 Back
BackCarbohydrates: Structure, Diversity, and Biological Roles
Study Guide - Smart Notes

Overview of Carbohydrates
Introduction to Carbohydrates
Carbohydrates are one of the four major classes of biomolecules essential for life. They serve as energy sources, structural components, and information carriers in biological systems. Carbohydrates are classified based on their complexity into monosaccharides, disaccharides, oligosaccharides, and polysaccharides.
Monosaccharides: The simplest carbohydrates, consisting of single sugar units.
Disaccharides: Composed of two monosaccharide units linked by glycosidic bonds.
Oligosaccharides: Short chains of monosaccharide units.
Polysaccharides: Long chains of monosaccharide units, which may be branched or unbranched.
Functions of Carbohydrates:
Energy storage and supply (e.g., glucose, starch, glycogen)
Structural roles (e.g., cellulose in plant cell walls, glycosaminoglycans in extracellular matrix)
Information carriers (e.g., cell-cell recognition via glycoproteins and glycolipids)
Chemical Components of Carbohydrates
Monosaccharide Structure and Diversity
Monosaccharides are the building blocks of carbohydrates. They contain three to seven carbon atoms and are classified by the number of carbons:
Triose (3 carbons)
Tetrose (4 carbons)
Pentose (5 carbons)
Hexose (6 carbons)
Heptose (7 carbons)
Monosaccharides contain a carbonyl group and multiple hydroxyl groups. They are further classified as aldoses (aldehyde group) or ketoses (ketone group).
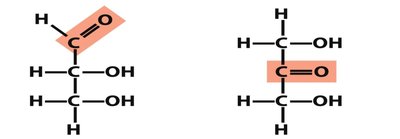
Isomerism in Monosaccharides
Monosaccharides exhibit several types of isomerism:
Constitutional isomers: Same molecular formula, different connectivity of atoms.
Stereoisomers: Same connectivity, different spatial arrangement.
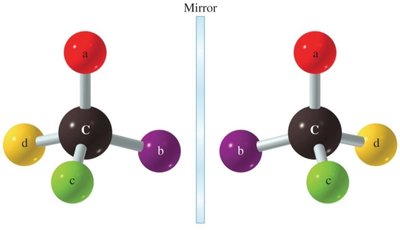
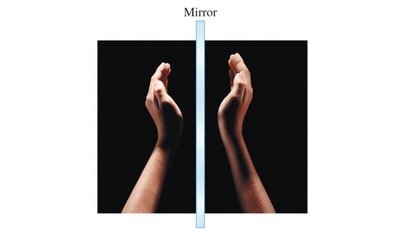
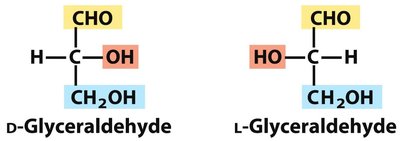
Enantiomers: Non-superimposable mirror images (D- and L- forms).
Diastereomers: Stereoisomers that are not mirror images.
Epimers: Differ in configuration around only one specific carbon atom.
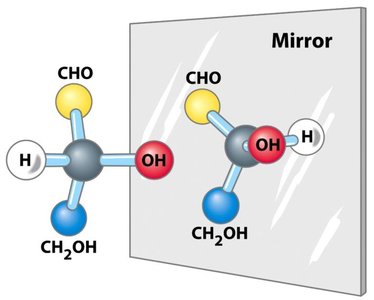
Fischer and Haworth Projections
The structure of monosaccharides is often depicted using Fischer projections (linear form) and Haworth projections (cyclic form). The Fischer projection is a two-dimensional representation, while the Haworth projection shows the ring structure.
Cyclic Forms of Monosaccharides
Monosaccharides with five or more carbons can cyclize to form ring structures. Glucose forms a six-membered ring (pyranose), while fructose forms a five-membered ring (furanose). Cyclization creates a new chiral center at the anomeric carbon, resulting in α and β anomers.
