 Back
BackCH 5 - Bioenergetics: The Flow of Energy in the Cell (Ch. 5 Study Notes)
Study Guide - Smart Notes

Bioenergetics: The Flow of Energy in the Cell
Overview of Cellular Energy Needs
All living cells require a continuous supply of energy to sustain life. This energy is essential for driving chemical and physical changes necessary for cellular function. Cells have four essential needs:
Molecular building blocks
Chemical catalysts (enzymes)
Information to guide activities
Energy to drive reactions and processes
Categories of Cellular Work
Energy in cells is used for six main types of work:
Synthetic work: Formation of new chemical bonds and synthesis of molecules, essential for growth and maintenance.
Mechanical work: Physical changes in position or orientation, such as muscle contraction or movement of cilia/flagella.
Concentration work: Accumulation or removal of substances to create concentration gradients.
Electrical work: Movement of ions across membranes, generating membrane potentials.
Generation of heat: Maintaining body temperature in homeotherms.
Generation of light: Bioluminescence in certain organisms.
Synthetic Work
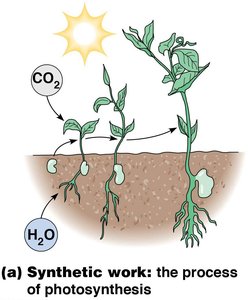
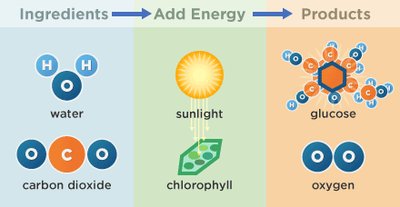
Synthetic work involves the biosynthesis of macromolecules, such as proteins, nucleic acids, and polysaccharides. Photosynthesis is a prime example, where plants convert carbon dioxide and water into glucose using sunlight.
Mechanical Work
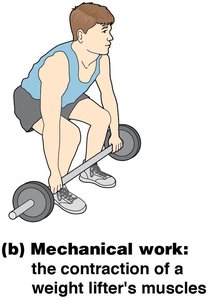
Mechanical work refers to movement, such as muscle contraction, cell division, or the beating of cilia and flagella. This type of work is powered by the hydrolysis of ATP.
Concentration Work
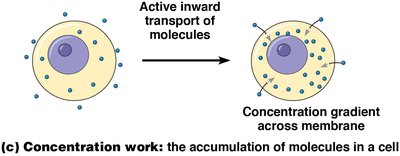
Concentration work is the active transport of molecules against their concentration gradients, such as the accumulation of nutrients or removal of waste products.
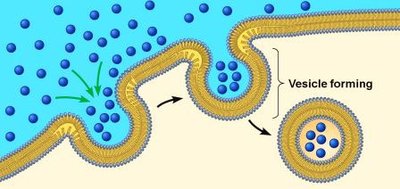
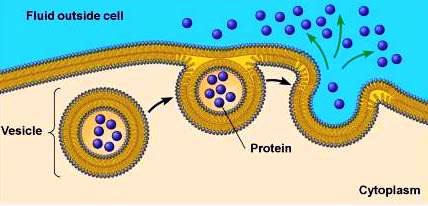
Electrical Work
Electrical work involves the movement of ions (e.g., Na+, K+) across membranes, creating electrical potentials essential for processes like nerve impulse transmission and ATP synthesis.
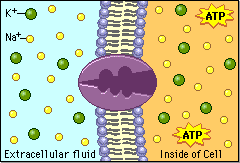
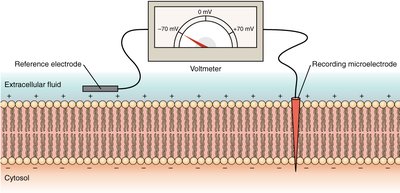
Heat Production
Some organisms use energy to generate heat, which is crucial for maintaining body temperature in homeotherms (warm-blooded animals).

Bioluminescence
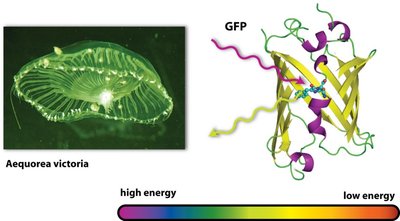
Bioluminescence is the production of light by living organisms, often through the reaction of ATP with luminescent compounds. Green fluorescent protein (GFP) from Aequorea victoria is widely used in cell biology research.
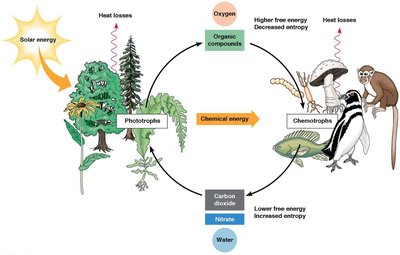
Energy Sources for Life
Life on Earth is sustained by energy from sunlight (phototrophs) or the oxidation of organic compounds (chemotrophs). Organisms are also classified by their carbon source: autotrophs (self-feeders) use CO2, while heterotrophs require organic carbon.
Energy Flow in the Biosphere
Energy flows unidirectionally from the sun through the biosphere, while matter cycles between phototrophs and chemotrophs. Photosynthesis stores energy in reduced compounds, which is later released by oxidation in consumers.
