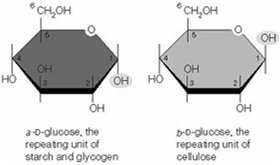
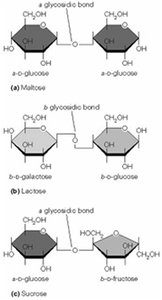
Other sugars and storage carbohydrates (e.g., glycogen, starch) are converted into glycolytic intermediates before entering the main pathway. The linkage between monosaccharides is a glycosidic bond.
Monosaccharides: Single sugar units (e.g., glucose).
Disaccharides and Polysaccharides: Multiple sugar units linked by glycosidic bonds. Starch and glycogen are storage polysaccharides with α-glycosidic bonds.
Substrate-Level Phosphorylation
ATP is generated during glycolysis by substrate-level phosphorylation, where a high-energy phosphate group is directly transferred to ADP from a metabolic intermediate (e.g., 1,3-bisphosphoglycerate). This is distinct from oxidative phosphorylation, which produces most ATP in aerobic organisms.
Summary Table: Standard Free Energies of Hydrolysis
Phosphorylated Compound | Hydrolysis Reaction | ΔG°' (kcal/mol) |
|---|---|---|
Phosphoenolpyruvate (PEP) | PEP + H2O → pyruvate + Pi | -14.8 |
1,3-bisphosphoglycerate | 1,3-bisphosphoglycerate + H2O → 3-phosphoglycerate + Pi | -11.8 |
Phosphocreatine | Phosphocreatine + H2O → creatine + Pi | -10.3 |
Adenosine triphosphate (ATP) | ATP + H2O → ADP + Pi | -7.3 |
Glucose-1-phosphate | Glucose-1-phosphate + H2O → glucose + Pi | -5.0 |
Glucose-6-phosphate | Glucose-6-phosphate + H2O → glucose + Pi | -3.3 |
Glycerol phosphate | Glycerol phosphate + H2O → glycerol + Pi | -2.2 |

Key Takeaways
Energy flow in cells is governed by the laws of thermodynamics and mediated by ATP.
Glycolysis is a central pathway for energy extraction from glucose, with alternative fates for pyruvate depending on oxygen availability.
Enzymes lower activation energy but do not change the overall free energy change (ΔG) of reactions.
Carbohydrates enter glycolysis after conversion to intermediates, and ATP is generated by substrate-level phosphorylation.