α-anomer: Hydroxyl group at the anomeric carbon is below the plane of the ring.
β-anomer: Hydroxyl group at the anomeric carbon is above the plane of the ring.
Conformations of Monosaccharide Rings
Monosaccharide rings are not planar. Six-membered rings (pyranoses) can adopt chair or boat conformations, with the chair form being more stable due to reduced steric hindrance.
Modified Monosaccharides
Biochemical Modifications
Monosaccharides can be chemically modified to increase their functional diversity:
Deoxysugars: Ribose (RNA) and deoxyribose (DNA)
Sugar phosphates: Important intermediates in metabolism (e.g., glucose 6-phosphate)
Amino sugars: Hydroxyl group replaced by an amino group, often acetylated
Glycosidic Bonds and Disaccharides
Formation of Glycosidic Bonds
Monosaccharides can be linked to other molecules via glycosidic bonds:
N-glycosidic bond: Between the anomeric carbon and a nitrogen atom (e.g., nucleotides)
O-glycosidic bond: Between the anomeric carbon and an oxygen atom (e.g., disaccharides)
Common Disaccharides
Disaccharides are composed of two monosaccharides joined by an O-glycosidic bond. Examples include:
Maltose: Two glucose units (α-1,4 linkage)
Sucrose: Glucose and fructose (α-1,2 linkage)
Lactose: Glucose and galactose (β-1,4 linkage)
Lactose intolerance results from a deficiency in the enzyme lactase, leading to gastrointestinal symptoms.
Polysaccharides
Structure and Function of Polysaccharides
Polysaccharides are long chains of monosaccharide units and serve as energy storage or structural molecules.
Starch: Main storage polysaccharide in plants; composed of amylose (unbranched, α-1,4) and amylopectin (branched, α-1,4 and α-1,6)
Glycogen: Main storage polysaccharide in animals; highly branched (α-1,4 and α-1,6)
Cellulose: Structural component in plant cell walls; linear chains of glucose (β-1,4)
Glycosaminoglycans (GAGs): Linear polysaccharides of repeating disaccharide units, important in the extracellular matrix
Glycoproteins and Biological Information
Glycoproteins and Glycoconjugates
Carbohydrates can be covalently attached to proteins (glycoproteins) or lipids (glycolipids). Glycoproteins are found on the cell surface and play roles in cell-cell recognition, signaling, and protection from degradation.
O-linked glycosylation: Carbohydrate attached to the oxygen atom of serine or threonine
N-linked glycosylation: Carbohydrate attached to the nitrogen atom of asparagine
Protein glycosylation occurs in the endoplasmic reticulum and Golgi apparatus.
Biological Roles of Carbohydrates
Energy storage (starch, glycogen)
Structural support (cellulose, GAGs)
Cell-cell communication (glycoproteins, glycolipids)
Recognition sites for pathogens (e.g., influenza virus binding to cell-surface carbohydrates)
Summary Table: Types of Carbohydrates and Their Functions
Type | Structure | Function | Example |
|---|---|---|---|
Monosaccharide | Single sugar unit | Energy source, building block | Glucose, fructose |
Disaccharide | Two sugar units | Energy transport | Sucrose, lactose |
Oligosaccharide | Few sugar units | Cell recognition | Blood group antigens |
Polysaccharide | Many sugar units | Energy storage, structure | Starch, glycogen, cellulose |
Key Points
Monosaccharides are the simplest carbohydrates and can exist as aldoses or ketoses.
Isomerism increases the diversity of monosaccharides, with D-isomers being most common in nature.
Monosaccharides cyclize to form pyranose (6-membered) or furanose (5-membered) rings, creating α and β anomers.
Disaccharides and polysaccharides are formed by glycosidic bonds between monosaccharides.
Polysaccharides serve as energy storage (starch, glycogen) or structural components (cellulose, GAGs).
Glycoproteins and glycolipids are essential for cell communication and recognition.