Thermodynamics and Bioenergetics
Bioenergetics applies the laws of thermodynamics to biological systems, governing how energy is transformed and utilized in cells.
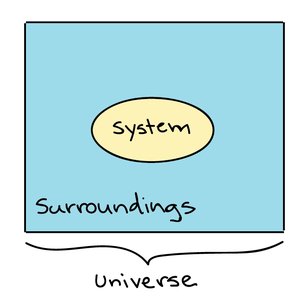
Systems and Surroundings
A system is the part of the universe under study, while the surroundings are everything else. Biological systems are typically open, exchanging energy and matter with their environment.
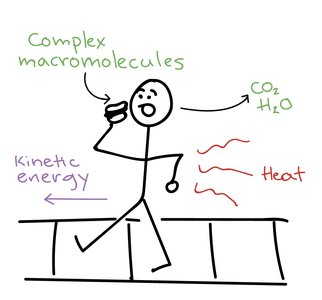
Heat and Work
Energy exchange between a system and its surroundings occurs as heat or work. Heat has limited utility in isothermal biological systems, so most cellular energy is used to perform work.
Quantifying Energy Change
Energy changes are measured in calories (cal) or joules (J). One calorie is the energy required to raise 1 gram of water by 1°C at 1 atm pressure. 1 kcal = 1000 cal; 1 J = 0.239 cal.
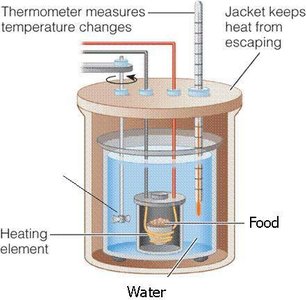
The First Law of Thermodynamics
The first law (law of conservation of energy) states that energy cannot be created or destroyed, only transformed. The total energy of the universe remains constant.
Internal Energy and Enthalpy
Internal energy (E): Total energy stored in a system.
Change in internal energy (ΔE):
Enthalpy (H): Heat content,
In biological systems,
The Second Law of Thermodynamics
The second law states that the universe tends toward greater disorder (entropy). This law predicts the direction of spontaneous processes and the amount of energy released.
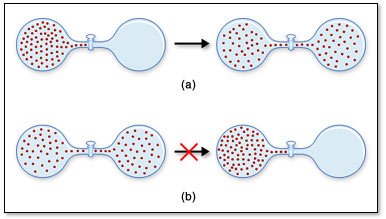
Entropy (S)
Entropy is a measure of randomness or disorder.
Spontaneous processes increase the entropy of the universe ().
Free Energy (G)
Free energy (G): Energy available to do work.
Free energy change (ΔG):
If , the reaction is exergonic (spontaneous).
If , the reaction is endergonic (non-spontaneous).
Biological Examples
Glucose oxidation: , kcal/mol (exergonic)
CO2 reduction: , kcal/mol (endergonic)
Equilibrium and Free Energy
The equilibrium constant () indicates the direction of a reaction. At equilibrium, . The relationship between and is given by:
Where is the gas constant (1.987 cal/mol·K) and is temperature in Kelvin.
Standard Free Energy Change ()
Measured under standard conditions: 25°C, 1 atm, pH 7.0, 1 M concentrations.
Directly related to :
Summary Table: Thermodynamic Parameters
Parameter | Definition | Significance |
|---|---|---|
ΔE | Change in internal energy | Total energy change in system |
ΔH | Change in enthalpy | Heat absorbed or released |
ΔS | Change in entropy | Change in disorder/randomness |
ΔG | Change in free energy | Predicts spontaneity of reaction |
Key Equations
Important Concepts
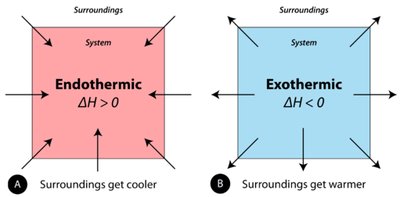
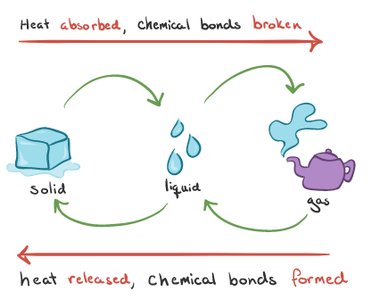
Endothermic vs. Endergonic: Endothermic reactions absorb heat (ΔH > 0), while endergonic reactions require energy input (ΔG > 0).
Exothermic vs. Exergonic: Exothermic reactions release heat (ΔH < 0), while exergonic reactions release free energy (ΔG < 0).
Not all endothermic reactions are endergonic, and not all exothermic reactions are exergonic.
Limitations of ΔG
ΔG indicates whether a reaction is thermodynamically possible and how much energy would be released, but it does not provide information about the reaction rate or mechanism. Enzymes (biological catalysts) are required to lower activation energy and allow reactions to proceed at biologically relevant rates.
Summary
Energy flow drives all life processes, coupling the flow of energy with the cycling of matter.
Thermodynamic principles govern the direction and efficiency of cellular reactions.
Cells maintain a steady state far from equilibrium, enabling life to persist.
